Cannabis and Cannabinoids (PDQ®)–Health Professional Version
Overview
This cancer information summary provides an overview of the use of Cannabis and its components as a treatment for people with cancer-related symptoms caused by the disease itself or its treatment.
This summary contains the following key information:
- Cannabis has been used for medicinal purposes for thousands of years.
- By federal law, the possession of Cannabis is illegal in the United States, except within approved research settings; however, a growing number of states, territories, and the District of Columbia have enacted laws to legalize its medical and/or recreational use.
- The U.S. Food and Drug Administration has not approved Cannabis as a treatment for cancer or any other medical condition.
- Chemical components of Cannabis, called cannabinoids, activate specific receptors throughout the body to produce pharmacological effects, particularly in the central nervous system and the immune system.
- Commercially available cannabinoids, such as dronabinol and nabilone, are approved drugs for the treatment of cancer-related side effects.
- Cannabinoids may have benefits in the treatment of cancer-related side effects.
Many of the medical and scientific terms used in this summary are hypertext linked (at first use in each section) to the NCI Dictionary of Cancer Terms, which is oriented toward nonexperts. When a linked term is clicked, a definition will appear in a separate window.
Reference citations in some PDQ cancer information summaries may include links to external websites that are operated by individuals or organizations for the purpose of marketing or advocating the use of specific treatments or products. These reference citations are included for informational purposes only. Their inclusion should not be viewed as an endorsement of the content of the websites, or of any treatment or product, by the PDQ Integrative, Alternative, and Complementary Therapies Editorial Board or the National Cancer Institute.
General Information
Cannabis, also known as marijuana, originated in Central Asia but is grown worldwide today. In the United States, it is a controlled substance and is classified as a Schedule I agent (a drug with a high potential for abuse, and no currently accepted medical use). The Cannabis plant produces a resin containing 21-carbon terpenophenolic compounds called cannabinoids, in addition to other compounds found in plants, such as terpenes and flavonoids. The highest concentration of cannabinoids is found in the female flowers of the plant.[1] Delta-9-tetrahydrocannabinol (THC) is the main psychoactive cannabinoid, but over 100 other cannabinoids have been reported to be present in the plant. Cannabidiol (CBD) does not produce the characteristic altered consciousness associated with Cannabis. CBD is thought to have potential therapeutic effectiveness and has recently been approved in the form of the pharmaceutical Epidiolex for the treatment of refractory seizure disorders in children. Other cannabinoids that are being investigated for potential medical benefits include cannabinol (CBN), cannabigerol (CBG), and tetrahydrocannabivarin (THCV).
Clinical trials conducted on medicinal Cannabis are limited. The U.S. Food and Drug Administration (FDA) has not approved the use of Cannabis as a treatment for any medical condition, although both isolated THC and CBD pharmaceuticals are licensed and approved. To conduct clinical drug research with botanical Cannabis in the United States, researchers must file an Investigational New Drug (IND) application with the FDA, obtain a Schedule I license from the U.S. Drug Enforcement Administration, and obtain approval from the National Institute on Drug Abuse.
In the 2018 United States Farm Bill, the term hemp is used to describe cultivars of the Cannabis species that contain less than 0.3% THC. Hemp oil or CBD oil are products manufactured from extracts of industrial hemp (i.e., low-THC cannabis cultivars), whereas hemp seed oil is an edible fatty oil that is essentially cannabinoid-free (see Table 1). Some products contain other botanical extracts and/or over-the-counter analgesics. These products are readily available as oral and topical tinctures or other formulations often advertised for pain management and other purposes. Hemp products containing less than 0.3% of delta-9-THC are not scheduled drugs and could be considered as Farm Bill compliant. While CBD is a controlled substance, hemp is not.
| Name/Material | Constituents/Composition | |
|---|---|---|
| CBD = cannabidiol; THC = tetrahydrocannabinol. | ||
| Cannabis species, including C. sativa | Cannabinoids; also terpenoids and flavonoids | |
| • Hemp (aka industrial hemp) | Low Δ9-THC (<0.3%); high CBD | |
| • Marijuana/marihuana | High Δ9-THC (>0.3%); low CBD | |
| Nabiximols (trade name: Sativex) | Mixture of ethanol extracts of Cannabis species; contains Δ9-THC and CBD in a 1:1 ratio | |
| Hemp oil/CBD oil | Solution of a solvent extract from Cannabis flowers and/or leaves dissolved in an edible oil; typically contains 1%–5% CBD | |
| Hemp seed oil | Edible, fatty oil produced from Cannabis seeds; contains no or only traces of cannabinoids | |
| Dronabinol (trade names: Marinol and Syndros) | Synthetic Δ9-THC | |
| Nabilone (trade names: Cesamet and Canemes) | Synthetic THC analogue | |
| Cannabidiol (trade name: Epidiolex) | Highly purified (>98%), plant-derived CBD | |
The potential benefits of medicinal Cannabis for people living with cancer include the following:[2]
- Antiemetic effects.
- Appetite stimulation.
- Pain relief.
- Improved sleep.
A study of 13,180 patients with various cancers treated at 12 NCI-designated cancer centers revealed important insights into the prevalence, patterns, and perceptions of Cannabis use among people with cancer.[3] Estimates of frequency of use were obtained in a subset of 12,614 respondents from the 10 sites that drew probability samples, with 33% of the respondents reporting Cannabis use since their cancer diagnoses. Reported use showed little variation based on state legal status of Cannabis, with 26% usage reported in the one survey site in a state where Cannabis was fully illegal. Nearly 90% of Cannabis-using respondents reported various benefits: pain management (75%), better sleep (67%), nausea relief (65%), and improved appetite (56%). Sixty-five percent of patients with cancer reported some adverse effects such as inability to drive, difficulty concentrating, lung damage, and impaired memory. Reported risks of addiction and legal problems were more common in states where Cannabis was fully illegal. While 70% of patients expressed interest in learning more about medical Cannabis, only 21.5% discussed it with their cancer providers, and 15% reported receiving guidance from their healthcare team. The study highlights a significant gap between patient interest and provider engagement, emphasizing the need for better education and communication about Cannabis use in oncology care.
Although few relevant surveys of practice patterns exist, it appears that physicians caring for patients with cancer in the United States recommend medicinal Cannabis primarily for symptom management.[4]
This summary will review the role of Cannabis and the cannabinoids in the treatment of people with cancer and disease-related or treatment-related side effects. The NCI hosted a virtual meeting, the NCI Cannabis, Cannabinoids, and Cancer Research Symposium, on December 15–18, 2020. The seven sessions are summarized in the Journal of the National Cancer Institute Monographs and contain basic science and clinical information as well as a summary of the barriers to conducting Cannabis research.[5-11]
References
- Adams IB, Martin BR: Cannabis: pharmacology and toxicology in animals and humans. Addiction 91 (11): 1585-614, 1996. [PUBMED Abstract]
- Abrams DI: Integrating cannabis into clinical cancer care. Curr Oncol 23 (2): S8-S14, 2016. [PUBMED Abstract]
- Ellison GL, Helzlsouer KJ, Rosenfield SM, et al.: Perceptions, prevalence, and patterns of cannabis use among cancer patients treated at 12 NCI-Designated Cancer Centers. J Natl Cancer Inst Monogr 2024 (66): 202-217, 2024. [PUBMED Abstract]
- Doblin RE, Kleiman MA: Marijuana as antiemetic medicine: a survey of oncologists' experiences and attitudes. J Clin Oncol 9 (7): 1314-9, 1991. [PUBMED Abstract]
- Ellison GL, Alejandro Salicrup L, Freedman AN, et al.: The National Cancer Institute and Cannabis and Cannabinoids Research. J Natl Cancer Inst Monogr 2021 (58): 35-38, 2021. [PUBMED Abstract]
- Sexton M, Garcia JM, Jatoi A, et al.: The Management of Cancer Symptoms and Treatment-Induced Side Effects With Cannabis or Cannabinoids. J Natl Cancer Inst Monogr 2021 (58): 86-98, 2021. [PUBMED Abstract]
- Cooper ZD, Abrams DI, Gust S, et al.: Challenges for Clinical Cannabis and Cannabinoid Research in the United States. J Natl Cancer Inst Monogr 2021 (58): 114-122, 2021. [PUBMED Abstract]
- Braun IM, Abrams DI, Blansky SE, et al.: Cannabis and the Cancer Patient. J Natl Cancer Inst Monogr 2021 (58): 68-77, 2021. [PUBMED Abstract]
- Ward SJ, Lichtman AH, Piomelli D, et al.: Cannabinoids and Cancer Chemotherapy-Associated Adverse Effects. J Natl Cancer Inst Monogr 2021 (58): 78-85, 2021. [PUBMED Abstract]
- McAllister SD, Abood ME, Califano J, et al.: Cannabinoid Cancer Biology and Prevention. J Natl Cancer Inst Monogr 2021 (58): 99-106, 2021. [PUBMED Abstract]
- Abrams DI, Velasco G, Twelves C, et al.: Cancer Treatment: Preclinical & Clinical. J Natl Cancer Inst Monogr 2021 (58): 107-113, 2021. [PUBMED Abstract]
History
Cannabis use for medicinal purposes dates back at least 3,000 years.[1-5] It was introduced into Western medicine in 1839 by W.B. O’Shaughnessy, a surgeon who learned of its medicinal properties while working in India for the British East India Company. Its use was promoted for reported analgesic, sedative, anti-inflammatory, antispasmodic, and anticonvulsant effects.
In 1937, the U.S. Treasury Department introduced the Marihuana Tax Act. This Act imposed a levy of $1 per ounce for medicinal use of Cannabis and $100 per ounce for nonmedical use. Physicians in the United States were the principal opponents of the Act. The American Medical Association (AMA) opposed the Act because physicians were required to pay a special tax for prescribing Cannabis, use special order forms to procure it, and keep special records concerning its professional use. In addition, the AMA believed that objective evidence that Cannabis was harmful was lacking and that passage of the Act would impede further research into its medicinal worth.[6] In 1942, Cannabis was removed from the U.S. Pharmacopoeia because of persistent concerns about its potential to cause harm.[2,3] Recently, there has been renewed interest in Cannabis by the U.S. Pharmacopeia.[7]
In 1951, Congress passed the Boggs Act, which for the first time included Cannabis with narcotic drugs. In 1970, with the passage of the Controlled Substances Act, marijuana was classified by Congress as a Schedule I drug. Drugs in Schedule I are distinguished as having no currently accepted medicinal use in the United States. Other Schedule I substances include heroin, LSD, mescaline, and methaqualone.
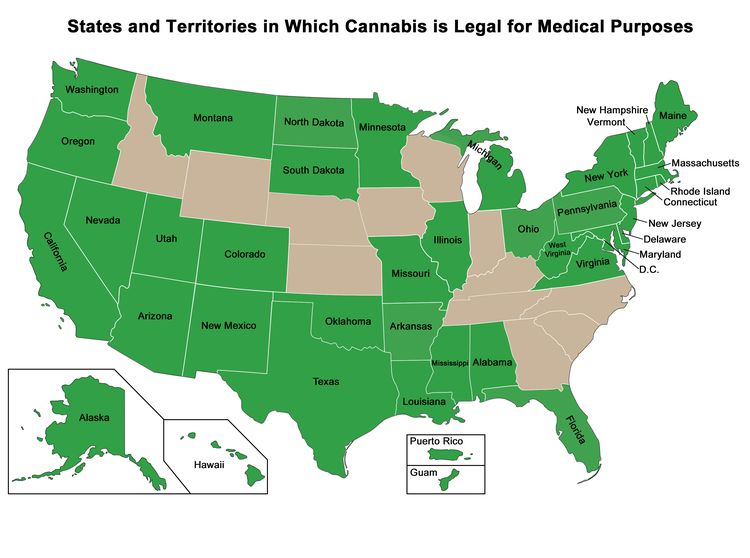
Despite its designation as having no medicinal use, Cannabis was distributed by the U.S. government to patients on a case-by-case basis under the Compassionate Use Investigational New Drug program established in 1978. Distribution of Cannabis through this program was closed to new patients in 1992.[1-4] Although federal law prohibits the use of Cannabis, Figure 1 below shows the states and territories that have legalized Cannabis use for medical purposes. Additional states have legalized only one ingredient in Cannabis, such as cannabidiol (CBD), and are not included in the map. Some medical marijuana laws are broader than others, and there is state-to-state variation in the types of medical conditions for which treatment is allowed.[8]
The main psychoactive constituent of Cannabis was identified as delta-9-tetrahydrocannabinol (THC). In 1986, an isomer of synthetic delta-9-THC in sesame oil was licensed and approved for the treatment of chemotherapy-associated nausea and vomiting under the generic name dronabinol. Clinical trials determined that dronabinol was as effective as or better than other antiemetic agents available at the time.[9] Dronabinol was also studied for its ability to stimulate weight gain in patients with AIDS in the late 1980s. Thus, the indications were expanded to include treatment of anorexia associated with human immunodeficiency virus infection in 1992. Clinical trial results showed no statistically significant weight gain, although patients reported an improvement in appetite.[10,11] Another important cannabinoid found in Cannabis is CBD.[12] This is a nonpsychoactive cannabinoid, which is an analogue of THC.
