Insights into a signaling process that enhances the effectiveness of cancer immunotherapy
- Posted:
240-760-6600
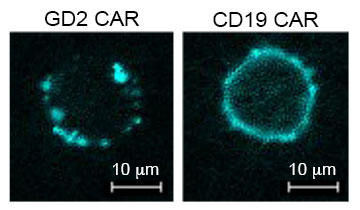
Fluorescent imaging of T cells with CAR receptors on the surface. The GD2 CAR (left) spontaneously aggregates, resulting in antigen-independent signaling, T cell exhaustion, and poor efficacy. The highly effective CD19 CAR (right) does not aggregate and does not signal in the absence of antigen.
Researchers at NCI have found that costimulatory signals can regulate T cell exhaustion, a process that limits the effectiveness of cancer immunotherapies. T cells are white blood cells that help fight infection and cancer. In this study, the investigators engineered T cells to produce artificial receptors called chimeric antigen receptors (CARs) on their surface, a promising approach to cancer treatment. CARs contain antigen recognition regions that allow the CAR T cells to recognize specific molecules (antigens) on tumor cells. CARs also contain intracellular stimulatory and costimulatory domains that provide signals necessary for full activation of the T cells. T cells engineered to express these CARs can then target and destroy tumor cells. However, during prolonged exposure to infection or tumor cells, T cells can lose their ability to function, a process known as T cell exhaustion. The NCI researchers found that some CAR T cells undergo early T cell exhaustion, a functional state that limits their effectiveness in fighting cancer. The researchers also found that changing the types of costimulatory signals that are included in the CAR can improve the function of CAR T cells by preventing exhaustion. This effort was led by Crystal Mackall, M.D., chief of the Pediatric Oncology Branch, Center for Cancer Research, NCI, and Adrienne Long, an M.D.-Ph.D. student in the laboratory. Their findings appeared May 4, 2015, in Nature Medicine.
While developing CAR T cells targeting an antigen called GD2, the scientists observed that some CARs cause continuous activation of the T cells that express them, independent of the presence of antigen. This antigen-independent signaling triggered CAR T cell exhaustion, which limited the cells’ ability to kill cancer in mice. The antigen-independent CAR signaling and CAR T cell exhaustion was seen to varying degrees with all CARs they tested, with the exception of the CD19 CAR, which has demonstrated effectiveness against acute lymphoblastic leukemia in early-phase trials. These findings indicate that exhaustion is a critical factor limiting the efficacy of some CAR T cells and provides a biologic basis to explain the dramatic anti-tumor effects of CD19 CARs. But the effect is less impressive, thus far, using CARs targeting other antigens. The researchers then used the GD2 CAR as a model of chronic CAR T cell signaling and exhaustion to study how changing the costimulatory molecule in the CAR affects CAR T cell exhaustion. They found that the costimulatory signals CD28 and 4-1BB had opposing effects on CAR T cell exhaustion, with CD28 increasing T cell exhaustion and 4-1BB decreasing it. These findings suggest that incorporating the 4-1BB costimulatory signal into CARs may be important in developing CAR T cells for clinical application. Experiments are underway to further explain the mechanism by which 4-1BB signaling prevents exhaustion in T cells, according to Mackall.