NCI study shows feasibility of cancer screening protocol for patients with Li-Fraumeni syndrome
- Posted:
240-760-6600
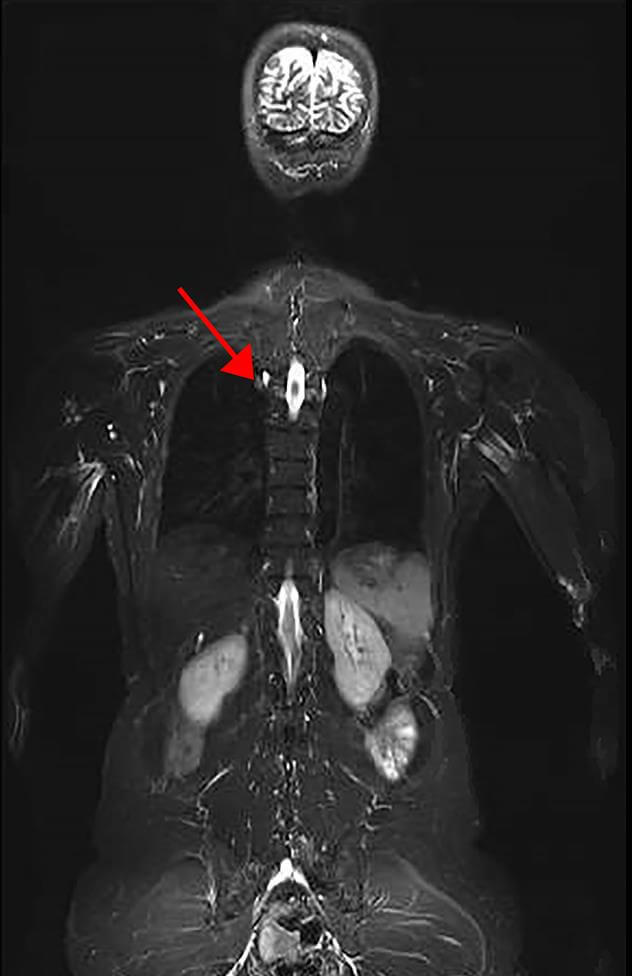
Part of a representative image of a whole body MRI of an LFS patient. Arrow denotes lesion found to be lung adenocarcinoma.
Credit: NCI
In a new study from the National Cancer Institute (NCI), part of the National Institutes of Health, researchers found a higher than expected prevalence of cancer at baseline screening in individuals with Li-Fraumeni syndrome (LFS), a rare inherited disorder that leads to a higher risk of developing certain cancers. The research demonstrates the feasibility of a new, comprehensive cancer screening protocol for this high-risk population.
The study was led by Sharon A. Savage, M.D., of NCI’s Division of Cancer Epidemiology and Genetics (DCEG), and was published with a companion meta-analysis on August 3, 2017, in JAMA Oncology.
LFS is most often caused by germline, or hereditary, mutations in a tumor suppressor gene known as TP53. The disorder results in many kinds of cancers—including bone and soft-tissue cancers (sarcomas), breast cancer, brain tumors, and cancer of the adrenal gland—that frequently occur at young ages. Individuals with LFS have an approximately 50 percent chance of developing cancer by age 40, and up to a 90 percent chance by age 60. Many patients with LFS develop more than one primary cancer over their lifetimes.
Dr. Frederick Pei Li and Dr. Joseph F. Fraumeni, Jr., first described LFS at NCI in 1969. “Researchers at the NCI have evaluated families with LFS extensively to better understand how germline mutations in TP53 influence risk, and how best to prevent cancers or treat them at the earliest possible stage,” said Dr. Fraumeni, Scientist Emeritus and Founding Director of DCEG. “However, because of the broad spectrum of cancers in LFS families, it has been challenging to put in place universally accepted cancer strategies.”
To address this gap in clinical care, researchers modified a cancer surveillance protocol from a previously published study and screened 116 LFS patients with germline TP53 mutations using a variety of tools including whole body, brain, and breast MRI, as well as mammography, colonoscopy, bloodwork, and abdominal ultrasound. The study used modalities that do not utilize ionizing radiation for imaging (except mammography), since patients with LFS appear to be radiosensitive.
They found that 40 trial participants (34 percent) had abnormalities on baseline screening examination with whole body, brain, or breast MRI that required further evaluation. Eight of these patients (7 percent) were diagnosed with a new primary cancer. All but one of the cancers found through screening were fully removed with surgery. In contrast, the non-MRI techniques used in the trial did not lead to a diagnosis of cancer at baseline screening.
“For high-risk populations, like families with LFS, personalized prevention approaches like this are critical to the early detection of the many kinds of cancers seen in this group,” Dr. Savage explained. “This protocol, along with other published studies, offers patients with LFS a new road map for early cancer detection going forward.”
The meta-analysis, published in the same issue of the journal, involved 578 participants with LFS in 13 cohorts at multiple research centers around the world. Similarly utilizing rapid whole body MRI, the investigators had an overall detection rate of 7 percent for new primary cancers, confirming the results of the study conducted at NCI.
“The findings from this international team effort further demonstrate the utility of whole body MRI screening for individuals with LFS,” Dr. Savage said. “With long term follow-up, additional refinement, and through international collaborations, we hope to establish a screening regimen that could extend and improve the lives of this unique population.”
About the National Cancer Institute (NCI): NCI leads the National Cancer Program and NIH’s efforts to dramatically reduce the prevalence of cancer and improve the lives of cancer patients and their families, through research into prevention and cancer biology, the development of new interventions, and the training and mentoring of new researchers. For more information about cancer, please visit the NCI website at cancer.gov or call NCI’s Contact Center (formerly known as the Cancer Information Service) at 1-800-4-CANCER (1-800-422-6237).
About the National Institutes of Health (NIH): NIH, the nation's medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit nih.gov.