In recent decades, the neurobiology of cannabinoids has been analyzed.[13-16] The first cannabinoid receptor, CB1, was identified in the brain in 1988. A second cannabinoid receptor, CB2, was identified in 1993. The highest expression of CB2 receptors is located on B lymphocytes and natural killer cells, suggesting a possible role in immunity. Endogenous cannabinoids (endocannabinoids) have been identified and appear to have a role in pain modulation, control of movement, feeding behavior, mood, bone growth, inflammation, neuroprotection, and memory.[17]
Nabiximols (Sativex), a Cannabis extract with a 1:1 ratio of THC:CBD, is approved in Canada (under the Notice of Compliance with Conditions) for symptomatic relief of pain in advanced cancer and multiple sclerosis.[18] Nabiximols is an herbal preparation containing a defined quantity of specific cannabinoids formulated for oromucosal spray administration with potential analgesic activity. Nabiximols contains extracts from two Cannabis plant varieties. The extracts mixture is standardized to the concentrations of the psychoactive delta-9-THC and the nonpsychoactive CBD. The preparation also contains other, more minor cannabinoids, flavonoids, and terpenoids.[19] Canada, New Zealand, and most countries in western Europe also have approved nabiximols for spasticity of multiple sclerosis, a common symptom that may include muscle stiffness, reduced mobility, and pain, and for which existing therapy is unsatisfactory.
References
- Abel EL: Marihuana, The First Twelve Thousand Years. Plenum Press, 1980. Also available online. Last accessed June 2, 2021.
- Joy JE, Watson SJ, Benson JA, eds.: Marijuana and Medicine: Assessing the Science Base. National Academy Press, 1999. Also available online. Last accessed June 2, 2021.
- Mack A, Joy J: Marijuana As Medicine? The Science Beyond the Controversy. National Academy Press, 2001. Also available online. Last accessed June 2, 2021.
- Booth M: Cannabis: A History. St Martin's Press, 2003.
- Russo EB, Jiang HE, Li X, et al.: Phytochemical and genetic analyses of ancient cannabis from Central Asia. J Exp Bot 59 (15): 4171-82, 2008. [PUBMED Abstract]
- Schaffer Library of Drug Policy: The Marihuana Tax Act of 1937: Taxation of Marihuana. Washington, DC: House of Representatives, Committee on Ways and Means, 1937. Available online. Last accessed June 2, 2021.
- Sarma ND, Waye A, ElSohly MA, et al.: Cannabis Inflorescence for Medical Purposes: USP Considerations for Quality Attributes. J Nat Prod 83 (4): 1334-1351, 2020. [PUBMED Abstract]
- National Academies of Sciences, Engineering, and Medicine: The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. The National Academies Press, 2017.
- Sallan SE, Zinberg NE, Frei E: Antiemetic effect of delta-9-tetrahydrocannabinol in patients receiving cancer chemotherapy. N Engl J Med 293 (16): 795-7, 1975. [PUBMED Abstract]
- Gorter R, Seefried M, Volberding P: Dronabinol effects on weight in patients with HIV infection. AIDS 6 (1): 127, 1992. [PUBMED Abstract]
- Beal JE, Olson R, Laubenstein L, et al.: Dronabinol as a treatment for anorexia associated with weight loss in patients with AIDS. J Pain Symptom Manage 10 (2): 89-97, 1995. [PUBMED Abstract]
- Adams R, Hunt M, Clark JH: Structure of cannabidiol: a product isolated from the marihuana extract of Minnesota wild hemp. J Am Chem Soc 62 (1): 196-200, 1940. Also available online. Last accessed June 2, 2021.
- Devane WA, Dysarz FA, Johnson MR, et al.: Determination and characterization of a cannabinoid receptor in rat brain. Mol Pharmacol 34 (5): 605-13, 1988. [PUBMED Abstract]
- Devane WA, Hanus L, Breuer A, et al.: Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258 (5090): 1946-9, 1992. [PUBMED Abstract]
- Pertwee RG, Howlett AC, Abood ME, et al.: International Union of Basic and Clinical Pharmacology. LXXIX. Cannabinoid receptors and their ligands: beyond CB₁ and CB₂. Pharmacol Rev 62 (4): 588-631, 2010. [PUBMED Abstract]
- Felder CC, Glass M: Cannabinoid receptors and their endogenous agonists. Annu Rev Pharmacol Toxicol 38: 179-200, 1998. [PUBMED Abstract]
- Pacher P, Bátkai S, Kunos G: The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev 58 (3): 389-462, 2006. [PUBMED Abstract]
- Howard P, Twycross R, Shuster J, et al.: Cannabinoids. J Pain Symptom Manage 46 (1): 142-9, 2013. [PUBMED Abstract]
- Nabiximols. Bethesda, MD: National Center for Biotechnology Information, 2009. Available online. Last accessed June 2, 2021.
Laboratory/Animal/Preclinical Studies
Cannabinoids are a group of 21-carbon–containing terpenophenolic compounds produced uniquely by Cannabis species (e.g., Cannabis sativa L.).[1,2] These plant-derived compounds may be referred to as phytocannabinoids. Although delta-9-tetrahydrocannabinol (THC) is the primary psychoactive ingredient, other known compounds with biological activity are cannabinol, cannabidiol (CBD), cannabichromene, cannabigerol, tetrahydrocannabivarin, and delta-8-THC. CBD, in particular, is thought to have significant analgesic, anti-inflammatory, and anxiolytic activity without the psychoactive effect (high) of delta-9-THC.
Antitumor Effects
One study in mice and rats suggested that cannabinoids may have a protective effect against the development of certain types of tumors.[3] During this 2-year study, groups of mice and rats were given various doses of THC by gavage. A dose-related decrease in the incidence of hepatic adenoma tumors and hepatocellular carcinoma (HCC) was observed in the mice. Decreased incidences of benign tumors (polyps and adenomas) in other organs (mammary gland, uterus, pituitary, testis, and pancreas) were also noted in the rats. In another study, delta-9-THC, delta-8-THC, and cannabinol were found to inhibit the growth of Lewis lung adenocarcinoma cells in vitro and in vivo.[4] In addition, other tumors have been shown to be sensitive to cannabinoid-induced growth inhibition.[5-8]
Cannabinoids may cause antitumor effects by various mechanisms, including induction of cell death, inhibition of cell growth, and inhibition of tumor angiogenesis invasion and metastasis.[9-12] Two reviews summarize the molecular mechanisms of action of cannabinoids as antitumor agents.[13,14] Cannabinoids appear to kill tumor cells but do not affect their nontransformed counterparts and may even protect them from cell death. For example, these compounds have been shown to induce apoptosis in glioma cells in culture and induce regression of glioma tumors in mice and rats, while they protect normal glial cells of astroglial and oligodendroglial lineages from apoptosis mediated by the CB1 receptor.[9]
The effects of delta-9-THC and a synthetic agonist of the CB2 receptor were investigated in HCC.[15] Both agents reduced the viability of HCC cells in vitro and demonstrated antitumor effects in HCC subcutaneous xenografts in nude mice. The investigations documented that the anti-HCC effects are mediated by way of the CB2 receptor. Similar to findings in glioma cells, the cannabinoids were shown to trigger cell death through stimulation of an endoplasmic reticulum stress pathway that activates autophagy and promotes apoptosis. Other investigations have confirmed that CB1 and CB2 receptors may be potential targets in non-small cell lung carcinoma [16] and breast cancer.[17]
An in vitro study of the effect of CBD on programmed cell death in breast cancer cell lines found that CBD induced programmed cell death, independent of the CB1, CB2, or vanilloid receptors. CBD inhibited the survival of both estrogen receptor–positive and estrogen receptor–negative breast cancer cell lines, inducing apoptosis in a concentration-dependent manner while having little effect on nontumorigenic mammary cells.[18] Other studies have also shown the antitumor effect of cannabinoids (i.e., CBD and THC) in preclinical models of breast cancer.[19,20]
CBD has also been demonstrated to exert a chemopreventive effect in a mouse model of colon cancer.[21] In this experimental system, azoxymethane increased premalignant and malignant lesions in the mouse colon. Animals treated with azoxymethane and CBD concurrently were protected from developing premalignant and malignant lesions. In in vitro experiments involving colorectal cancer cell lines, the investigators found that CBD protected DNA from oxidative damage, increased endocannabinoid levels, and reduced cell proliferation. In a subsequent study, the investigators found that the antiproliferative effect of CBD was counteracted by selective CB1 but not CB2 receptor antagonists, suggesting an involvement of CB1 receptors.[22]
Another investigation into the antitumor effects of CBD examined the role of intercellular adhesion molecule-1 (ICAM-1).[12] ICAM-1 expression in tumor cells has been reported to be negatively correlated with cancer metastasis. In lung cancer cell lines, CBD upregulated ICAM-1, leading to decreased cancer cell invasiveness.
In an in vivo model using severe combined immunodeficient mice, subcutaneous tumors were generated by inoculating the animals with cells from human non-small cell lung carcinoma cell lines.[23] Tumor growth was inhibited by 60% in THC-treated mice compared with vehicle-treated control mice. Tumor specimens revealed that THC had antiangiogenic and antiproliferative effects. However, research with immunocompetent murine tumor models has demonstrated immunosuppression and enhanced tumor growth in mice treated with THC.[24,25]
In addition, both plant-derived and endogenous cannabinoids have been studied for anti-inflammatory effects. A mouse study demonstrated that endogenous cannabinoid system signaling is likely to provide intrinsic protection against colonic inflammation.[26] As a result, a hypothesis that phytocannabinoids and endocannabinoids may be useful in the risk reduction and treatment of colorectal cancer has been developed.[27-30]
CBD may also enhance uptake of cytotoxic drugs into malignant cells. Activation of transient receptor potential vanilloid type 2 (TRPV2) has been shown to inhibit proliferation of human glioblastoma multiforme cells and overcome resistance to the chemotherapy agent carmustine. [31] One study showed that coadministration of THC and CBD over single-agent usage had greater antiproliferative activity in an in vitro study with multiple human glioblastoma multiforme cell lines.[32] In an in vitro model, CBD increased TRPV2 activation and increased uptake of cytotoxic drugs, leading to apoptosis of glioma cells without affecting normal human astrocytes. This suggests that coadministration of CBD with cytotoxic agents may increase drug uptake and potentiate cell death in human glioma cells. Also, CBD together with THC may enhance the antitumor activity of classic chemotherapeutic drugs such as temozolomide in some mouse models of cancer.[13,33] A meta-analysis of 34 in vitro and in vivo studies of cannabinoids in glioma reported that all but one study confirmed that cannabinoids selectively kill tumor cells.[34]
Antiemetic Effects
Preclinical research suggests that emetic circuitry is tonically controlled by endocannabinoids. The antiemetic action of cannabinoids is believed to be mediated via interaction with the 5-hydroxytryptamine 3 (5-HT3) receptor. CB1 receptors and 5-HT3 receptors are colocalized on gamma-aminobutyric acid (GABA)-ergic neurons, where they have opposite effects on GABA release.[35] There also may be direct inhibition of 5-HT3 gated ion currents through non–CB1 receptor pathways. CB1 receptor antagonists have been shown to elicit emesis in the least shrew that is reversed by cannabinoid agonists.[36] The involvement of CB1 receptor in emesis prevention has been shown by the ability of CB1 antagonists to reverse the effects of THC and other synthetic cannabinoid CB1 agonists in suppressing vomiting caused by cisplatin in the house musk shrew and lithium chloride in the least shrew. In the latter model, CBD was also shown to be efficacious.[37,38]
Appetite Stimulation
Many animal studies have previously demonstrated that delta-9-THC and other cannabinoids have a stimulatory effect on appetite and increase food intake. It is believed that the endogenous cannabinoid system may serve as a regulator of feeding behavior. The endogenous cannabinoid anandamide potently enhances appetite in mice.[39] Moreover, CB1 receptors in the hypothalamus may be involved in the motivational or reward aspects of eating.[40]
Analgesia
Understanding the mechanism of cannabinoid-induced analgesia has been increased through the study of cannabinoid receptors, endocannabinoids, and synthetic agonists and antagonists. Cannabinoids produce analgesia through supraspinal, spinal, and peripheral modes of action, acting on both ascending and descending pain pathways.[41] The CB1 receptor is found in both the central nervous system (CNS) and in peripheral nerve terminals. Similar to opioid receptors, increased levels of the CB1 receptor are found in regions of the brain that regulate nociceptive processing.[42] CB2 receptors, located predominantly in peripheral tissue, exist at very low levels in the CNS. With the development of receptor-specific antagonists, additional information about the roles of the receptors and endogenous cannabinoids in the modulation of pain has been obtained.[43,44]
Cannabinoids may also contribute to pain modulation through an anti-inflammatory mechanism; a CB2 effect with cannabinoids acting on mast cell receptors to attenuate the release of inflammatory agents, such as histamine and serotonin, and on keratinocytes to enhance the release of analgesic opioids has been described.[45-47] One study reported that the efficacy of synthetic CB1- and CB2-receptor agonists were comparable with the efficacy of morphine in a murine model of tumor pain.[48]
Cannabinoids have been shown to prevent chemotherapy-induced neuropathy in animal models exposed to paclitaxel, vincristine, or cisplatin.[49-51]
Anxiety and Sleep
The endocannabinoid system is believed to be centrally involved in the regulation of mood and the extinction of aversive memories. Animal studies have shown CBD to have anxiolytic properties. It was shown in rats that these anxiolytic properties are mediated through unknown mechanisms.[52] Anxiolytic effects of CBD have been shown in several animal models.[53,54]
The endocannabinoid system has also been shown to play a key role in the modulation of the sleep-waking cycle in rats.[55,56]
References
- Adams IB, Martin BR: Cannabis: pharmacology and toxicology in animals and humans. Addiction 91 (11): 1585-614, 1996. [PUBMED Abstract]
- Grotenhermen F, Russo E, eds.: Cannabis and Cannabinoids: Pharmacology, Toxicology, and Therapeutic Potential. The Haworth Press, 2002.
