Detection and Diagnosis
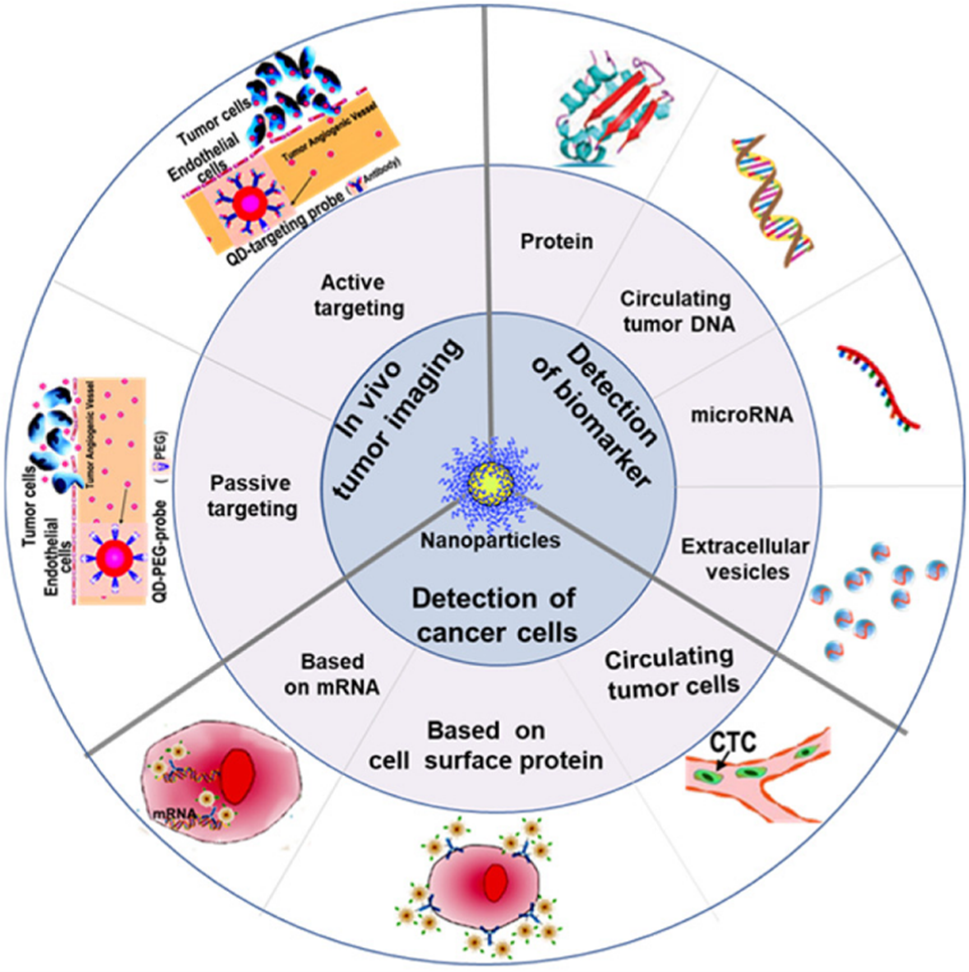
In the fight against cancer, half of the battle is won based on its early diagnosis. Cancer identified in its early stages leads to a significantly higher 5-year survival rate and a typically less aggressive treatment course. Given the limitations of classic imaging techniques and morphologic analysis of tissues or cells in achieving early diagnosis of cancer, nanotechnology has the potential of providing new molecular contrast agents and in vitro assays to enable earlier and more accurate initial diagnosis as well as ongoing monitoring of cancer patient treatments, as illustrated in the diagram below.
Nano-enable biosensors (as exemplified in the right image) exhibit exceptional sensitivity, specificity, and the capacity to recognize multiple molecules, making them suitable for detecting extracellular cancer biomarkers like cancer-associated proteins, circulating tumor DNA, and extracellular vesicles. Nanomaterials also come into play for the sensitive detection of cancer cells. For example, the isolation and characterization of circulating tumor cells (CTCs) presents challenges due to their relatively low abundance; nevertheless, diverse nanomaterials, including magnetic nanoparticles, nanowires, and dendrimers, are extensively developed and studied for precise CTCs detection. Additionally, nanoparticle probes accumulate within tumor tissues and can serve as contrast agents for in vivo cancer imaging, enhancing anatomical structure visualization. Lastly, these nanotechnology-based approaches also play a crucial role in measuring therapy response, optimizing treatment strategies.
