University of California, Davis — The Rodent Eye as a Non-Invasive Window for Understanding Cancer Nanotherapeutics
Principal Investigators: Kit Lam, M.D., Ph.D.
Co-Investigators: Edward Pugh, Ph.D., Edward Zawadzki, Ph.D., Yuanpei Li, Ph.D., Li Xing, Ph.D., and Wenwu Xiao, M.D., Ph.D.
Project Summary
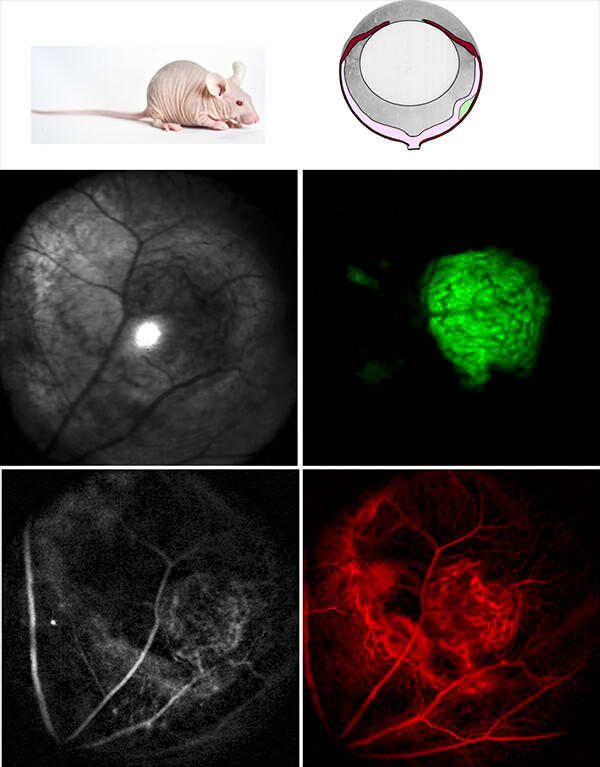
The overarching premise of our project is to use the mouse eye as a non-surgical window for highly efficient, optical investigation of all aspects of syngeneic or xenograft models, using a state-of-the-art ocular imaging facility, the "EyePod." The EyePod employs single-cell resolution intravital confocal microscopy and optical coherence tomography, performed completely non-invasively through the natural optics of the eye. This technology enables repeatable in vivo imaging over weeks and even months, quantitative tracking of tumor development and delivery of theranostic nanoparticles, and the measurement of tumor and tissue responses. Moreover, the retina is a highly "approachable part of the brain", so that non-invasive study of ocular tumors provides a platform for examining such critical issues as drug delivery across the blood brain barrier (BBB) and neurotoxicity. We will employ our recently reported nanoporphyrin in this research.
This novel multifunctional porphyrin-based micellar nanoplatform allows:
- efficient encapsulation of hydrophobic chemotherapeutic drugs or fluorescent dyes,
- near-infra red fluorescent (NIRF) detection of the tumor via the intrinsic fluorescence of porphyrins,
- photodynamic therapy (PDT) and photothermal therapy (PTT) via efficient free radical and heat generation at the tumor site, respectively,
- (Gd(III) loading for MRI imaging,
- 64Cu loading for PET imaging, and
- convenient ligation of cancer-targeting ligands to the surface of the micelle for cancer-specific targeted delivery.
Thorough understanding of how this nanocarrier distributes within the tumor microenvironment, and how it responds to controlled optical stimulation will enable us to maximize its therapeutic potential as a nano-theranostic agent. We will also study the effect of active targeting of nanocarriers with tumor and brain endothelial cell targeting peptides.
In more detail, our specific aims include;
- To develop intraocular glioblastoma and breast cancer xenograft models in eyes of nude mice, and to use non-invasive optical techniques (e.g., EyePod) to study the development of these tumor models at cellular resolution longitudinally over days and weeks, and their response to treatment with nanodoxorubicin;
- To use the tumor models and EyePod to study the biodistribution and photo-response of tumor targeting and non-targeting nanoporphyrins within the tumor micro-environment in vivo, and to use cryo-electron microscopy to "dissect" the nanodelivery at the ultrastructural level; and
- To optimize brain vascular endothelial cell-penetrating ligands, and to use in vivo EyePod imaging and cryoelectron microscopy to study their nanodelivery into retinal vasculature, and across the blood retinal barrier, which is very similar to blood brain barrier.
Project Expertise
For this effort, we have assembled a team of interdisciplinary scientists covering expertise across the areas of oncology, optics, imaging / microscopy, ophthalmology, nanomedicine, and cancer biology.