- National Toxicology Program: NTP toxicology and carcinogenesis studies of 1-trans-delta(9)-tetrahydrocannabinol (CAS No. 1972-08-3) in F344 rats and B6C3F1 mice (gavage studies). Natl Toxicol Program Tech Rep Ser 446: 1-317, 1996. [PUBMED Abstract]
- Bifulco M, Laezza C, Pisanti S, et al.: Cannabinoids and cancer: pros and cons of an antitumour strategy. Br J Pharmacol 148 (2): 123-35, 2006. [PUBMED Abstract]
- Sánchez C, de Ceballos ML, Gomez del Pulgar T, et al.: Inhibition of glioma growth in vivo by selective activation of the CB(2) cannabinoid receptor. Cancer Res 61 (15): 5784-9, 2001. [PUBMED Abstract]
- McKallip RJ, Lombard C, Fisher M, et al.: Targeting CB2 cannabinoid receptors as a novel therapy to treat malignant lymphoblastic disease. Blood 100 (2): 627-34, 2002. [PUBMED Abstract]
- Casanova ML, Blázquez C, Martínez-Palacio J, et al.: Inhibition of skin tumor growth and angiogenesis in vivo by activation of cannabinoid receptors. J Clin Invest 111 (1): 43-50, 2003. [PUBMED Abstract]
- Blázquez C, González-Feria L, Alvarez L, et al.: Cannabinoids inhibit the vascular endothelial growth factor pathway in gliomas. Cancer Res 64 (16): 5617-23, 2004. [PUBMED Abstract]
- Guzmán M: Cannabinoids: potential anticancer agents. Nat Rev Cancer 3 (10): 745-55, 2003. [PUBMED Abstract]
- Blázquez C, Casanova ML, Planas A, et al.: Inhibition of tumor angiogenesis by cannabinoids. FASEB J 17 (3): 529-31, 2003. [PUBMED Abstract]
- Vaccani A, Massi P, Colombo A, et al.: Cannabidiol inhibits human glioma cell migration through a cannabinoid receptor-independent mechanism. Br J Pharmacol 144 (8): 1032-6, 2005. [PUBMED Abstract]
- Ramer R, Bublitz K, Freimuth N, et al.: Cannabidiol inhibits lung cancer cell invasion and metastasis via intercellular adhesion molecule-1. FASEB J 26 (4): 1535-48, 2012. [PUBMED Abstract]
- Velasco G, Sánchez C, Guzmán M: Towards the use of cannabinoids as antitumour agents. Nat Rev Cancer 12 (6): 436-44, 2012. [PUBMED Abstract]
- Cridge BJ, Rosengren RJ: Critical appraisal of the potential use of cannabinoids in cancer management. Cancer Manag Res 5: 301-13, 2013. [PUBMED Abstract]
- Vara D, Salazar M, Olea-Herrero N, et al.: Anti-tumoral action of cannabinoids on hepatocellular carcinoma: role of AMPK-dependent activation of autophagy. Cell Death Differ 18 (7): 1099-111, 2011. [PUBMED Abstract]
- Preet A, Qamri Z, Nasser MW, et al.: Cannabinoid receptors, CB1 and CB2, as novel targets for inhibition of non-small cell lung cancer growth and metastasis. Cancer Prev Res (Phila) 4 (1): 65-75, 2011. [PUBMED Abstract]
- Nasser MW, Qamri Z, Deol YS, et al.: Crosstalk between chemokine receptor CXCR4 and cannabinoid receptor CB2 in modulating breast cancer growth and invasion. PLoS One 6 (9): e23901, 2011. [PUBMED Abstract]
- Shrivastava A, Kuzontkoski PM, Groopman JE, et al.: Cannabidiol induces programmed cell death in breast cancer cells by coordinating the cross-talk between apoptosis and autophagy. Mol Cancer Ther 10 (7): 1161-72, 2011. [PUBMED Abstract]
- Caffarel MM, Andradas C, Mira E, et al.: Cannabinoids reduce ErbB2-driven breast cancer progression through Akt inhibition. Mol Cancer 9: 196, 2010. [PUBMED Abstract]
- McAllister SD, Murase R, Christian RT, et al.: Pathways mediating the effects of cannabidiol on the reduction of breast cancer cell proliferation, invasion, and metastasis. Breast Cancer Res Treat 129 (1): 37-47, 2011. [PUBMED Abstract]
- Aviello G, Romano B, Borrelli F, et al.: Chemopreventive effect of the non-psychotropic phytocannabinoid cannabidiol on experimental colon cancer. J Mol Med (Berl) 90 (8): 925-34, 2012. [PUBMED Abstract]
- Romano B, Borrelli F, Pagano E, et al.: Inhibition of colon carcinogenesis by a standardized Cannabis sativa extract with high content of cannabidiol. Phytomedicine 21 (5): 631-9, 2014. [PUBMED Abstract]
- Preet A, Ganju RK, Groopman JE: Delta9-Tetrahydrocannabinol inhibits epithelial growth factor-induced lung cancer cell migration in vitro as well as its growth and metastasis in vivo. Oncogene 27 (3): 339-46, 2008. [PUBMED Abstract]
- Zhu LX, Sharma S, Stolina M, et al.: Delta-9-tetrahydrocannabinol inhibits antitumor immunity by a CB2 receptor-mediated, cytokine-dependent pathway. J Immunol 165 (1): 373-80, 2000. [PUBMED Abstract]
- McKallip RJ, Nagarkatti M, Nagarkatti PS: Delta-9-tetrahydrocannabinol enhances breast cancer growth and metastasis by suppression of the antitumor immune response. J Immunol 174 (6): 3281-9, 2005. [PUBMED Abstract]
- Massa F, Marsicano G, Hermann H, et al.: The endogenous cannabinoid system protects against colonic inflammation. J Clin Invest 113 (8): 1202-9, 2004. [PUBMED Abstract]
- Patsos HA, Hicks DJ, Greenhough A, et al.: Cannabinoids and cancer: potential for colorectal cancer therapy. Biochem Soc Trans 33 (Pt 4): 712-4, 2005. [PUBMED Abstract]
- Liu WM, Fowler DW, Dalgleish AG: Cannabis-derived substances in cancer therapy--an emerging anti-inflammatory role for the cannabinoids. Curr Clin Pharmacol 5 (4): 281-7, 2010. [PUBMED Abstract]
- Malfitano AM, Ciaglia E, Gangemi G, et al.: Update on the endocannabinoid system as an anticancer target. Expert Opin Ther Targets 15 (3): 297-308, 2011. [PUBMED Abstract]
- Sarfaraz S, Adhami VM, Syed DN, et al.: Cannabinoids for cancer treatment: progress and promise. Cancer Res 68 (2): 339-42, 2008. [PUBMED Abstract]
- Nabissi M, Morelli MB, Santoni M, et al.: Triggering of the TRPV2 channel by cannabidiol sensitizes glioblastoma cells to cytotoxic chemotherapeutic agents. Carcinogenesis 34 (1): 48-57, 2013. [PUBMED Abstract]
- Marcu JP, Christian RT, Lau D, et al.: Cannabidiol enhances the inhibitory effects of delta9-tetrahydrocannabinol on human glioblastoma cell proliferation and survival. Mol Cancer Ther 9 (1): 180-9, 2010. [PUBMED Abstract]
- Torres S, Lorente M, Rodríguez-Fornés F, et al.: A combined preclinical therapy of cannabinoids and temozolomide against glioma. Mol Cancer Ther 10 (1): 90-103, 2011. [PUBMED Abstract]
- Rocha FC, Dos Santos Júnior JG, Stefano SC, et al.: Systematic review of the literature on clinical and experimental trials on the antitumor effects of cannabinoids in gliomas. J Neurooncol 116 (1): 11-24, 2014. [PUBMED Abstract]
- Pacher P, Bátkai S, Kunos G: The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev 58 (3): 389-462, 2006. [PUBMED Abstract]
- Darmani NA: Delta(9)-tetrahydrocannabinol and synthetic cannabinoids prevent emesis produced by the cannabinoid CB(1) receptor antagonist/inverse agonist SR 141716A. Neuropsychopharmacology 24 (2): 198-203, 2001. [PUBMED Abstract]
- Darmani NA: Delta-9-tetrahydrocannabinol differentially suppresses cisplatin-induced emesis and indices of motor function via cannabinoid CB(1) receptors in the least shrew. Pharmacol Biochem Behav 69 (1-2): 239-49, 2001 May-Jun. [PUBMED Abstract]
- Parker LA, Kwiatkowska M, Burton P, et al.: Effect of cannabinoids on lithium-induced vomiting in the Suncus murinus (house musk shrew). Psychopharmacology (Berl) 171 (2): 156-61, 2004. [PUBMED Abstract]
- Mechoulam R, Berry EM, Avraham Y, et al.: Endocannabinoids, feeding and suckling--from our perspective. Int J Obes (Lond) 30 (Suppl 1): S24-8, 2006. [PUBMED Abstract]
- Fride E, Bregman T, Kirkham TC: Endocannabinoids and food intake: newborn suckling and appetite regulation in adulthood. Exp Biol Med (Maywood) 230 (4): 225-34, 2005. [PUBMED Abstract]
- Baker D, Pryce G, Giovannoni G, et al.: The therapeutic potential of cannabis. Lancet Neurol 2 (5): 291-8, 2003. [PUBMED Abstract]
- Walker JM, Hohmann AG, Martin WJ, et al.: The neurobiology of cannabinoid analgesia. Life Sci 65 (6-7): 665-73, 1999. [PUBMED Abstract]
- Meng ID, Manning BH, Martin WJ, et al.: An analgesia circuit activated by cannabinoids. Nature 395 (6700): 381-3, 1998. [PUBMED Abstract]
- Walker JM, Huang SM, Strangman NM, et al.: Pain modulation by release of the endogenous cannabinoid anandamide. Proc Natl Acad Sci U S A 96 (21): 12198-203, 1999. [PUBMED Abstract]
- Facci L, Dal Toso R, Romanello S, et al.: Mast cells express a peripheral cannabinoid receptor with differential sensitivity to anandamide and palmitoylethanolamide. Proc Natl Acad Sci U S A 92 (8): 3376-80, 1995. [PUBMED Abstract]
- Ibrahim MM, Porreca F, Lai J, et al.: CB2 cannabinoid receptor activation produces antinociception by stimulating peripheral release of endogenous opioids. Proc Natl Acad Sci U S A 102 (8): 3093-8, 2005. [PUBMED Abstract]
- Richardson JD, Kilo S, Hargreaves KM: Cannabinoids reduce hyperalgesia and inflammation via interaction with peripheral CB1 receptors. Pain 75 (1): 111-9, 1998. [PUBMED Abstract]
- Khasabova IA, Gielissen J, Chandiramani A, et al.: CB1 and CB2 receptor agonists promote analgesia through synergy in a murine model of tumor pain. Behav Pharmacol 22 (5-6): 607-16, 2011. [PUBMED Abstract]
- Ward SJ, McAllister SD, Kawamura R, et al.: Cannabidiol inhibits paclitaxel-induced neuropathic pain through 5-HT(1A) receptors without diminishing nervous system function or chemotherapy efficacy. Br J Pharmacol 171 (3): 636-45, 2014. [PUBMED Abstract]
- Rahn EJ, Makriyannis A, Hohmann AG: Activation of cannabinoid CB1 and CB2 receptors suppresses neuropathic nociception evoked by the chemotherapeutic agent vincristine in rats. Br J Pharmacol 152 (5): 765-77, 2007. [PUBMED Abstract]
- Khasabova IA, Khasabov S, Paz J, et al.: Cannabinoid type-1 receptor reduces pain and neurotoxicity produced by chemotherapy. J Neurosci 32 (20): 7091-101, 2012. [PUBMED Abstract]
- Campos AC, Guimarães FS: Involvement of 5HT1A receptors in the anxiolytic-like effects of cannabidiol injected into the dorsolateral periaqueductal gray of rats. Psychopharmacology (Berl) 199 (2): 223-30, 2008. [PUBMED Abstract]
- Crippa JA, Zuardi AW, Hallak JE: [Therapeutical use of the cannabinoids in psychiatry]. Rev Bras Psiquiatr 32 (Suppl 1): S56-66, 2010. [PUBMED Abstract]
- Guimarães FS, Chiaretti TM, Graeff FG, et al.: Antianxiety effect of cannabidiol in the elevated plus-maze. Psychopharmacology (Berl) 100 (4): 558-9, 1990. [PUBMED Abstract]
- Méndez-Díaz M, Caynas-Rojas S, Arteaga Santacruz V, et al.: Entopeduncular nucleus endocannabinoid system modulates sleep-waking cycle and mood in rats. Pharmacol Biochem Behav 107: 29-35, 2013. [PUBMED Abstract]
- Pava MJ, den Hartog CR, Blanco-Centurion C, et al.: Endocannabinoid modulation of cortical up-states and NREM sleep. PLoS One 9 (2): e88672, 2014. [PUBMED Abstract]
Human/Clinical Studies
Cannabis Pharmacology
When oral Cannabis is ingested, there is a low (6%–20%) and variable oral bioavailability.[1,2] Peak plasma concentrations of delta-9-tetrahydrocannabinol (THC) occur after 1 to 6 hours and remain elevated with a terminal half-life of 20 to 30 hours. Taken by mouth, delta-9-THC is initially metabolized in the liver to 11-OH-THC, a potent psychoactive metabolite. Inhaled cannabinoids are rapidly absorbed into the bloodstream with a peak concentration in 2 to 10 minutes, declining rapidly for a period of 30 minutes and with less generation of the psychoactive 11-OH metabolite.
