Lutathera Shows Promise as Part of Initial Therapy for Some Neuroendocrine Tumors
, by Shana Spindler
People with advanced neuroendocrine tumors in the digestive system may benefit from a treatment combination that includes the drug Lu 177-dotatate (Lutathera), according to the results of a first-of-its-kind clinical trial.
All participants in the trial, called NETTER-2, had advanced neuroendocrine tumors in the gastrointestinal tract or pancreas that had not yet been treated.
Those who received Lu 177-dotatate plus octreotide (Sandostatin) lived almost three times as long without their cancer getting worse (progression-free survival) as those who received octreotide alone—a median of nearly 23 months, versus 8.5 months. And the number of people whose tumors shrank, sometimes completely, was nearly five times greater in those who received both drugs compared to those who only received octreotide.
“[These are] some of the highest response rates we’ve seen in neuroendocrine cancers to date, so that’s exciting,” said Simron Singh, M.D., of the University of Toronto, who led the NETTER-2 trial. Dr. Singh presented the findings at the ASCO Gastrointestinal Cancers Symposium on January 19.
Update: The study results were published subsequently on June 5 in The Lancet.
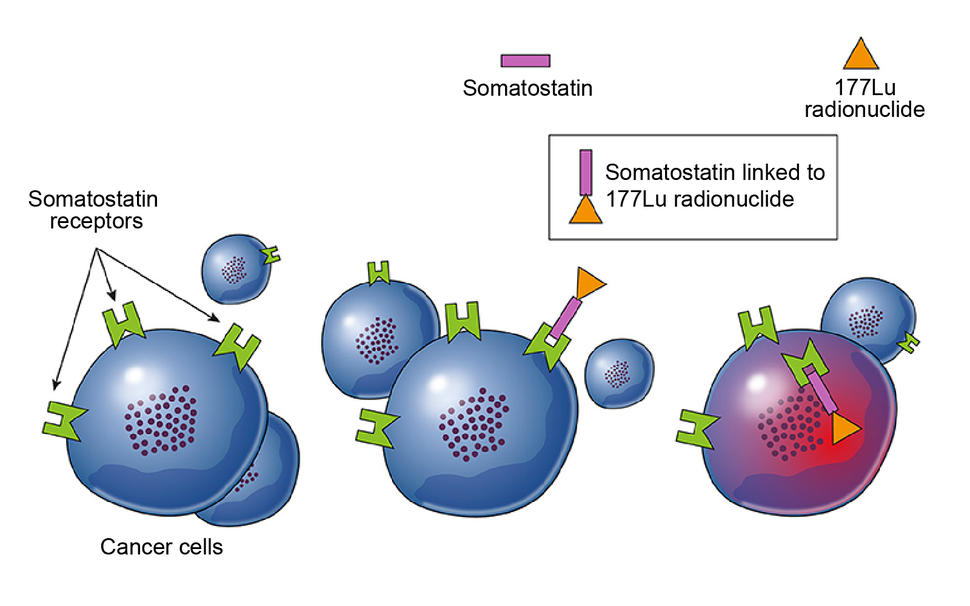
Known as a radiopharmaceutical, Lu 177-dotatate works differently than many other cancer drugs. It latches on to a specific protein on neuroendocrine tumor cells, enters the cells, and releases a radioactive particle (lutetium-177) that kills them.
Although Lu 177-dotatate is already approved by the Food and Drug Administration (FDA) for the treatment of advanced gastrointestinal and pancreatic neuroendocrine tumors, often called GEP-NETs, no trial had yet tested when and how best to use it along with other drugs commonly used to treat these cancers.
Jaydira del Rivero, M.D., of NCI’s Center for Cancer Research, who studies neuroendocrine cancers but was not involved in the NETTER-2 trial, called the work “a groundbreaking study.”
Giving Lu 177-dotatate as an initial treatment for these cancers “can be practice changing,” Dr. del Rivero said.
Rare tumors with uncertain treatment plans
As their name implies, neuroendocrine tumors (NETs) are a group of uncommon tumors that start in specialized cells in the neuroendocrine system that are involved in diverse functions in the body, from food digestion to heart rate control. These tumors are rare overall, affecting an estimated 4 in 100,000 adults.
NETs can occur in different areas throughout the body, but they’re often found in the small intestine, pancreas, and rectum. These tumors have the capacity to make hormones that may cause various symptoms, such as pain in the abdomen, nausea, and diarrhea.
Neuroendocrine tumors vary in their characteristics, and the treatment is different from patient to patient, explained Dr. del Rivero. Some people have neuroendocrine tumors that can be treated with surgery alone, she continued, whereas others will need treatments that target the specific molecular characteristics of their tumor.
