FDA Approvals - Cancer Currents Blog
News on recent approvals of cancer therapies by the Food and Drug Administration. Includes expert comments on how the approval will influence patient care and future research.
-
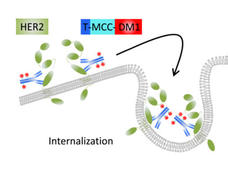
T-DM1 Approval Expanded to Include Some Women with Early-Stage HER2-Positive Breast Cancer
FDA has expanded the approved use of the drug ado-trastuzumab emtansine (Kadcyla), also called T-DM1, to include adjuvant treatment in some women with early-stage HER2-positive breast cancer.
-
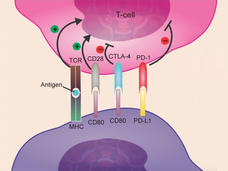
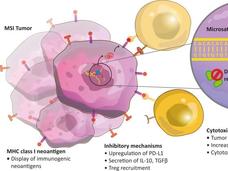
Atezolizumab Approved for Some Patients with Triple-Negative Breast Cancer
FDA has approved atezolizumab (Tecentriq) in combination with chemotherapy for the treatment of some women with advanced triple-negative breast cancer. This is the first FDA-approved regimen for breast cancer to include immunotherapy.
-
Pembrolizumab Now Second Immunotherapy Approved to Treat Merkel Cell Carcinoma
FDA has approved pembrolizumab (Keytruda) to treat people with Merkel cell carcinoma, a rare and deadly form of skin cancer. The approval covers use of the drug to treat locally advanced or metastatic forms of the disease.
-
Atezolizumab Approved for Initial Treatment of Metastatic Lung Cancer
On December 6, 2018, the Food and Drug Administration (FDA) approved atezolizumab (Tecentriq) in combination with a standard three-drug regimen as an initial treatment for advanced lung cancer that does not have EGFR or ALK mutations.
-
FDA Approvals Bring New Options for Older Patients with AML
FDA has approved venetoclax (Venclexta) and glasdegib (Daurismo) for use in people with acute myeloid leukemia aged 75 and older and those with health conditions that prevent them from receiving the intensive chemotherapy regimen that is the standard initial treatment.
-
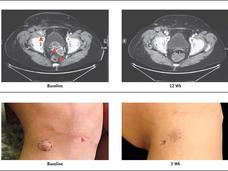
Immunotherapy Drug Cemiplimab Approved for Advanced Squamous Cell Skin Cancer
The Food and Drug Administration approved the immunotherapy drug cemiplimab (Libtayo) for an advanced form of cutaneous squamous cell carcinoma (SCC), a common type of skin cancer. It is the first agent to be approved specifically for advanced SCC.
-
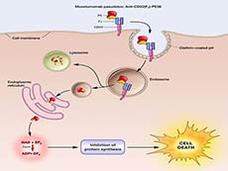
Moxetumomab Approved by FDA for Hairy Cell Leukemia
The FDA has approved moxetumomab pasudotox (Lumoxiti), a bacterial toxin–based drug, for the treatment of some patients with hairy cell leukemia (HCL). Moxetumomab is approved to treat patients with HCL who have already undergone at least two lines of standard treatments.
-
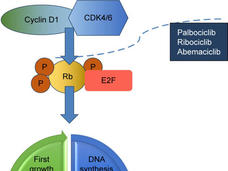
Ribociclib Approval Expanded for Some Women with Advanced Breast Cancer
FDA has expanded the approved uses of ribociclib (Kisqali) for women with advanced breast cancer, including new uses in pre- and postmenopausal women. It’s the first approval under a new FDA program to speed the review of cancer drugs.
-
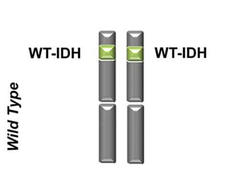
Targeted Drug Approved for Acute Myeloid Leukemia with IDH1 Gene Mutations
The FDA has approved ivosidenib (Tibsovo) for the treatment of adults with acute myeloid leukemia (AML) that has a specific mutation in a gene called IDH1. Ivosidenib becomes the first FDA-approved IDH1-targeted treatment.
