FDA Approvals - Cancer Currents Blog
News on recent approvals of cancer therapies by the Food and Drug Administration. Includes expert comments on how the approval will influence patient care and future research.
-
FDA Approval of KRAS Inhibitor Sotorasib for Lung Cancer Hailed as Milestone
FDA has approved the first KRAS-blocking drug, sotorasib (Lumakras). The approval, which covers the use of sotorasib to treat some patients with advanced lung cancer, sets the stage for other KRAS inhibitors already in development, researchers said.
-
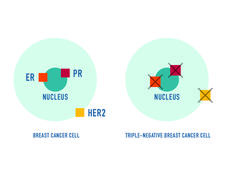
Sacituzumab Govitecan Earns Full Approval for Triple-Negative Breast Cancer
Sacituzumab govitecan (Trodelvy) now has regular FDA approval for people with locally advanced or metastatic triple-negative breast cancer (TNBC). The update follows last year’s accelerated approval of the drug for people with TNBC.
-
FDA Approves BCMA-Targeted CAR T-Cell Therapy for Multiple Myeloma
The Food and Drug Administration has approved idecabtagene vicleucel (Abecma) for some people with multiple myeloma. The approval is based, in part, on a small study in which ide-cel partially or completely shrank tumors in 72% of patients.
-
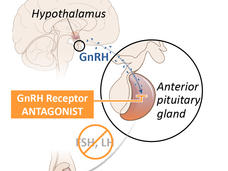
Relugolix Approval Expected to Alter Treatment for Advanced Prostate Cancer
FDA’s recent approval of relugolix (Orgovyx) is expected to affect the treatment of men with advanced prostate cancer. A large clinical trial showed that relugolix was more effective at reducing testosterone levels than another common treatment.
-
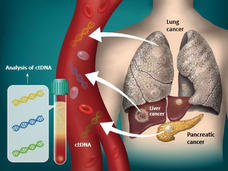
Cancer “Liquid Biopsy” Blood Test Gets Expanded FDA Approval
FDA has expanded the approved uses of the FoundationOne Liquid CDx blood test, known as a liquid biopsy, that can help doctors pick specific treatments for some people with cancer. When used in this way, the test is known as a companion diagnostic.
-
FDA Approves Blood Tests That Can Help Guide Cancer Treatment
FDA has recently approved two blood tests, known as liquid biopsies, that gather genetic information to help inform treatment decisions for people with cancer. This Cancer Currents story explores how the tests are used and who can get the tests.
-
CAR T-Cell Therapy Approved by FDA for Mantle Cell Lymphoma
A CAR T-cell therapy called brexucabtagene autoleucel (Tecartus) has been approved by the Food and Drug Administration (FDA) for some patients with mantle cell lymphoma. This is the third CAR T-cell therapy approved by FDA for patients with cancer.
-
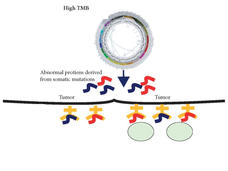
A New FDA Approval Furthers the Role of Genomics in Cancer Care
FDA’s approval of pembrolizumab (Keytruda) to treat people whose cancer is tumor mutational burden-high highlights the importance of genomic testing to guide treatment, including for children with cancer, according to NCI Director Dr. Ned Sharpless.
-
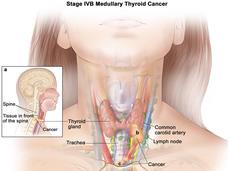
Selpercatinib Approved for Thyroid and Lung Cancers with RET Gene Alterations
FDA has granted accelerated approval for selpercatinib (Retevmo) to treat certain patients with thyroid cancer or non-small cell lung cancer whose tumors have RET gene alterations. The drug, which works by blocking the activity of RET proteins, was approved based on the results of the LIBRETTO-001 trial.