Sensing In Vitro
The discovery of cancer-associated molecular alterations in blood at multiple molecular spaces (genome, epigenome, transcriptome, proteome, and metabolome) has led to new opportunities in early cancer detection. Nanotechnology-enabled liquid biopsy diagnostic devices offer the capability for the enrichment and subsequent omics analysis of the blood-circulating ‘cancer fingerprints’ with high selectivity and sensitivity. Through well-established fabrication techniques (e.g., lithography), compact devices or point-of-care systems can be crafted. These diagnostic tools utilize biochemical reactions to interrogate the presence, activity, or concentration of specific biological molecules in a bodily fluid sample. These reactions, such as antigen-antibody binding, hybridization of DNA fragments, or ligand adhering to cell surface epitopes, can subsequently be transformed into measurable detection signals using methods like optical, magnetic, or electronic means.
Many cancers detection and diagnostic devices utilizing nanoparticles have been developed (as evident in the table below). A pioneering study (Jwa-min Nam, Shad Thaxton, and Chad A. Mirkin, Science, 2003) introduced an ultrasensitive method for detecting prostate-specific antigen (PSA) down to 3 attomolar sensitivity level, which is six orders of magnitude more sensitive as compared to the clinically accepted conventional assays. This approach, known as the bio-barcode assay, employs a sandwich immunoassay design by combining magnetic nanoparticles with PSA-specific antibodies and DNA-encoded gold nanoparticles. The elevated PSA level can be used to trigger prostate-specific membrane antigen (PSMA) targeted PET imaging (Steven P. Rowe, Michael A. Gorin, and Martin G. Pomper, Annual Review of Medicine, 2019) for prostate cancer detection and therapy monitoring. Another sandwich immunoassay device has been designed, relying on DNA-encoded antibody libraries (DEAL, as shown in the right figure). DEAL technique (Rong Fan, et al., Nature Biotechnology, 2008; Chao Ma, et al., Nature Methods, 2011) uses DNA-directed immobilization of antibodies in microfluidic channels that can convert a pre-patterned single stranded DNA barcode microarray into an antibody microarray. This enables multiplex immunoassays, on-chip blood separation, and measurement of large protein panels directly from blood. Looking forward, new advances in the coupling of complex microfluidics and nanoscale materials offer the potential to derive comprehensive in vitro cancer diagnostic insights from a patient sample.
Examples of nanomaterial detection platforms used in cancer liquid biopsy studies. EpCAM, epithelial cell adhesion molecule. Adapted from Lois Gardner, Kostas Kostarelos, Parag Mallick, Caroline Dive, and Marilena Hadjidemetriou, Nature Reviews Clinical Oncology, 2022
| Nanomaterial platform | Enrichment mechanism | Biofluid (volume) | Type of cancer | Reference |
|---|---|---|---|---|
| Proteins | ||||
| Nanotrap hydrogel core–shell nanoparticles | Affinity capture | Serum (0.2 ml) | Ovarian or prostate cancer | Fredolini, C. et al. AAPS J. 2010 |
| PEGylated liposomal doxorubicin | Protein corona | Plasma (1 ml) | Ovarian cancer | Hadjidemetriou, M. et al. Nano Today 2020 |