For example, many GEP-NETs have a protein on their surface that binds to a hormone called somatostatin. Octreotide is a drug that mimics somatostatin and tricks the tumors into halting growth and hormone production. This is the standard initial treatment for people with advanced GEP-NETs that cannot be removed by surgery but are growing relatively slowly, which are classified as low and intermediate grade.
Typically, treatment plans are tailored to each patient's individual characteristics, taking into account factors like the amount of hormones in the blood, tumor location, other health conditions, and the presence of somatostatin receptors on tumor cells.
Advanced GEP-NETs that grow at a faster rate (high grade) are particularly challenging to manage, with no standard initial treatment for these tumors.
Although GEP-NETs are already rare, the high-grade advanced GEP-NETs are rarer still. So, studies testing the best treatments for the high-grade form of this cancer have been very difficult to do.
“Until now, we have [had] no idea of any of the effects of our approved therapies” for patients with high-grade advanced GEP-NETs, explained Jordan Berlin, M.D., of the Vanderbilt University Medical Center during a discussion of the NETTER-2 trial at the ASCO symposium.
The NETTER-2 study, however, included a substantial number of these trickier-to-treat tumors.
Lu 177-dotatate combination therapy beats octreotide alone
In early 2018, FDA approved Lu 177-dotatate for the treatment of GEP-NETs in adults. Like octreotide, the drug mimics somatostatin. But it also sneaks something into the cell—a radioactive molecule that helps kill GEP-NETs by damaging the tumor cells’ DNA.
The approval was based on findings from a trial called NETTER-1, which did not enroll people with the high-grade type of gastrointestinal neuroendocrine tumors. In addition, all participants in NETTER-1 had received at least one previous treatment.
In the NETTER-2 trial—funded by Advanced Accelerator Applications, the manufacturer of Lu 177-dotatate—the researchers tested Lu 177-dotatate as the initial treatment of intermediate- and high-grade advanced GEP-NETs. The study was the first trial ever to test a radiopharmaceutical as initial therapy for a solid tumor.
More than 200 people with advanced GEP-NETs participated in the trial. For most of the patients, their tumors originated in the pancreas or small intestine, and about one-third of the patients had the highest-grade type of tumor, called grade 3 tumors.
All participants were randomly assigned to receive Lu 177-dotatate and octreotide every 8 weeks or a larger dose of octreotide alone every 4 weeks, until their cancer started to get worse. The researchers tracked the patients for up to 3 years.
In addition to the substantial improvements in progression-free survival among participants treated with the combination, there was an equally impressive improvement in the percentage of participants whose tumors shrank, or responded, to the treatment: 43% versus 9%.
This is a “striking finding,” said Dr. del Rivero, “and unexpectedly high.”
For a little more than 5% of participants who received the combination treatment, their tumors shrank completely (a complete response). None of the participants who received octreotide alone had such an outcome.
“These data have clinical practice–changing implications,” Dr. Singh said.
Debate about Lu 177-dotatate’s long-term side effects and when to use it
The most common immediate side effects for both treatments were nausea, diarrhea, and abdominal pain, and both groups of participants reported a similar quality of life during treatment.
Some experts, however, have voiced concern about the risk of blood diseases that can occur long after Lu 177-dotatate treatment ends. In the NETTER-2 trial, one patient who received Lu 177-dotatate developed a blood cancer called myelodysplastic syndrome (MDS) following treatment.
“Neither treatment was particularly toxic, which is nice, but it is still early, and we don’t know what the long-term [blood] toxicities will be,” Dr. Berlin cautioned.
“The risk of MDS is low,” said Dr. del Rivero. People should not be hesitant to receive Lu 177-dotatate in their initial treatment if they and their doctor agree it would be the best option for them, she continued. “All of the patient’s [tumor] characteristics need to be taken into consideration … so we can tailor the best treatment option.”
Dr. Berlin agreed that Lu 177-dotatate can be an option for initial treatment, although he added that it will be important to see if it improves how long people live overall. And without much improvement on patients’ quality of life compared with octreotide alone, he added, Lu 177-dotatate adds to—rather than replaces—other initial treatment options, for now.
Making Lu 177-dotatate accessible to all who need it
In the United States, most hospitals where patients can be treated with Lu 177-dotatate are large academic centers. That’s because these centers have the infrastructure required to handle radioactive therapies, including the specialized disposal of radioactive waste, Dr. del Rivero explained.
These specialized requirements limit access to the treatment for people with GEP-NETs being treated at smaller community hospitals.
In addition, many areas around the world—such as several countries in South America, certain parts of Africa, and smaller countries in Europe and Asia—have no access to Lu 177-dotatate.
“We know it works, and we have seen it can shrink tumors based on the NETTER-2 study,” Dr. del Rivero continued. “Now, how can we make the treatment accessible not only within the [United] States but globally?”