-
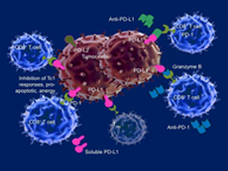
Combination of Immunotherapy Drugs Approved for Metastatic Colorectal Cancer
The FDA has approved the combination of the immune checkpoint inhibitors ipilimumab (Yervoy) and nivolumab (Opdivo) for the treatment of patients with metastatic colorectal cancer whose tumor cells have defects that affect their ability to repair DNA.
-
New Immunotherapy Option Approved for Cervical Cancer, Rare Lymphoma
FDA has approved pembrolizumab (Keytruda) for some women with advanced cervical cancer and some patients with primary mediastinal large B-cell lymphoma (PMBCL), a rare type of non-Hodgkin lymphoma.
-
FDA Alters Approved Use of Two Checkpoint Inhibitors for Bladder Cancer
FDA has changed the approved uses of the immunotherapy drugs pembrolizumab (Keytruda) and atezolizumab (Tecentriq) to treat the most common form of bladder cancer. The change is based on whether patients’ tumors have a specific biomarker.
-
Approval Expanded for Venetoclax in Chronic Lymphocytic Leukemia
FDA expanded the approval of venetoclax (Venclexta) for people with chronic lymphocytic leukemia (CLL) to include those whose cancer has progressed after previous treatment, regardless of whether their cancer cells have the deletion 17p gene alteration.
-
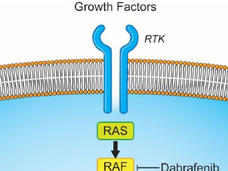
Dabrafenib–Trametinib Combination Approved for Melanoma, Anaplastic Thyroid Cancer
FDA recently approved the targeted-drug combination to treat patients with advanced melanoma and a subset of patients with a rare and aggressive form of thyroid cancer whose tumors have a specific mutation in the BRAF gene.
-
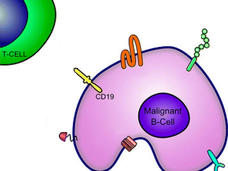
FDA Approves Second CAR T-Cell Therapy for Lymphoma
FDA has approved tisagenlecleucel (Kymriah) for certain kinds of non-Hodgkin lymphoma. Read about the trial that led to the approval and what the approval means for people with lymphoma.
-
FDA Approves Nivolumab and Ipilimumab Combination for Advanced Kidney Cancer
FDA has approved the combination of two immunotherapy drugs, nivolumab (Opdivo) and ipilimumab (Yervoy), as an initial treatment for some patients with advanced kidney cancer. Learn how this approval will affect patient care.
-
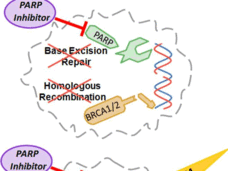
Rucaparib Approved as Maintenance Treatment for Some Recurrent Ovarian Cancers
FDA has expanded its approval of rucaparib (Rubraca) as a maintenance therapy for women with recurrent ovarian, fallopian tube, or primary peritoneal cancer whose tumors shrank after subsequent treatment with a platinum-based chemotherapy.
-
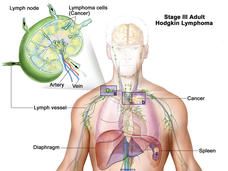
Brentuximab Approved for Initial Treatment of Advanced Hodgkin Lymphoma
The FDA has expanded the approved uses of brentuximab (Adcetris) in people with Hodgkin lymphoma. Under the new approval, brentuximab can be used in combination with three other chemotherapy drugs as an initial treatment in patients with advanced disease.
-
Abemaciclib Approval Expands Initial Treatment Options for Advanced Breast Cancer
FDA has approved the CDK4/6 inhibitor abemaciclib (Verzenio) as a first-line treatment in some women with advanced or metastatic breast cancer. Under the approval, the drug must be used in combination with an aromatase inhibitor.
-
FDA Approves Apalutamide for Some Men with Prostate Cancer
In the trial that led to the approval, apalutamide (Erleada) delayed cancer metastasis for men with prostate cancer that is resistant to androgen deprivation therapy.