-
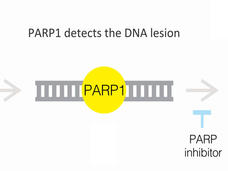
With Two FDA Approvals, Prostate Cancer Treatment Enters the PARP Era
FDA has approved olaparib (Lynparza) and rucaparib (Rubraca) to treat some men with metastatic prostate cancer. The PARP inhibitors are approved for men whose cancers have stopped responding to hormone treatment and have specific genetic alterations.
-
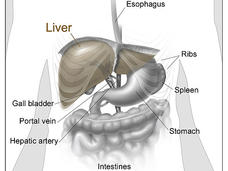
Atezolizumab Plus Bevacizumab Approved to Treat Liver Cancer
FDA has approved atezolizumab (Tecentriq) plus bevacizumab (Avastin) as an initial treatment for some people with advanced liver cancer. This is the first approval in 13 years for a treatment that is more effective than the current standard, sorafenib.
-
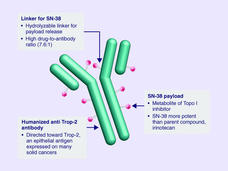
Sacituzumab Govitecan Approved for Metastatic Triple-Negative Breast Cancer
FDA has approved sacituzumab govitecan (Trodelvy) for the treatment of triple-negative breast cancer that has spread to other parts of the body. Under the approval, patients must have already undergone at least two prior treatment regimens.
-
Encorafenib, Cetuximab Combination Approved for Metastatic Colorectal Cancer
The Food and Drug Administration has approved encorafenib (Braftovi) in combination with cetuximab (Erbitux) to treat adults with metastatic colorectal cancer whose tumors have a specific mutation in the BRAF gene, called V600E.
-
Selumetinib Approved by FDA to Treat Children with NF1
The Food and Drug Administration (FDA) has approved selumetinib (Koselugo) to treat children with neurofibromatosis 1 (NF1), a genetic disorder that causes tumors, called plexiform neurofibromas, to form throughout the nervous system.
-
Avapritinib Approved to Treat GIST with a Rare Gene Alteration
Avapritinib (Ayvakit) has been approved for adults with gastrointestinal stromal tumors (GIST) whose tumors have an alteration in a portion of the PDGFRA gene called exon 18. The approval applies to those whose tumors cannot be removed with surgery or have spread.
-
Enfortumab Vedotin Approved for Recurrent Bladder Cancer
Enfortumab vedotin-ejfv (Padcev) has been approved for people with advanced bladder cancer. FDA granted the drug accelerated approval for cancers that have progressed despite previous treatments.
-
For Metastatic HER2-Positive Breast Cancer, New Treatments Emerge
Tucatinib improved survival for women in the HER2CLIMB trial, including some whose cancer had spread to the brain. Trastuzumab deruxtecan improved survival and shrank many tumors in the DESTINY-Breast01 trial, which led to its accelerated approval.
-
FDA Approves Entrectinib Based on Tumor Genetics Rather Than Cancer Type
FDA has approved entrectinib (Rozlytrek) for the treatment of children and adults with tumors bearing an NTRK gene fusion. The approval also covers adults with non-small cell lung cancer harboring a ROS1 gene fusion.
-
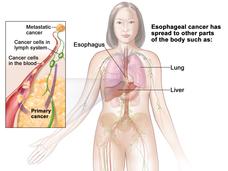
Pembrolizumab Approved for Some Patients with Advanced Esophageal Cancer
FDA has approved the immunotherapy drug pembrolizumab (Keytruda) to treat some patients with advanced esophageal cancer. Patients must have certain levels of the protein PD-L1 on their tumors, as determined by an FDA-approved test.
-
Combination Therapy with Venetoclax Approved for Chronic Lymphocytic Leukemia
The Food and Drug Administration has approved venetoclax (Venclexta) in combination with obinutuzumab (Gazyva) for the initial treatment of adults with chronic lymphocytic leukemia or small lymphocytic lymphoma.