Cannabinoids are known to interact with the hepatic cytochrome P450 enzyme system.[3,4] In one study, 24 patients with cancer were treated with intravenous irinotecan (600 mg, n = 12) or docetaxel (180 mg, n = 12), followed 3 weeks later by the same drugs concomitant with medicinal Cannabis taken in the form of an herbal tea for 15 consecutive days, starting 12 days before the second treatment.[4] The administration of Cannabis did not significantly influence exposure to and clearance of irinotecan or docetaxel, although the herbal tea route of administration may not reproduce the effects of inhalation or oral ingestion of fat-soluble cannabinoids.
Highly concentrated THC or cannabidiol (CBD) oil extracts are illegally promoted as potential cancer cures.[5] These oils have not been evaluated in any clinical trials for anticancer activity or safety. Because CBD is a potential inhibitor of certain cytochrome P450 enzymes, highly concentrated CBD oils used concurrently with conventional therapies that are metabolized by these enzymes could potentially increase toxicity or decrease the effectiveness of these therapies.[6,7]
Additionally, multiple reliability and quality concerns have been raised about Cannabis analytical labs, alleging the inflation of cannabinoids content in different Cannabis products. One study showed major inconsistencies in reported THC content because of an insufficient stabilization of sample preparation and testing methods.[8]
Cancer Risk
A number of studies have yielded conflicting evidence regarding the risks of various cancers associated with Cannabis smoking.
A pooled analysis of three case-cohort studies of men in northwestern Africa (430 cases and 778 controls) showed a significantly increased risk of lung cancer among tobacco smokers who also inhaled Cannabis.[9]
A large, retrospective cohort study of 64,855 men aged 15 to 49 years from the United States found that Cannabis use was not associated with tobacco-related cancers and a number of other common malignancies. However, the study did find that, among nonsmokers of tobacco, ever having used Cannabis was associated with an increased risk of prostate cancer.[10]
A population-based case-control study of 611 patients with lung cancer revealed that chronic low Cannabis exposure was not associated with an increased risk of lung cancer or other upper aerodigestive tract cancers. The study also found no positive associations with any cancer type (oral, pharyngeal, laryngeal, lung, or esophageal) when adjusting for several confounders, including cigarette smoking.[11]
A systematic review of 19 studies evaluating premalignant or malignant lung lesions in people 18 years or older who inhaled Cannabis concluded that observational studies failed to demonstrate a statistically significant association between Cannabis inhalation and lung cancer after adjusting for tobacco use.[12] In the review of the published meta-analyses, the National Academies of Sciences, Engineering, and Medicine (NASEM) report concluded that there was moderate evidence of no statistical association between Cannabis smoking and the incidence of lung cancer.[13]
Epidemiological studies examining one association of Cannabis use with head and neck squamous cell carcinomas have also been inconsistent in their findings. A pooled analysis of nine case-control studies from the U.S./Latin American International Head and Neck Cancer Epidemiology (INHANCE) Consortium included information from 1,921 oropharyngeal cancer cases, 356 tongue cancer cases, and 7,639 controls. The study found that Cannabis smokers had an elevated risk of oropharyngeal cancers and a reduced risk of tongue cancer compared with those who never smoked Cannabis. These study results both reflect the inconsistent effects of cannabinoids on cancer incidence noted in previous studies and suggest that more research is needed to understand the potential role of human papillomavirus infection.[14] A systematic review and meta-analysis of nine case-control studies involving 13,931 participants also concluded that there was insufficient evidence to demonstrate a positive or negative association between Cannabis smoking and the incidence of head and neck cancers.[15]
Stimulated by the hypothesis that chronic marijuana use produces adverse effects on the human endocrine and reproductive systems, the association between Cannabis use and incidence of testicular germ cell tumors (TGCTs) has been examined.[16-18] Three population-based case-control studies reported an association between Cannabis use and elevated risk of TGCTs, especially nonseminoma or mixed-histology tumors.[16-18] However, the sample sizes in these studies were inadequate to address Cannabis dose by addressing associations among recency, frequency, and duration of use. In a study of 49,343 Swedish men aged 19 to 21 years enrolled in the military between 1969 and 1970, participants were asked at the time of conscription about their use of Cannabis and were followed up for 42 years.[19] This study found no significant correlation between “ever” Cannabis use and the development of testicular cancer, but it did find that “heavy” Cannabis use (more than 50 times in a lifetime) was associated with a 2.5-fold increased risk. Limitations of the study were that it relied on indirect assessment of Cannabis use, and no information was collected on Cannabis use after the conscription-assessment period or on whether the testicular cancers were seminoma or nonseminoma subtypes. These reports established the need for larger, well-powered, prospective studies, especially studies evaluating the role of endocannabinoid signaling and cannabinoid receptors in TGCTs.
An analysis of 84,170 participants in the California Men’s Health Study was performed to investigate the association between Cannabis use and the incidence of bladder cancer. During 16 years of follow-up, 89 Cannabis users (0.3%) developed bladder cancer compared with 190 (0.4%) of the men who did not report Cannabis use (P < .001). After adjusting for age, race, ethnicity, and body mass index, Cannabis use was associated with a 45% reduction in bladder cancer incidence (hazard ratio, 0.55; 95% confidence interval [CI], 0.33–1.00).[20]
A comprehensive Health Canada monograph on marijuana concluded that while there are many cellular and molecular studies that provide strong evidence that inhaled marijuana is carcinogenic, the epidemiological evidence of a link between marijuana use and cancer is still inconclusive.[21]
Patterns of Cannabis Use Among Cancer Patients
A cross-sectional survey of patients with cancer seen at the Seattle Cancer Care Alliance was conducted over a 6-week period between 2015 and 2016.[22] In Washington State, Cannabis was legalized for medicinal use in 1998 and for recreational use in 2012. Of the 2,737 possible participants, 936 (34%) completed the anonymous questionnaire. Twenty-four percent of patients considered themselves active Cannabis users. Similar numbers of patients inhaled (70%) or used edibles (70%), with dual use (40%) being common. Non–mutually exclusive reasons for Cannabis use were physical symptoms (75%), neuropsychiatric symptoms (63%), recreational use/enjoyment (35%), and treatment of cancer (26%). The physical symptoms most commonly cited were pain, nausea, and loss of appetite. Most patients (74%) stated that they would prefer to obtain information about Cannabis from their cancer team, but less than 15% reported receiving information from their cancer physician or nurse.
Data from 2,970 Israeli patients with cancer who used government-issued Cannabis were collected over a 6-month period to assess for improvement in baseline symptoms.[23] The most improved symptoms from baseline include the following:
- Nausea and vomiting (91.0%).
- Sleep disorders (87.5%).
- Restlessness (87.5%).
- Anxiety and depression (84.2%).
- Pruritus (82.1%).
- Headaches (81.4%).
Before treatment initiation, 52.9% of patients reported pain scores in the 8 to 10 range, while only 4.6% of patients reported this intensity at the 6-month assessment time point. It is difficult to assess from the observational data if the improvements were caused by the Cannabis or the cancer treatment.[23] Similarly, a study of a subset of patients with cancer in the Minnesota medical Cannabis program explored changes in the severity of eight symptoms (i.e., anxiety, appetite loss, depression, disturbed sleep, fatigue, nausea, pain, and vomiting) experienced by these patients.[24] Significant symptomatic improvements were noted (38.4%–56.2%) in patients with each symptom. Because of the observational and uncontrolled nature of this study, the findings are not generalizable, but as the authors suggested, may be useful in designing more rigorous research studies in the future.
Forty-two percent of women (257 of 612) with a diagnosis of breast cancer within the past 5 years who participated in an anonymous online survey reported using Cannabis for the relief of symptoms, particularly pain (78%), insomnia (70%), anxiety (57%), stress (51%), and nausea and vomiting (46%).[25] Among Cannabis users, 79% used Cannabis during their cancer treatment, and 75% reported that Cannabis was extremely or very helpful for relieving symptoms. Forty-nine percent of Cannabis users felt that Cannabis could be useful in treating the cancer itself. Only 39% of the participants reported discussing Cannabis use with their physicians.
Patients with cancer turn to medical Cannabis use even in states where it has not been legalized. Researchers from the Medical University of South Carolina conducted a survey study that explored the prevalence, patterns, and motivations behind Cannabis use among patients with cancer and survivors in a state without legal access to Cannabis as of 2023.[26] Despite the absence of legal access, a significant proportion of participants reported using Cannabis for symptom management including pain, nausea, and other symptoms. Moreover, participants reported improvements in mood, sleep, and overall better quality of life. This study gives insight into why many patients with cancer and survivors use Cannabis for symptom management, highlighting the importance of understanding and addressing patient' needs even in states with no legal access to Cannabis.
Cancer Treatment
No ongoing clinical trials of Cannabis as a treatment for cancer in humans were identified in a PubMed search. The first published trial of any cannabinoid in patients with cancer was a small pilot study of intratumoral injection of delta-9-THC in patients with recurrent glioblastoma multiforme, which demonstrated no significant clinical benefit.[27,28] A small double-blind exploratory phase IB study, conducted in the United Kingdom, used nabiximols, a 1:1 ratio of THC:CBD in a Cannabis-based medicinal extract oromucosal spray, in conjunction with dose-dense temozolomide in treating patients with recurrent glioblastoma multiforme.[29][Level of evidence: 1iA] Of the 27 patients enrolled, 6 were part of an open-label group and 21 were part of a randomized group (12 treated with nabiximols and 9 treated with placebo). Progression-free survival at 6 months was seen in 33% of patients in both arms of the study. However, at 1 year, 83.3% of the patients who received nabiximols were alive compared with 44.4% of those who received placebo (P = .042). The investigators cautioned that this early-phase study was not powered for a survival end point. Overall survival rates at 2 years continued to favor the nabiximols arm (50%) compared with the placebo arm (22%) (these rates included results for the 6 patients in the open-label group who received nabiximols).[29]
In a 2016 consecutive case series study, nine patients with varying stages of brain tumors, including six with glioblastoma multiforme, received CBD 200 mg twice daily in addition to surgical excision and chemoradiation.[30][Level of evidence: 3iiiA] The authors reported that all but one of the cohort remained alive at the time of publication. However, the heterogeneity of the brain tumor patients probably contributed to the findings.
Another Israeli group postulated that the anti-inflammatory and immunosuppressive effects of CBD might make it a valuable adjunct in the treatment of acute graft-versus-host disease (GVHD) in patients who have undergone allogeneic hematopoietic stem cell transplant. The authors investigated CBD 300 mg/d in addition to standard GVHD prophylaxis in 48 adult patients who had undergone a transplant, predominantly for acute leukemia or myelodysplastic syndrome (NCT01385124 and NCT01596075).[31] The combination of CBD with standard GVHD prophylaxis was found to be safe. Compared with 101 historical controls treated with standard prophylaxis, patients who received CBD appeared to have a lower incidence of grade II to grade IV GVHD, suggesting that a randomized controlled trial (RCT) is warranted.
