Northwestern University — Nucleic Acid-Based Nanoconstructs for the Treatment of Cancer
Center Overview
Current cancer treatment methodologies fall short of providing efficacious, targeted, precision therapies geared towards the individual patient. Due to their novel chemical, biological, and physical properties, nucleic-acid based nanoconstructs can be used to gain access to privileged intracellular environments, discover new aspects of cancer biology, and exploit nanostructure-biomolecular interactions to create effective treatment options. The Northwestern CCNE will explore these vast possibilities by applying a novel class of nanostructure genetic constructs - the spherical nucleic acid (SNA) and variants of it - for the study and treatment of brain and prostate cancer.
SNAs enter cells in large quantities as single-entity agents and have been shown to cross the blood-brain-barrier (BBB) and the blood-tumor-barrier (BTB) in a systemic delivery scheme, and the epidermis in a topical approach. Once inside the cellular environment, SNAs resist enzymatic degradation, do not elicit toxicity or immunogenicity, and can be used to regulate gene expression via antisense and RNAi pathways. SNAs form the foundation of the technological focus of the Northwestern CCNE which, once successful, will generate a large body of fundamental knowledge that will stand to unravel a portion of the complex landscape of cancer genetics and biology, and deliver two first-in-class SNA nanostructure-based agents - a therapeutic for precision neuro-oncology that can be used to treat glioblastoma multiforme and a therapeutic cancer vaccine that can be used to treat prostate cancer.
The Northwestern CCNE is led by International Institute for Nanotechnology Director, Chad A. Mirkin, PhD, and Robert H. Lurie Comprehensive Cancer Center Director, Leonidas Platanias, MD. Dr. Mirkin and Dr. Platanias bring extensive knowledge, skill, and experience to the enterprise, and together, share the authority and responsibility for leading and directing the project.
Projects
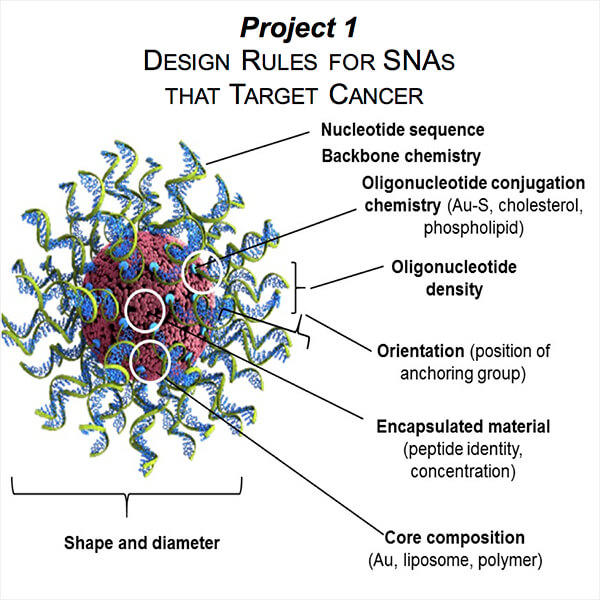
Project 1: Design Rules for SNAs that Target Cancer
Project Investigator: Milan Mrksich, Ph.D.
Led by Professor of Chemistry Milan Mrksich, this project will develop the design rules for the synthesis of SNAs with maximum / optimal gene regulatory or immunostimulatory capacity. Major goals include the analysis of the bulk responses from thousands of cells and the use of NanoFlare technology and microfluidic systems to probe and analyze the individual responses of single cells. The data from these measurements will be used to develop structure-activity rules that guide the optimization of nanoparticle therapeutics for Projects 2 and 3. The work will identify those structural features that promote (as well as detract from) the wanted pharmacological effects.
Project 2: SNAs for Metabolic Reprogramming of Malignant Glioma
Project Investigator: Alexander Stegh, Ph.D.
Project 2 is led by Alexander Stegh, an Assistant Professor and Zell Scholar in the Department of Neurology. The project will preferentially inhibit, in GBM cells, one of the most fundamentally important energy generating pathways, the tricarboxylic acid (TCA) cycle, and, in so doing, trigger tumor cell killing induced by inhibitors of receptor tyrosine kinases (RTKi). Major goals of this project include functionally evaluating a library of IDH1-targeting SNAs to identify high-activity constructs, evaluating siIDH1-SNAs as adjuvant for RTKi in GBM subtype-defined PDX models, and determining anti-glioma effects of siIDH1-SNAs and RTKIs in a genetically engineered mouse (GEM) model for GBM. This project will comprehensively characterize IDH1 as a novel therapeutic target in GBM, and provide preclinical proof-of-concept that the systemic transfer of siRNAs targeted to IDH1 as adjuvant for RTKi is an effective new strategy to combat malignant brain cancers.
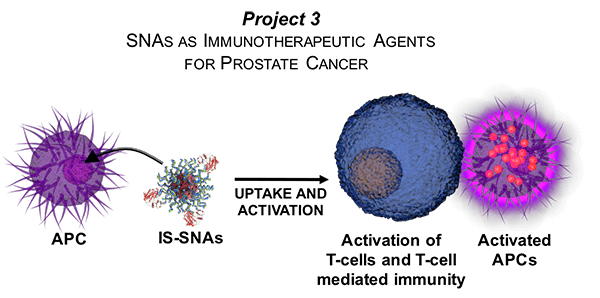
Project 3: SNAs as Immunotherapeutic Agents for Prostate Cancer
Project Investigator: Chad A. Mirkin, Ph.D.
This is a translational project led by Center PI Chad Mirkin. This project will develop a new class of therapeutic vaccines based on SNAs for the treatment of prostate cancer. SNAs represent a therapeutic platform for the rational design of nanostructures that induce potent immunostimulatory responses directed towards prostate tumor antigens. Major goals of this project include the design and synthesis of immunostimulatory SNAs (IS-SNA) for lymphocyte activation, analysis of the immunostimulatory activity of IS-SNAs with a panel of standardized in vitro assays, assessment and characterization of IS-SNA activity in immunocompetent mouse models, and development of combination therapies for an optimized cancer immunotherapy: immunostimulation by IS-SNAs combined with modulation of the immunosuppression of solid tumors. The IS-SNA platform is expected to show excellent immunostimulatory properties in the treatment of prostate cancer. Furthermore, the combinations of IS-SNAs and tumor microenvironment modulators are expected to show synergy, and lead to improved therapeutic outcomes over those of either agent alone. These results will inform the design of early stage clinical studies that involve the combination of these agents to be conducted by the commercialization partner.
Cores
Oligonucleotide Synthesis and Nanoconstructs Core
Core Investigator: Teri W. Odom, Ph.D.
This core will provide large quantities of optimized, high-quality SNAs to the projects for preclinical translational research. The core will also seek to better understand how the architecture of the SNA affects its behavior in biological environments.
Developmental Program
Center Program Director: Chad Mirkin, Ph.D.
The Northwestern CCNE will include a developmental program designed to broaden capabilities by providing support for junior faculty to engage in pilot projects in emerging high risk/high reward areas and for trans-Alliance research and training projects as well as projects to cross-test novel frameworks.