| Proteograph superparamagnetic iron oxide nanoparticles | Protein corona | Plasma (0.1 ml) | Non-small-cell lung cancer | Blume, J. E. et al. Nat. Commun. 2020 |
| Circulating cell- free DNA (cfDNA) | ||||
| GMACS chip (silica magnetic nanoparticles) | Label free | Serum (0.5 ml) | Breast cancer | Gwak, H. et al. Biomicrofluidics 2019 |
| Polypyrrole-coated gold nanowires | Label free | Plasma (0.2-1 ml) | Breast or lung cancer | Jeon, S. H. et al. Theranostics 2016 |
| Circulating tumor cells (CTCs) | ||||
| Magnetic nanowires | Immunocapture (anti-pCAM, anti-EGFR, anti-N-cadherin, anti-TROP2 and anti-vimentin) | Blood (0.2-1 ml) | Non-metastatic, early stage breast cancer | Hong, W. et al. Biomaterials 2016 |
| TiO2 nanofibers | Immunocapture (anti-EpCAM) | Blood (1 ml) | Colorectal or gastric cancer | Zhang, N. et al. Adv. Mater.2012 |
| PL–PEG–NH2- functionalized graphene oxide nanosheets | Immunocapture (anti-EpCAM) | Blood (1 ml) | Lung, breast, or pancreatic cancer | Yoon, H. J. et al. Nat. Nanotechnol. 2013 |
| Carbon nanotube chip | Label free | Blood (4-8.5 ml) | Breast cancer | Loeian, M. S. et al. Lab. Chip 2019 |
| Extracellular vesicles (EVs) | ||||
| Nano-interfaced microfluidic exosome chip (graphene oxide–polydopamine coated) | Immunocapture (anti-CD81) | Plasma (2 μl) | Ovarian cancer | Zhang, P. et al. Lab. Chip 2016 |
| Silicon nanowires | Immunocapture (anti-EpCAM, anti-ASGPR1 and anti-CD147) | Plasma (0.5 ml) | Hepatocellular carcinoma | Sun, N. et al. Nat. Commun.2020 |
Imaging In Vivo
Current imaging methods, such as MRI, PET, and CT, can detect cancers with sensitivity levels sufficient only to provide detection by the time thousands of cancer cells have grown and may have spread. Nanotechnology based imaging contrast agents for conventional imaging modalities, now in development and translation, offer the ability to specifically target and significantly amplify the ability of in vivo tumor detection. Moreover, current nanoscale imaging platforms are enabling novel imaging modalities not traditionally utilized for clinical cancer treatment and diagnosis, for example photoacoustic tomography (PAT), Raman spectroscopic imaging, and multimodal imaging (i.e., contrast agents designed for several imaging modalities, simultaneously). Nanotechnology enables these imaging platforms by designing nanoparticles simultaneously carrying multiple components (e.g., conventional contrast agents) and ligands to target tumor cells.
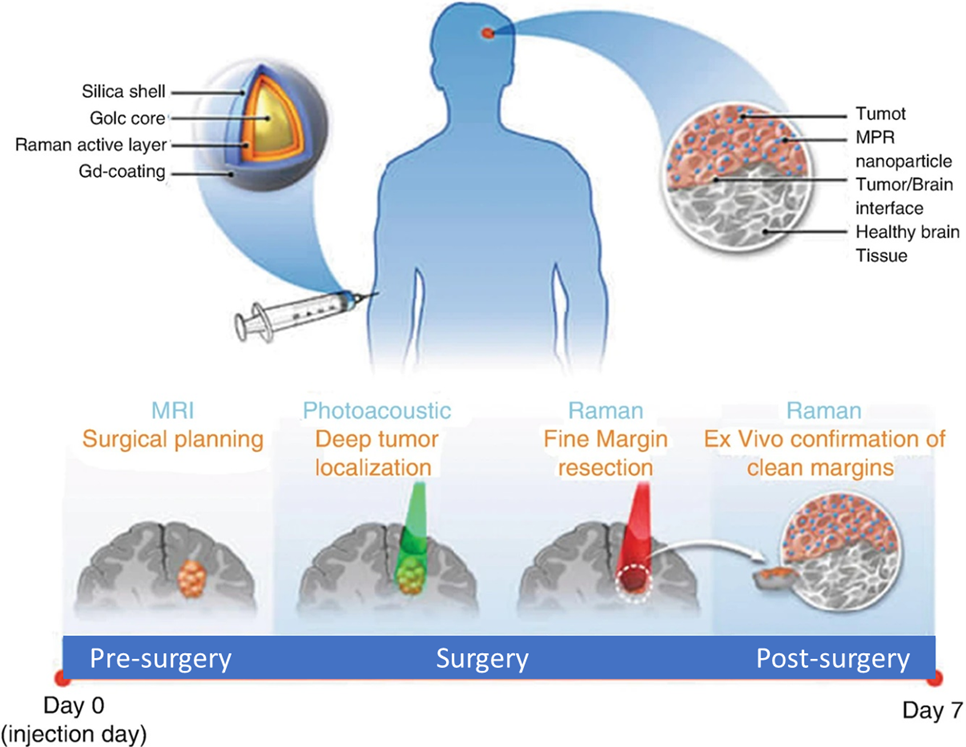
NCI-funded research has produced many notable examples of nanotechnology-based imaging agents over the past decade. For instance, researchers have engineered multimodal nanoparticles that can delineate the margins of brain tumors before and during surgery (Moritz F Kircher, et al., Nature Methods, 2012). These nanoparticles, combining MRI, PAT, and Raman detection, are capable of tracking tumor growth and surgical staging via MRI, but also grant the surgeon the ability to visualize brain tumor down to the single cancer cell level, increasing the potential for precise removal of cancerous tissue (as shown in the figure below). Similarly, scientists have designed silica-hybrid nanoparticles (‘C-dots’) capable of providing both PET and optical imaging contrast in the same platform (Evan Philips, et al., Science Trans Med., 2016). These nanoparticles can actively target to metastatic melanoma with cRGDY peptides, making it successfully through initial clinical trials.