Clinical data regarding Cannabis as an anticancer agent in pediatric use are limited to a few case reports.[32,33]
A review of medical literature shows evidence of a growing interest in research of the use of Cannabis in cancer management.[34]
Antiemetic Effect
Cannabinoids
Despite advances in pharmacological and nonpharmacological management, nausea and vomiting (N/V) remain distressing side effects for patients with cancer and their families. Dronabinol, a synthetically produced delta-9-THC, was approved in the United States in 1986 as an antiemetic to be used in cancer chemotherapy. Nabilone, a synthetic derivative of delta-9-THC, was first approved in Canada in 1982 and is now also available in the United States.[35] The U.S. Food and Drug Administration (FDA) has approved both dronabinol and nabilone for the treatment of N/V associated with cancer chemotherapy in patients who have failed to respond to conventional antiemetic therapy. Numerous clinical trials and meta-analyses have shown that dronabinol and nabilone are effective in the treatment of N/V induced by chemotherapy.[36-39] The National Comprehensive Cancer Network Guidelines recommend cannabinoids as breakthrough treatment for chemotherapy-related N/V.[40] The American Society for Clinical Oncology (ASCO) antiemetic guidelines updated in 2017 recommends that the FDA-approved cannabinoids, dronabinol or nabilone, be used to treat N/V that is resistant to standard antiemetic therapies.[41]
One systematic review studied 30 randomized comparisons of delta-9-THC preparations with placebo or other antiemetics from which data on efficacy and harm were available.[42] Oral nabilone, oral dronabinol, and intramuscular levonantradol (a synthetic analogue of dronabinol) were tested. Inhaled Cannabis trials were not included. Among all 1,366 patients included in the review, cannabinoids were found to be more effective than the conventional antiemetics prochlorperazine, metoclopramide, chlorpromazine, thiethylperazine, haloperidol, domperidone, and alizapride. Cannabinoids, however, were not more effective for patients receiving very low or very high emetogenic chemotherapy. Side effects included a feeling of being high, euphoria, sedation or drowsiness, dizziness, dysphoria or depression, hallucinations, paranoia, and hypotension.[42]
Another analysis of 15 controlled studies compared nabilone with placebo or available antiemetic drugs.[43] Among 600 patients with cancer, nabilone was found to be superior to prochlorperazine, domperidone, and alizapride, with nabilone favored for continuous use.
A Cochrane meta-analysis of 23 RCTs reviewed studies conducted between 1975 and 1991 that investigated dronabinol or nabilone, either as monotherapy or as an adjunct to the conventional dopamine antagonists that were the standard antiemetics at that time.[44] The chemotherapy regimens involved drugs with low, moderate, or high emetic potential. The meta-analysis graded the quality of evidence as low for most outcomes. The review concluded that individuals were more likely to report complete absence of N/V when they received cannabinoids compared with placebo, although they were more likely to withdraw from the study because of an adverse event. Individuals reported a higher preference for cannabinoids than placebo or prochlorperazine. There was no difference in the antiemetic effect of cannabinoids when compared with prochlorperazine. The authors concluded that Cannabis-based medications may be useful for treating refractory chemotherapy-induced N/V; however, they cautioned that their assessment may change with the availability of newer antiemetic regimens.
At least 50% of patients who receive moderately emetogenic chemotherapy may experience delayed chemotherapy-induced N/V. Although selective neurokinin 1 antagonists that inhibit substance P have been approved for delayed N/V, a study was conducted before their availability to assess dronabinol, ondansetron, or their combination in preventing delayed-onset chemotherapy-induced N/V.[45] Ondansetron, a serotonin 5-hydroxytryptamine 3 (5-HT3) receptor antagonist, is one of the mainstay agents in the current antiemetic armamentarium. In this trial, the primary objective was to assess the response 2 to 5 days after moderately to severely emetogenic chemotherapy. Sixty-one patients were analyzed for efficacy. The total response—a composite end point—including nausea intensity, vomiting/retching, and use of rescue medications, was similar with dronabinol (54%), ondansetron (58%), and combination therapy (47%) when compared with placebo (20%). Nausea absence was greater in the active treatment groups (dronabinol 71%, ondansetron 64%, combination therapy 53%) when compared with placebo (15%; P < .05 vs. placebo for all). Occurrence rates for nausea intensity and vomiting/retching episodes were the lowest in patients treated with dronabinol, suggesting that dronabinol compares favorably with ondansetron in this situation where a substance P inhibitor would currently be the drug of choice.
For more information, see the Cannabis section in Nausea and Vomiting Related to Cancer Treatment.
Cannabis
Three trials have evaluated the efficacy of inhaled Cannabis in chemotherapy-induced N/V.[46-49] In two of the studies, inhaled Cannabis was made available only after dronabinol failure. In the first trial, no antiemetic effect was achieved with marijuana in patients receiving cyclophosphamide or doxorubicin,[46] but in the second trial, a statistically significant superior antiemetic effect of inhaled Cannabis versus placebo was found among patients receiving high-dose methotrexate.[47] The third trial was a randomized, double-blind, placebo-controlled, crossover trial involving 20 adults in which both inhaled marijuana and oral THC were evaluated. One-quarter of the patients reported a favorable antiemetic response to the cannabinoid therapies. This latter study was reported in abstract form in 1984. A full report, detailing the methods and outcomes apparently has not been published, which limits a thorough interpretation of the significance of these findings.[48]
Newer antiemetics (e.g., 5-HT3 receptor antagonists) have not been directly compared with Cannabis or cannabinoids in patients with cancer. However, the Cannabis-extract oromucosal spray, nabiximols, formulated with 1:1 THC:CBD was shown in a small pilot randomized, placebo-controlled, double-blinded clinical trial in Spain to treat chemotherapy-related N/V.[50][Level of evidence: 1iC]
A phase II/III Australian/New Zealand trial included patients with a solid tumor or hematological malignancy of any stage (n = 147). Patients received a THC:CBD capsule or placebo on days -1 to 5 for the secondary prevention of refractory chemotherapy-induced N/V in combination with guideline recommended antiemetics for chemotherapy of moderate or high emetogenic risk.[51] The primary end point was complete response, defined as no emesis or use of rescue medications during hours 0–120. The THC:CBD group had a higher response rate (24%) compared with the placebo group (8%). Increased moderate- to-severe side effects were observed in the intervention group with more frequent sedation, transient anxiety, and dizziness.
ASCO antiemetic guidelines updated in 2020 state that evidence remains insufficient to recommend medical marijuana for either the prevention or treatment of N/V in patients with cancer who receive chemotherapy or radiation therapy.[41]
Appetite Stimulation
Patients with cancer may experience anorexia, early satiety, weight loss, and cachexia. Such patients face not only the disfigurement associated with wasting but also cannot engage in the social interaction of meals.
Cannabinoids
Four controlled trials have assessed the effect of oral THC on measures of appetite, food appreciation, calorie intake, and weight loss in patients with advanced malignancies. Three relatively small, placebo-controlled trials (N = 52; N = 46; N = 65) each found that oral THC produced improvements in one or more of these outcomes.[52-54] The one study that used an active control evaluated the efficacy of dronabinol alone or with megestrol acetate compared with that of megestrol acetate alone for managing cancer-associated anorexia.[55] In this randomized, double-blind study of 469 adults with advanced cancer and weight loss, patients received 2.5 mg of oral THC twice daily, 800 mg of oral megestrol daily, or both. After 8 to 11 weeks of treatment, the megestrol group had a 75% increase in appetite and an 11% increase in weight, compared with a 49% increase in appetite and a 3% increase in weight in the oral THC group. The between-group differences were statistically significant in favor of megestrol acetate. Furthermore, the combined therapy did not offer additional benefits beyond those provided by megestrol acetate alone. The authors concluded that dronabinol, compared with megestrol acetate, did little to promote appetite or weight gain in patients with advanced cancer.
Cannabis
In trials conducted in the 1980s that involved healthy control subjects, inhaling Cannabis led to an increase in caloric intake, mainly in the form of between-meal snacks, with increased intakes of fatty and sweet foods.[56,57]
Despite patients' great interest in oral preparations of Cannabis to improve appetite, there is only one trial of Cannabis extract used for appetite stimulation. In an RCT, researchers compared the safety and effectiveness of orally administered Cannabis extract (2.5 mg THC and 1 mg CBD), THC (2.5 mg), or placebo for the treatment of cancer-related anorexia-cachexia. A total of 243 patients with advanced cancer received treatment twice daily for 6 weeks. While all extracts were well tolerated, no differences were observed in patients' appetite or quality of life among the three groups at this dose level and duration of intervention.[58]
No published studies have explored the effect of inhaled Cannabis on appetite in patients with cancer.
Analgesia
Cannabinoids
Pain management improves a patient’s quality of life throughout all stages of cancer. Through the study of cannabinoid receptors, endocannabinoids, and synthetic agonists and antagonists, the mechanisms of cannabinoid-induced analgesia have been analyzed.[59][Level of evidence: 1iC] The CB1 receptor is found in the central nervous system (CNS) and in peripheral nerve terminals.[60] CB2 receptors are located mainly in peripheral tissue and are expressed in only low amounts in the CNS. Whereas only CB1 agonists exert analgesic activity in the CNS, both CB1 and CB2 agonists have analgesic activity in peripheral tissue.[61,62]
Cancer pain results from inflammation, invasion of bone or other pain-sensitive structures, or nerve injury. When cancer pain is severe and persistent, it is often resistant to treatment with opioids.
Two studies examined the effects of oral delta-9-THC on cancer pain. The first, a double-blind, placebo-controlled study involving ten patients, measured both pain intensity and pain relief.[63] It was reported that 15 mg and 20 mg doses of the cannabinoid delta-9-THC were associated with substantial analgesic effects, with antiemetic effects and appetite stimulation.
In a follow-up, single-dose study involving 36 patients, 10 mg of delta-9-THC produced analgesic effects during a 7-hour observation period that were comparable to 60 mg doses of codeine, and 20 mg doses of delta-9-THC induced effects equivalent to 120 mg doses of codeine.[64] Higher doses of THC were found to be more sedating than codeine.
Another study examined the effects of a plant extract with controlled cannabinoid content in an oromucosal spray. In a multicenter, double-blind, placebo-controlled study, the THC:CBD nabiximols extract and THC extract alone were compared in the analgesic management of patients with advanced cancer and with moderate-to-severe cancer-related pain. Patients were assigned to one of three treatment groups: THC:CBD extract, THC extract, or placebo. The researchers concluded that the THC:CBD extract was efficacious for pain relief in patients with advanced cancer whose pain was not fully relieved by strong opioids.[65] In a randomized, placebo-controlled, graded-dose trial, opioid-treated patients with cancer with poorly controlled chronic pain demonstrated significantly better control of pain and sleep disruption with THC:CBD oromucosal spray at lower doses (1–4 and 6–10 sprays/d), compared with placebo. Adverse events were dose related, with only the high-dose group (11–16 sprays/d) comparing unfavorably with the placebo arm. These studies provide promising evidence of an adjuvant analgesic effect of THC:CBD in this opioid-refractory patient population and may provide an opportunity to address this significant clinical challenge.[66] An open-label extension study of 43 patients who had participated in the randomized trial found that some patients continued to obtain relief of their cancer-related pain with long-term use of the THC:CBD oromucosal spray without increasing their dose of the spray or the dose of their other analgesics.[67]
An observational study assessed the effectiveness of nabilone in patients with advanced cancer who were experiencing pain and other symptoms (anorexia, depression, and anxiety). The researchers reported that patients who used nabilone experienced improved management of pain, nausea, anxiety, and distress when compared with untreated patients. Nabilone was also associated with a decreased use of opioids, nonsteroidal anti-inflammatory drugs, tricyclic antidepressants, gabapentin, dexamethasone, metoclopramide, and ondansetron.[68]
Cannabis
Animal studies have suggested a synergistic analgesic effect when cannabinoids are combined with opioids. The results from one pharmacokinetic interaction study have been reported. In this study, 21 patients with chronic pain were administered vaporized Cannabis along with sustained-release morphine or oxycodone for 5 days.[69] The patients who received vaporized Cannabis and sustained-release morphine had a statistically significant decrease in their mean pain score over the 5-day period; those who received vaporized Cannabis and oxycodone did not. These findings should be verified by further studies before recommendations favoring such an approach are warranted in general clinical practice.
Patients with cancer may experience neuropathic pain, especially if treated with platinum-based chemotherapy or taxanes. Two RCTs of inhaled Cannabis in patients with peripheral neuropathy or neuropathic pain of various etiologies found that pain was reduced in patients who received inhaled Cannabis, compared with those who received placebo.[70,71] A retrospective analysis examined the effect of Cannabis on chemotherapy-induced peripheral neuropathy (CIPN) in Israeli patients with cancer who received oxaliplatin-based regimens for gastrointestinal malignancies.[72][Level of evidence: 2Diii] Patients were divided into three groups on the basis of their exposure to Cannabis: Cannabis-first group (received Cannabis before starting oxaliplatin), oxaliplatin-first group (received oxaliplatin before starting Cannabis), and controls (no Cannabis use). A significant difference in grade 2 to 3 CIPN was seen between the Cannabis-exposed patients (15.3%) and controls (27.9%) (P < .001). The neuropathy-sparing effect was more pronounced among those treated with Cannabis first (75%) compared with those who received oxaliplatin first (46.2%) (P < .001). Some limitations of this study were its retrospective design and documentation of Cannabis use as qualitative, not quantitative.
A randomized, placebo-controlled, crossover, pilot study of nabiximols in 16 patients with chemotherapy-induced neuropathic pain showed no significant difference between the treatment and placebo groups. A responder analysis, however, demonstrated that five patients reported a reduction in their pain of at least 2 points on an 11-point scale, suggesting that a larger follow-up study may be warranted.[73]
One real-world randomized controlled trial explored Cannabis use in patients with advanced cancer who received care in a community oncology practice setting (148 screened; 30 randomly assigned; 18 analyzed).[74] Once certified by their oncologists, participants were randomly assigned to receive early Cannabis (EC) or delayed start of medical Cannabis (DC) for 3 months as part of a state-sponsored Cannabis program. The EC group had stable opioid usage compared with the DC group who had an increase in opioid usage during the 3-month study period. Overall, there were no significant changes in quality of life or symptom scores between the groups, with no overall Cannabis-related adverse events. Limitations included a variety of cancer types and no consistent use of Cannabis products (108 different Cannabis products were dispensed during the study period).
Anxiety and Sleep
Cannabinoids
In a small pilot study of analgesia involving ten patients with cancer pain, secondary measures showed that 15 mg and 20 mg doses of the cannabinoid delta-9-THC were associated with anxiolytic effects.[63][Level of evidence: 1iC]
A small placebo-controlled study of dronabinol in patients with cancer with altered chemosensory perception also noted increased quality of sleep and relaxation in THC-treated patients.[53][Level of evidence: 1iC]
Cannabis
Patients often experience mood elevation after exposure to Cannabis, depending on their previous experience. In a five-patient case series of inhaled Cannabis that examined analgesic effects in chronic pain, it was reported that patients who self-administered Cannabis had improved mood, improved sense of well-being, and less anxiety.[75]
Another common effect of Cannabis is sleepiness. A small placebo-controlled study of dronabinol in patients with cancer with altered chemosensory perception also noted increased quality of sleep and relaxation in THC-treated patients.[53]
Seventy-four patients with newly diagnosed head and neck cancer self-described as current Cannabis users were matched to 74 nonusers in a Canadian study investigating quality of life using the EuroQol-5D and Edmonton Symptom Assessment System instruments.[76] Cannabis users had significantly lower scores in the anxiety/depression (difference, 0.74; 95% CI, 0.557–0.930) and pain/discomfort (difference, 0.29; 95% CI, 0.037–1.541) domains. Cannabis users were also less tired, had more appetite, and better general well-being.
A single center, phase II, double-blind study of two ratios (1:1 [THC:CBD] and 4:1 [THC:CBD]) of an oral medical Cannabis oil enrolled patients with recurrent or inoperable high-grade glioma. Investigators assessed the side effects and Functional Assessment of Cancer Therapy-Brain (FACT-Br) at baseline and 12 weeks as a primary outcome.[77] There was no difference in the primary end point; however, some significant differences were noted in the subscores of the FACT-Br (i.e., physical, functional, and sleep favored the 1:1 ratio) and these end points would be appropriate for future research.
Symptom Management With Cannabidiol
A randomized, double-blind, placebo-controlled trial (n = 144) assessed the impact of oral cannabidiol oil (50–200 mg three times a day) on the total symptom distress score (TSDS), a measure of overall cancer symptom burden.[78] All patients received baseline care from palliative care specialists. No significant difference was found in the median TSDS between the two arms at the two assessment points (day 14 and day 28). This study had several limitations, including heterogeneity of the patient population, low symptom burden of patients at baseline, and a high level of patient withdrawals from both study arms.
Pediatric Population and Cannabis and Cannabinoid Medicinal Use
A growing number of pediatric patients are seeking symptom relief with Cannabis or cannabinoid treatment, although studies are limited.[79] The American Academy of Pediatrics has not endorsed Cannabis and cannabinoid use because of concerns about brain development.
A retrospective study from Israel of 50 pediatric oncology patients who were prescribed medicinal Cannabis over an 8-year period reported that the most common indications included the following:[80]
- Nausea and vomiting.
- Depressed mood.
- Sleep disturbances.
- Poor appetite and weight loss.
- Pain.
Most of the patients (n = 30) received Cannabis in the form of oral oil drops, with some of the older children inhaling vaporized Cannabis or combining inhalation with oral oils. Structured interviews with the parents, and their child when appropriate, revealed that 40 participants (80%) reported a high level of general satisfaction with the use of Cannabis with infrequent short-term side effects.[80] Survey studies revealed that most responding pediatricians in the United States and Canada supported the use of medical Cannabis for symptom management in pediatric patients with cancer.[81,82]
Clinical Studies of Cannabis and Cannabinoids
| Reference | Trial Design | Condition or Cancer Type | Treatment Groups (Enrolled; Treated; Placebo or No Treatment Control)b | Resultsc | Concurrent Therapy Usedd | Level of Evidence Scoree |
|---|---|---|---|---|---|---|
| 5-HT3 = 5-hydroxytryptamine 3; CINV = chemotherapy-induced nausea and vomiting; N/V = nausea and vomiting; RCT = randomized controlled trial. | ||||||
| aFor additional information and definition of terms, see text and the NCI Dictionary of Cancer Terms. | ||||||
| bNumber of patients treated plus number of patient controls may not equal number of patients enrolled; number of patients enrolled equals number of patients initially recruited/considered by the researchers who conducted a study; number of patients treated equals number of enrolled patients who were given the treatment being studied AND for whom results were reported. | ||||||
| cStrongest evidence reported that the treatment under study has activity or otherwise improves the well-being of patients with cancer. | ||||||
| dConcurrent therapy for symptoms treated (not cancer). | ||||||
| eFor information about levels of evidence analysis and scores, see Levels of Evidence for Human Studies of Integrative, Alternative, and Complementary Therapies. | ||||||
| [77] | RCT | High-grade gliomas | 88; 45 (1:1), 43 (4:1); None | No difference in the primary end point | Dexamethasone, temozolomide, bevacizumab, lomustine | 1iC |
| [46] | RCT | CINV | 8; 8; None | No antiemetic effect reported | No | 1iC |
| [47] | RCT | CINV | 15; 15; None | Decreased N/V | No | 1iiC |
| [50] | Pilot RCT | CINV | 16; 7; 9 | Decreased delayed N/V | 5-HT3 receptor antagonists | 1iC |
| [69] | Nonrandomized trial | Chronic pain | 21;10 (morphine), 11 (oxycodone); None | Decreased pain | Yes, morphine, oxycodone | 2C |
| [76] | Prospective cohort study | Anxiety, pain, depression, loss of appetite | 148; 74; 74 | Decreased pain, anxiety, depression, increased appetite | Unknown | 2C |
| Reference | Trial Design | Condition or Cancer Type | Treatment Groups (Enrolled; Treated; Placebo or No Treatment Control)b | Resultsc | Concurrent Therapy Usedd | Level of Evidence Scoree |
|---|---|---|---|---|---|---|
| CBD = cannabidiol; No. = number; NSAIDs = nonsteroidal anti-inflammatory drugs; QoL = quality of life; RCT = randomized controlled trial; THC = delta-9-tetrahydrocannabinol. | ||||||
| aFor additional information and definition of terms, see text and the NCI Dictionary of Cancer Terms. | ||||||
| bNumber of patients treated plus number of patient controls may not equal number of patients enrolled; number of patients enrolled equals number of patients initially recruited/considered by the researchers who conducted a study; number of patients treated equals number of enrolled patients who were given the treatment being studied AND for whom results were reported. | ||||||
| cStrongest evidence reported that the treatment under study has activity or otherwise improves the well-being of patients with cancer. | ||||||
| dConcurrent therapy for symptoms treated (not cancer). | ||||||
| eFor information about levels of evidence analysis and scores, see Levels of Evidence for Human Studies of Integrative, Alternative, and Complementary Therapies. | ||||||
| [55] | RCT | Cancer-associated anorexia | 469; dronabinol 152, megestrol acetate 159, or both 158; None | Megestrol acetate provided increased appetite and weight gain, among patients with advanced cancer compared with dronabinol alone | No | 1iC |
| [53] | Pilot RCT | Appetite | 21; 11; 10 | THC, compared with placebo, improved and enhanced taste and smell | No | 1iC |
| [58] | RCT | Cancer-related anorexia-cachexia syndrome | 243; Cannabis extract 95, THC 100; 48 | No differences in patients’ appetite or QoL were found | No | 1iC |
| [83] | RCT | Appetite | 139; 72; 67 | Increase in appetite | No | 1iC |
| [54] | RCT | Anorexia | 47; 22; 25 | Increased calorie intake | No | 1iC |
| [63] | RCT | Pain | 10; 10; None | Pain relief | No | 1iC |
| [65] | RCT | Pain | 177; 60 (THC:CBD), 58 (THC); 59 | THC:CBD extract group had reduced pain | Yes, opioids | 1iC |
| [66] | RCT | Pain | 360; 269; 91 | Decreased pain in low-dose group | Yes, opioids | 1iC |
| [67] | Open-label extension | Pain | 43; 39 (THC:CBD), 4 (THC), None | Decreased pain | Yes, opioids | 2C |
| [68] | Observational study | Pain | 112; 47; 65 | Decreased pain | Yes, opioids, NSAIDs, gabapentin | 2C |
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Adams IB, Martin BR: Cannabis: pharmacology and toxicology in animals and humans. Addiction 91 (11): 1585-614, 1996. [PUBMED Abstract]
- Agurell S, Halldin M, Lindgren JE, et al.: Pharmacokinetics and metabolism of delta 1-tetrahydrocannabinol and other cannabinoids with emphasis on man. Pharmacol Rev 38 (1): 21-43, 1986. [PUBMED Abstract]
- Yamamoto I, Watanabe K, Narimatsu S, et al.: Recent advances in the metabolism of cannabinoids. Int J Biochem Cell Biol 27 (8): 741-6, 1995. [PUBMED Abstract]
- Engels FK, de Jong FA, Sparreboom A, et al.: Medicinal cannabis does not influence the clinical pharmacokinetics of irinotecan and docetaxel. Oncologist 12 (3): 291-300, 2007. [PUBMED Abstract]
- FDA Warns Companies Marketing Unproven Products, Derived From Marijuana, That Claim to Treat or Cure Cancer [News Release]. Silver Spring, Md: Food and Drug Administration, 2017. Available online. Last accessed January 4, 2019.
- Yamaori S, Okamoto Y, Yamamoto I, et al.: Cannabidiol, a major phytocannabinoid, as a potent atypical inhibitor for CYP2D6. Drug Metab Dispos 39 (11): 2049-56, 2011. [PUBMED Abstract]
- Jiang R, Yamaori S, Okamoto Y, et al.: Cannabidiol is a potent inhibitor of the catalytic activity of cytochrome P450 2C19. Drug Metab Pharmacokinet 28 (4): 332-8, 2013. [PUBMED Abstract]
- Azwell T, Ciotti C, Adams A: Variation among hemp (Cannabis sativus L.) analytical testing laboratories evinces regulatory and quality control issues for the industry. J Appl Res Med Aromat Plants 31: 100434, 2022. Available online. Last accessed October 17, 2024.
- Berthiller J, Straif K, Boniol M, et al.: Cannabis smoking and risk of lung cancer in men: a pooled analysis of three studies in Maghreb. J Thorac Oncol 3 (12): 1398-403, 2008. [PUBMED Abstract]
- Sidney S, Quesenberry CP, Friedman GD, et al.: Marijuana use and cancer incidence (California, United States). Cancer Causes Control 8 (5): 722-8, 1997. [PUBMED Abstract]
- Hashibe M, Morgenstern H, Cui Y, et al.: Marijuana use and the risk of lung and upper aerodigestive tract cancers: results of a population-based case-control study. Cancer Epidemiol Biomarkers Prev 15 (10): 1829-34, 2006. [PUBMED Abstract]
- Mehra R, Moore BA, Crothers K, et al.: The association between marijuana smoking and lung cancer: a systematic review. Arch Intern Med 166 (13): 1359-67, 2006. [PUBMED Abstract]
- National Academies of Sciences, Engineering, and Medicine: The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. The National Academies Press, 2017.
- Marks MA, Chaturvedi AK, Kelsey K, et al.: Association of marijuana smoking with oropharyngeal and oral tongue cancers: pooled analysis from the INHANCE consortium. Cancer Epidemiol Biomarkers Prev 23 (1): 160-71, 2014. [PUBMED Abstract]
- de Carvalho MF, Dourado MR, Fernandes IB, et al.: Head and neck cancer among marijuana users: a meta-analysis of matched case-control studies. Arch Oral Biol 60 (12): 1750-5, 2015. [PUBMED Abstract]
- Daling JR, Doody DR, Sun X, et al.: Association of marijuana use and the incidence of testicular germ cell tumors. Cancer 115 (6): 1215-23, 2009. [PUBMED Abstract]
- Trabert B, Sigurdson AJ, Sweeney AM, et al.: Marijuana use and testicular germ cell tumors. Cancer 117 (4): 848-53, 2011. [PUBMED Abstract]
- Lacson JC, Carroll JD, Tuazon E, et al.: Population-based case-control study of recreational drug use and testis cancer risk confirms an association between marijuana use and nonseminoma risk. Cancer 118 (21): 5374-83, 2012. [PUBMED Abstract]
- Callaghan RC, Allebeck P, Akre O, et al.: Cannabis Use and Incidence of Testicular Cancer: A 42-Year Follow-up of Swedish Men between 1970 and 2011. Cancer Epidemiol Biomarkers Prev 26 (11): 1644-1652, 2017. [PUBMED Abstract]
- Thomas AA, Wallner LP, Quinn VP, et al.: Association between cannabis use and the risk of bladder cancer: results from the California Men's Health Study. Urology 85 (2): 388-92, 2015. [PUBMED Abstract]
- Health Canada: Marihuana (Marijuana, Cannabis): Dried Plant for Administration by Ingestion or Other Means. Ottawa, Canada: Health Canada, 2010. Available online. Last accessed October 18, 2017.