A longstanding vision in cancer nanotechnology is the capability to simultaneously image and administer therapy in vivo, leading to the emergence of ‘theranostic’ nanoscale platforms. A diagnostic platform for pancreatic cancer has been designed to break through the fibrotic stromal tissue of these tumors (Lei Zhu, Hui Mao, and Lily Yang, WIREs Nanomedicine and Nanobiotechnology, 2022). Once past this barrier, the nanoparticles, containing magnetic iron cores, facilitate MRI contrast for diagnosis and concurrently deliver small-molecule drugs directly to cancer cells for treatment.
Measuring Response to Therapy
The cornerstone of precise and prognostic medical care lies in measuring an individual patient’s response to targeted therapy, immunotherapy, or surgery as their disease progresses. Accurate and relevant disease monitoring can allow for optimizing treatment regimens (e.g., therapeutic course correction, use of drug combinations, and fine-tuning drug doses), preemptive clinical decision making (e.g., separating therapeutic responders from non-responders), and patient stratification for clinical trials. In vivo imaging, tissue biopsy, and in vitro diagnostics are all available for this purpose. Being able to track cancer cell death in vivo is important for delivering effective dosing regimens and/or precisely administering novel therapies. For example, scientist have designed a target-enabled in situ ligand assembly (TESLA) nanoparticle system, in which nanoparticles can self-assemble within the body when cleaved by enzymes from therapy-induced cell death (Deju Ye, et al., Nature Chemistry, 2014). These nanoparticles also carry various image contrast agents to monitor (PET, MRI, etc.) local tumor response to therapies.
While tissue biopsy serves as the traditional gold standards for cancer identification, it is an invasive procedure and has limited space sampling capability to track disease progression relative to the course of therapy, effectively. In vitro cancer diagnosis and monitoring introduces a simpler method of tracking therapy response by leveraging the tumor’s shed biological material (e.g., cells, DNA, other cancer-specific biomolecules) circulating in blood over time. They typically exist at incredibly low concentrations as compared to other blood components, the need arises for specific and highly sensitive tools to detect, capture, and purify them. Nanotechnology is paving the way for the realization of these essential tools. For instance, microfluidic nano chips (one example shown in the right figure) have been developed for multiplex protein detection of potential cancer markers at sub-picomolar concentration level (Sebastian J. Osterfeld, et, al., PNAS, 2008) and for CTCs detection with over 95% sensitivity (Millicent Lin, et al., Accounts of Chemical Research, 2014), enabling effective monitoring of cells and proteins in blood samples to understand and enhance therapy outcomes.