- Pergam SA, Woodfield MC, Lee CM, et al.: Cannabis use among patients at a comprehensive cancer center in a state with legalized medicinal and recreational use. Cancer 123 (22): 4488-4497, 2017. [PUBMED Abstract]
- Bar-Lev Schleider L, Mechoulam R, Lederman V, et al.: Prospective analysis of safety and efficacy of medical cannabis in large unselected population of patients with cancer. Eur J Intern Med 49: 37-43, 2018. [PUBMED Abstract]
- Anderson SP, Zylla DM, McGriff DM, et al.: Impact of Medical Cannabis on Patient-Reported Symptoms for Patients With Cancer Enrolled in Minnesota's Medical Cannabis Program. J Oncol Pract 15 (4): e338-e345, 2019. [PUBMED Abstract]
- Weiss MC, Hibbs JE, Buckley ME, et al.: A Coala-T-Cannabis Survey Study of breast cancer patients' use of cannabis before, during, and after treatment. Cancer 128 (1): 160-168, 2022. [PUBMED Abstract]
- McClure EA, Walters KJ, Tomko RL, et al.: Cannabis use prevalence, patterns, and reasons for use among patients with cancer and survivors in a state without legal cannabis access. Support Care Cancer 31 (7): 429, 2023. [PUBMED Abstract]
- Guzmán M, Duarte MJ, Blázquez C, et al.: A pilot clinical study of Delta9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br J Cancer 95 (2): 197-203, 2006. [PUBMED Abstract]
- Velasco G, Sánchez C, Guzmán M: Towards the use of cannabinoids as antitumour agents. Nat Rev Cancer 12 (6): 436-44, 2012. [PUBMED Abstract]
- Twelves C, Sabel M, Checketts D, et al.: A phase 1b randomised, placebo-controlled trial of nabiximols cannabinoid oromucosal spray with temozolomide in patients with recurrent glioblastoma. Br J Cancer 124 (8): 1379-1387, 2021. [PUBMED Abstract]
- Likar R, Koestenberger M, Stultschnig M, et al.: Concomitant Treatment of Malignant Brain Tumours With CBD - A Case Series and Review of the Literature. Anticancer Res 39 (10): 5797-5801, 2019. [PUBMED Abstract]
- Yeshurun M, Shpilberg O, Herscovici C, et al.: Cannabidiol for the Prevention of Graft-versus-Host-Disease after Allogeneic Hematopoietic Cell Transplantation: Results of a Phase II Study. Biol Blood Marrow Transplant 21 (10): 1770-5, 2015. [PUBMED Abstract]
- Singh Y, Bali C: Cannabis extract treatment for terminal acute lymphoblastic leukemia with a Philadelphia chromosome mutation. Case Rep Oncol 6 (3): 585-92, 2013. [PUBMED Abstract]
- Foroughi M, Hendson G, Sargent MA, et al.: Spontaneous regression of septum pellucidum/forniceal pilocytic astrocytomas--possible role of Cannabis inhalation. Childs Nerv Syst 27 (4): 671-9, 2011. [PUBMED Abstract]
- Goyal S, Kubendran S, Kogan M, et al.: High expectations: The landscape of clinical trials of medical marijuana in oncology. Complement Ther Med 49: 102336, 2020. [PUBMED Abstract]
- Sutton IR, Daeninck P: Cannabinoids in the management of intractable chemotherapy-induced nausea and vomiting and cancer-related pain. J Support Oncol 4 (10): 531-5, 2006 Nov-Dec. [PUBMED Abstract]
- Ahmedzai S, Carlyle DL, Calder IT, et al.: Anti-emetic efficacy and toxicity of nabilone, a synthetic cannabinoid, in lung cancer chemotherapy. Br J Cancer 48 (5): 657-63, 1983. [PUBMED Abstract]
- Chan HS, Correia JA, MacLeod SM: Nabilone versus prochlorperazine for control of cancer chemotherapy-induced emesis in children: a double-blind, crossover trial. Pediatrics 79 (6): 946-52, 1987. [PUBMED Abstract]
- Johansson R, Kilkku P, Groenroos M: A double-blind, controlled trial of nabilone vs. prochlorperazine for refractory emesis induced by cancer chemotherapy. Cancer Treat Rev 9 (Suppl B): 25-33, 1982. [PUBMED Abstract]
- Niiranen A, Mattson K: A cross-over comparison of nabilone and prochlorperazine for emesis induced by cancer chemotherapy. Am J Clin Oncol 8 (4): 336-40, 1985. [PUBMED Abstract]
- National Comprehensive Cancer Network: NCCN Clinical Practice Guidelines in Oncology: Antiemesis. Version 1.2021. Plymouth Meeting, Pa: National Comprehensive Cancer Network, 2021. Available online with free registration. Last accessed August 26, 2021..
- Hesketh PJ, Kris MG, Basch E, et al.: Antiemetics: ASCO Guideline Update. J Clin Oncol 38 (24): 2782-2797, 2020. [PUBMED Abstract]
- Tramèr MR, Carroll D, Campbell FA, et al.: Cannabinoids for control of chemotherapy induced nausea and vomiting: quantitative systematic review. BMJ 323 (7303): 16-21, 2001. [PUBMED Abstract]
- Ben Amar M: Cannabinoids in medicine: A review of their therapeutic potential. J Ethnopharmacol 105 (1-2): 1-25, 2006. [PUBMED Abstract]
- Smith LA, Azariah F, Lavender VT, et al.: Cannabinoids for nausea and vomiting in adults with cancer receiving chemotherapy. Cochrane Database Syst Rev (11): CD009464, 2015. [PUBMED Abstract]
- Meiri E, Jhangiani H, Vredenburgh JJ, et al.: Efficacy of dronabinol alone and in combination with ondansetron versus ondansetron alone for delayed chemotherapy-induced nausea and vomiting. Curr Med Res Opin 23 (3): 533-43, 2007. [PUBMED Abstract]
- Chang AE, Shiling DJ, Stillman RC, et al.: A prospective evaluation of delta-9-tetrahydrocannabinol as an antiemetic in patients receiving adriamycin and cytoxan chemotherapy. Cancer 47 (7): 1746-51, 1981. [PUBMED Abstract]
- Chang AE, Shiling DJ, Stillman RC, et al.: Delta-9-tetrahydrocannabinol as an antiemetic in cancer patients receiving high-dose methotrexate. A prospective, randomized evaluation. Ann Intern Med 91 (6): 819-24, 1979. [PUBMED Abstract]
- Levitt M, Faiman C, Hawks R, et al.: Randomized double blind comparison of delta-9-tetrahydrocannabinol and marijuana as chemotherapy antiemetics. [Abstract] Proceedings of the American Society of Clinical Oncology 3: A-C354, 91, 1984.
- Musty RE, Rossi R: Effects of smoked cannabis and oral delta-9-tetrahydrocannabinol on nausea and emesis after cancer chemotherapy: a review of state clinical trials. Journal of Cannabis Therapeutics 1 (1): 29-56, 2001. Also available online. Last accessed October 18, 2017.
- Duran M, Pérez E, Abanades S, et al.: Preliminary efficacy and safety of an oromucosal standardized cannabis extract in chemotherapy-induced nausea and vomiting. Br J Clin Pharmacol 70 (5): 656-63, 2010. [PUBMED Abstract]
- Grimison P, Mersiades A, Kirby A, et al.: Oral Cannabis Extract for Secondary Prevention of Chemotherapy-Induced Nausea and Vomiting: Final Results of a Randomized, Placebo-Controlled, Phase II/III Trial. J Clin Oncol 42 (34): 4040-4050, 2024. [PUBMED Abstract]
- Regelson W, Butler JR, Schulz J, et al.: Delta-9-tetrahydrocannabinol as an effective antidepressant and appetite-stimulating agent in advanced cancer patients. In: Braude MC, Szara S: The Pharmacology of Marihuana. Raven Press, 1976, pp 763-76.
- Brisbois TD, de Kock IH, Watanabe SM, et al.: Delta-9-tetrahydrocannabinol may palliate altered chemosensory perception in cancer patients: results of a randomized, double-blind, placebo-controlled pilot trial. Ann Oncol 22 (9): 2086-93, 2011. [PUBMED Abstract]
- Turcott JG, Del Rocío Guillen Núñez M, Flores-Estrada D, et al.: The effect of nabilone on appetite, nutritional status, and quality of life in lung cancer patients: a randomized, double-blind clinical trial. Support Care Cancer 26 (9): 3029-3038, 2018. [PUBMED Abstract]
- Jatoi A, Windschitl HE, Loprinzi CL, et al.: Dronabinol versus megestrol acetate versus combination therapy for cancer-associated anorexia: a North Central Cancer Treatment Group study. J Clin Oncol 20 (2): 567-73, 2002. [PUBMED Abstract]
- Foltin RW, Brady JV, Fischman MW: Behavioral analysis of marijuana effects on food intake in humans. Pharmacol Biochem Behav 25 (3): 577-82, 1986. [PUBMED Abstract]
- Foltin RW, Fischman MW, Byrne MF: Effects of smoked marijuana on food intake and body weight of humans living in a residential laboratory. Appetite 11 (1): 1-14, 1988. [PUBMED Abstract]
- Strasser F, Luftner D, Possinger K, et al.: Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J Clin Oncol 24 (21): 3394-400, 2006. [PUBMED Abstract]
- Aggarwal SK: Cannabinergic pain medicine: a concise clinical primer and survey of randomized-controlled trial results. Clin J Pain 29 (2): 162-71, 2013. [PUBMED Abstract]
- Walker JM, Hohmann AG, Martin WJ, et al.: The neurobiology of cannabinoid analgesia. Life Sci 65 (6-7): 665-73, 1999. [PUBMED Abstract]
- Calignano A, La Rana G, Giuffrida A, et al.: Control of pain initiation by endogenous cannabinoids. Nature 394 (6690): 277-81, 1998. [PUBMED Abstract]
- Fields HL, Meng ID: Watching the pot boil. Nat Med 4 (9): 1008-9, 1998. [PUBMED Abstract]
- Noyes R, Brunk SF, Baram DA, et al.: Analgesic effect of delta-9-tetrahydrocannabinol. J Clin Pharmacol 15 (2-3): 139-43, 1975 Feb-Mar. [PUBMED Abstract]
- Noyes R, Brunk SF, Avery DA, et al.: The analgesic properties of delta-9-tetrahydrocannabinol and codeine. Clin Pharmacol Ther 18 (1): 84-9, 1975. [PUBMED Abstract]
- Johnson JR, Burnell-Nugent M, Lossignol D, et al.: Multicenter, double-blind, randomized, placebo-controlled, parallel-group study of the efficacy, safety, and tolerability of THC:CBD extract and THC extract in patients with intractable cancer-related pain. J Pain Symptom Manage 39 (2): 167-79, 2010. [PUBMED Abstract]
- Portenoy RK, Ganae-Motan ED, Allende S, et al.: Nabiximols for opioid-treated cancer patients with poorly-controlled chronic pain: a randomized, placebo-controlled, graded-dose trial. J Pain 13 (5): 438-49, 2012. [PUBMED Abstract]
- Johnson JR, Lossignol D, Burnell-Nugent M, et al.: An open-label extension study to investigate the long-term safety and tolerability of THC/CBD oromucosal spray and oromucosal THC spray in patients with terminal cancer-related pain refractory to strong opioid analgesics. J Pain Symptom Manage 46 (2): 207-18, 2013. [PUBMED Abstract]
- Maida V, Ennis M, Irani S, et al.: Adjunctive nabilone in cancer pain and symptom management: a prospective observational study using propensity scoring. J Support Oncol 6 (3): 119-24, 2008. [PUBMED Abstract]
- Abrams DI, Couey P, Shade SB, et al.: Cannabinoid-opioid interaction in chronic pain. Clin Pharmacol Ther 90 (6): 844-51, 2011. [PUBMED Abstract]
- Wilsey B, Marcotte T, Deutsch R, et al.: Low-dose vaporized cannabis significantly improves neuropathic pain. J Pain 14 (2): 136-48, 2013. [PUBMED Abstract]
- Wilsey B, Marcotte T, Tsodikov A, et al.: A randomized, placebo-controlled, crossover trial of cannabis cigarettes in neuropathic pain. J Pain 9 (6): 506-21, 2008. [PUBMED Abstract]
- Waissengrin B, Mirelman D, Pelles S, et al.: Effect of cannabis on oxaliplatin-induced peripheral neuropathy among oncology patients: a retrospective analysis. Ther Adv Med Oncol 13: 1758835921990203, 2021. [PUBMED Abstract]
- Lynch ME, Cesar-Rittenberg P, Hohmann AG: A double-blind, placebo-controlled, crossover pilot trial with extension using an oral mucosal cannabinoid extract for treatment of chemotherapy-induced neuropathic pain. J Pain Symptom Manage 47 (1): 166-73, 2014. [PUBMED Abstract]
- Zylla DM, Eklund J, Gilmore G, et al.: A randomized trial of medical cannabis in patients with stage IV cancers to assess feasibility, dose requirements, impact on pain and opioid use, safety, and overall patient satisfaction. Support Care Cancer 29 (12): 7471-7478, 2021. [PUBMED Abstract]
- Noyes R, Baram DA: Cannabis analgesia. Compr Psychiatry 15 (6): 531-5, 1974 Nov-Dec. [PUBMED Abstract]
- Zhang H, Xie M, Archibald SD, et al.: Association of Marijuana Use With Psychosocial and Quality of Life Outcomes Among Patients With Head and Neck Cancer. JAMA Otolaryngol Head Neck Surg 144 (11): 1017-1022, 2018. [PUBMED Abstract]
- Schloss J, Lacey J, Sinclair J, et al.: A Phase 2 Randomised Clinical Trial Assessing the Tolerability of Two Different Ratios of Medicinal Cannabis in Patients With High Grade Gliomas. Front Oncol 11: 649555, 2021. [PUBMED Abstract]
- Hardy J, Greer R, Huggett G, et al.: Phase IIb Randomized, Placebo-Controlled, Dose-Escalating, Double-Blind Study of Cannabidiol Oil for the Relief of Symptoms in Advanced Cancer (MedCan1-CBD). J Clin Oncol 41 (7): 1444-1452, 2023. [PUBMED Abstract]
- Chhabra M, Ben-Eltriki M, Paul A, et al.: Cannabinoids for symptom management in children with cancer: A systematic review and meta-analysis. Cancer 129 (22): 3656-3670, 2023. [PUBMED Abstract]
- Ofir R, Bar-Sela G, Weyl Ben-Arush M, et al.: Medical marijuana use for pediatric oncology patients: single institution experience. Pediatr Hematol Oncol 36 (5): 255-266, 2019. [PUBMED Abstract]
- Oberoi S, Protudjer JLP, Rapoport A, et al.: Perspectives of pediatric oncologists and palliative care physicians on the therapeutic use of cannabis in children with cancer. Cancer Rep (Hoboken) 5 (9): e1551, 2022. [PUBMED Abstract]
- Ananth P, Ma C, Al-Sayegh H, et al.: Provider Perspectives on Use of Medical Marijuana in Children With Cancer. Pediatrics 141 (1): , 2018. [PUBMED Abstract]
- Beal JE, Olson R, Laubenstein L, et al.: Dronabinol as a treatment for anorexia associated with weight loss in patients with AIDS. J Pain Symptom Manage 10 (2): 89-97, 1995. [PUBMED Abstract]
Adverse Effects
Cannabis and Cannabinoids
Because cannabinoid receptors, unlike opioid receptors, are not located in the brainstem areas controlling respiration, lethal overdoses from Cannabis and cannabinoids do not occur.[1-4] However, cannabinoid receptors are present in other tissues throughout the body, not just in the central nervous system, and adverse effects include the following:
- Tachycardia.
- Hypotension.
- Conjunctival injection.
- Bronchodilation.
- Muscle relaxation.
- Decreased gastrointestinal motility.
Although cannabinoids are considered by some to be addictive drugs, their addictive potential is considerably lower than that of other prescribed agents or substances of abuse.[2,4] The brain develops a tolerance to cannabinoids.
Withdrawal symptoms such as irritability, insomnia with sleep electroencephalogram disturbance, restlessness, hot flashes, and, rarely, nausea and cramping have been observed. However, these symptoms appear to be mild compared with withdrawal symptoms associated with opiates or benzodiazepines, and the symptoms usually dissipate after a few days.
Unlike other commonly used drugs, cannabinoids are stored in adipose tissue and excreted at a low rate (half-life 1–3 days), so even abrupt cessation of cannabinoid intake is not associated with rapid declines in plasma concentrations that would precipitate severe or abrupt withdrawal symptoms or drug cravings.
Cannabidiol (CBD) is an inhibitor of cytochrome P450 isoforms in vitro. Because many anticancer therapies are metabolized by these enzymes, highly concentrated CBD oils used concurrently could potentially increase the toxicity or decrease the effectiveness of these therapies.[5-7]
Since Cannabis smoke contains many of the same components as tobacco smoke, there are valid concerns about the adverse pulmonary effects of inhaled Cannabis. A longitudinal study in a noncancer population evaluated repeated measurements of pulmonary function over 20 years in 5,115 men and women whose smoking histories were known.[8] While tobacco exposure was associated with decreased pulmonary function, the investigators concluded that occasional and low-cumulative Cannabis use was not associated with adverse effects on pulmonary function (forced expiratory volume in the first second of expiration [FEV1] and forced vital capacity [FVC]).
Interactions With Conventional Cancer Therapies
The potential for cytochrome P450 interactions with highly concentrated oil preparations of delta-9-tetrahydrocannabinol and/or cannabidiol is a concern.[9] Few pharmacokinetic interaction studies have been conducted with Cannabis or cannabinoids and conventional cancer therapies. There is insufficient clinical evidence and concern to support the belief that relatively small amounts of active cannabinoids can substantially impact metabolism of relatively large doses of chemotherapy drugs.
An Israeli retrospective observational study assessed the impact of Cannabis use during nivolumab immunotherapy.[10] One hundred forty patients with advanced melanoma, non-small cell lung cancer, and renal cell carcinoma received the checkpoint inhibitor nivolumab (89 patients received nivolumab alone and 51 patients received nivolumab plus Cannabis). In a multivariate model, Cannabis was the only significant factor that reduced the response rate to immunotherapy (37.5% in patients who received nivolumab alone compared with 15.9% in patients who received nivolumab plus Cannabis [odds ratio, 3.13; 95% confidence interval, 1.24–8.1; P = .016]). There was no difference in progression-free survival or overall survival. A subsequent prospective observational study from the same investigators followed 102 patients with metastatic cancers initiating immunotherapy.[11][Level of evidence: 2Dii] Sixty-eight patients received immunotherapy alone while 34 patients used Cannabis during immunotherapy. Over half of the patients in each group had stage IV non-small cell lung cancer. Cannabis users were less likely to receive immunotherapy as a first-line intervention (24%) compared with nonusers (46%) (P = .03). Cannabis users showed a significantly lower percentage of clinical benefit (39% of Cannabis users with complete or partial responses or stable disease compared with 59% of nonusers [P = .035]). In this analysis, the median time to tumor progression was 3.4 months in Cannabis users compared with 13.1 months in nonusers and the overall survival was 6.4 months in Cannabis users compared with 28.5 months in nonusers. The investigators also noted that Cannabis users reported a lower rate of overall treatment-related adverse experiences compared with nonusers, with fewer immune-related adverse events (P = .057). The investigators postulated that this finding may be related to the possible immunosuppressive effects of Cannabis and concluded that Cannabis consumption should be carefully considered in patients with advanced malignancies who are treated with immunotherapy. Limitations noted by the authors that may be confounders in this analysis include the observational nature of the study, the relatively small sample size, and the high heterogeneity of the participants.
References
- Adams IB, Martin BR: Cannabis: pharmacology and toxicology in animals and humans. Addiction 91 (11): 1585-614, 1996. [PUBMED Abstract]
- Grotenhermen F, Russo E, eds.: Cannabis and Cannabinoids: Pharmacology, Toxicology, and Therapeutic Potential. The Haworth Press, 2002.
- Sutton IR, Daeninck P: Cannabinoids in the management of intractable chemotherapy-induced nausea and vomiting and cancer-related pain. J Support Oncol 4 (10): 531-5, 2006 Nov-Dec. [PUBMED Abstract]
- Guzmán M: Cannabinoids: potential anticancer agents. Nat Rev Cancer 3 (10): 745-55, 2003. [PUBMED Abstract]
- Yamaori S, Okamoto Y, Yamamoto I, et al.: Cannabidiol, a major phytocannabinoid, as a potent atypical inhibitor for CYP2D6. Drug Metab Dispos 39 (11): 2049-56, 2011. [PUBMED Abstract]
- Jiang R, Yamaori S, Okamoto Y, et al.: Cannabidiol is a potent inhibitor of the catalytic activity of cytochrome P450 2C19. Drug Metab Pharmacokinet 28 (4): 332-8, 2013. [PUBMED Abstract]
- Guedon M, Le Bozec A, Brugel M, et al.: Cannabidiol-drug interaction in cancer patients: A retrospective study in a real-life setting. Br J Clin Pharmacol 89 (7): 2322-2328, 2023. [PUBMED Abstract]
- Pletcher MJ, Vittinghoff E, Kalhan R, et al.: Association between marijuana exposure and pulmonary function over 20 years. JAMA 307 (2): 173-81, 2012. [PUBMED Abstract]
- Kocis PT, Vrana KE: Delta-9-tetrahydrocannabinol and cannabidiol drug-drug interactions. Med Cannabis Cannabinoids 3 (1): 61-73, 2020.
- Taha T, Meiri D, Talhamy S, et al.: Cannabis Impacts Tumor Response Rate to Nivolumab in Patients with Advanced Malignancies. Oncologist 24 (4): 549-554, 2019. [PUBMED Abstract]
- Bar-Sela G, Cohen I, Campisi-Pinto S, et al.: Cannabis Consumption Used by Cancer Patients during Immunotherapy Correlates with Poor Clinical Outcome. Cancers (Basel) 12 (9): , 2020. [PUBMED Abstract]
Summary of the Evidence for Cannabis and Cannabinoids
To assist readers in evaluating the results of human studies of integrative, alternative, and complementary therapies for people with cancer, the strength of the evidence (i.e., the levels of evidence) associated with each type of treatment is provided whenever possible. To qualify for a level of evidence analysis, a study must:
- Be published in a peer-reviewed scientific journal.
- Report on therapeutic outcome or outcomes, such as tumor response, improvement in survival, or measured improvement in quality of life.
- Describe clinical findings in sufficient detail for a meaningful evaluation to be made.
Separate levels of evidence scores are assigned to qualifying human studies on the basis of statistical strength of the study design and scientific strength of the treatment outcomes (i.e., end points) measured. The resulting two scores are then combined to produce an overall score. For an explanation of possible scores and additional information about levels of evidence analysis of Complementary and Alternative Medicine (CAM) treatments for people with cancer, see Levels of Evidence for Human Studies of Integrative, Alternative, and Complementary Therapies.
Cannabinoids
- Several controlled clinical trials have been performed, and meta-analyses of these support a beneficial effect of cannabinoids (dronabinol and nabilone) on chemotherapy-induced nausea and vomiting (N/V) compared with placebo. Both dronabinol and nabilone are approved by the U.S. Food and Drug Administration for the prevention or treatment of chemotherapy-induced N/V in patients with cancer but not for other symptom management.
Cannabis
- There have been ten clinical trials on the use of inhaled Cannabis in cancer patients that can be divided into two groups. In one group, four small studies assessed antiemetic activity, but each explored a different patient population and chemotherapy regimen. One study demonstrated no effect, the second study showed a positive effect versus placebo, and the report of the third study did not provide enough information to characterize the overall outcome as positive or neutral. Consequently, there are insufficient data to provide an overall level of evidence assessment for the use of Cannabis for chemotherapy-induced N/V. Apparently, there are no published controlled clinical trials on the use of inhaled Cannabis for other cancer-related or cancer treatment–related symptoms.
- An increasing number of trials are evaluating the oromucosal administration of Cannabis plant extract with fixed concentrations of cannabinoid components, with national drug regulatory agencies in Canada and in some European countries that issue approval for cancer pain.
- At present, there is insufficient evidence to recommend inhaling Cannabis as a treatment for cancer-related symptoms or cancer treatment–related symptoms or cancer treatment-related side effects; however, additional research is needed.
Latest Updates to This Summary (05/13/2025)
The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above.
Added text about the findings from a multicenter survey on perceptions, prevalence, and patterns of Cannabis use among 13,180 patients with various cancers treated at 12 National Cancer Institute-designated cancer centers (cited Ellison et al. as reference 4).
This summary is written and maintained by the PDQ Integrative, Alternative, and Complementary Therapies Editorial Board, which is editorially independent of NCI. The summary reflects an independent review of the literature and does not represent a policy statement of NCI or NIH. More information about summary policies and the role of the PDQ Editorial Boards in maintaining the PDQ summaries can be found on the About This PDQ Summary and PDQ® Cancer Information for Health Professionals pages.
About This PDQ Summary
Purpose of This Summary
This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about the use of Cannabis and cannabinoids in the treatment of people with cancer. It is intended as a resource to inform and assist clinicians in the care of their patients. It does not provide formal guidelines or recommendations for making health care decisions.
Reviewers and Updates
This summary is reviewed regularly and updated as necessary by the PDQ Integrative, Alternative, and Complementary Therapies Editorial Board, which is editorially independent of the National Cancer Institute (NCI). The summary reflects an independent review of the literature and does not represent a policy statement of NCI or the National Institutes of Health (NIH).
Board members review recently published articles each month to determine whether an article should:
- be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary.
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website's Email Us. Do not contact the individual Board Members with questions or comments about the summaries. Board members will not respond to individual inquiries.
Levels of Evidence
Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Integrative, Alternative, and Complementary Therapies Editorial Board uses a formal evidence ranking system in developing its level-of-evidence designations.
Permission to Use This Summary
PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary].”
The preferred citation for this PDQ summary is:
PDQ® Integrative, Alternative, and Complementary Therapies Editorial Board. PDQ Cannabis and Cannabinoids. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/about-cancer/treatment/cam/hp/cannabis-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389198]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in Visuals Online, a collection of over 2,000 scientific images.
Disclaimer
The information in these summaries should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s Email Us.