AI dual-stain approach improved accuracy, efficiency of cervical cancer screening in NCI study
- Posted:
240-760-6600
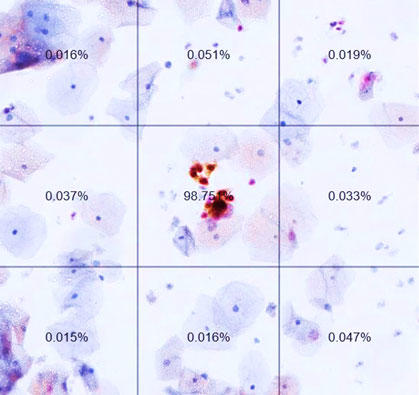
A slide from an automated dual-stain cytology test. The percentages are AI-generated likelihoods of positive results. The image at center (labeled 98.75%) shows a positive result.
Credit: Nicolas Wentzensen, M.D., Ph.D.
In a new study, a computer algorithm improved the accuracy and efficiency of cervical cancer screening compared with cytology (Pap test), the current standard for follow-up of women who test positive with primary human papillomavirus (HPV) screening. The new approach uses artificial intelligence (AI) to automate dual-stain evaluation and has clear implications for clinical care.
Findings from the study were published June 25, 2020, in the Journal of the National Cancer Institute. The algorithm was developed and the study conducted by investigators at the National Cancer Institute (NCI), part of the National Institutes of Health, in collaboration with researchers from several other institutions.
“We’re excited to show we have a fully automated approach to cervical cancer screening as a follow-up to a positive HPV test that outperformed the standard method in our study,” said Nicolas Wentzensen, M.D., Ph.D., of NCI’s Division of Cancer Epidemiology and Genetics, who led the study. “Based on our results, it could increase the efficiency of cervical cancer screening by finding more precancers and reducing false positives, which has the potential to eliminate a substantial number of unnecessary procedures among HPV-positive women.”
In recent years, clinicians have hoped to take advantage of advances in digital imaging and machine learning to improve cervical cancer screening. Women who test negative for HPV are at low risk for cervical cancer for the following decade, and even most cervical HPV infections—which cause positive HPV tests—will not result in precancer. The challenge is to identify which women with positive HPV test results are most likely to have precancerous changes in their cervical cells and should, therefore, have a colposcopy to examine the cervix and take samples for biopsy, or who need immediate treatment.
Currently, women with positive HPV tests may have additional HPV tests or Pap cytology tests to assess the need for colposcopy, biopsy, or treatment. Pap cytology, in which specially trained laboratory professionals (cytotechnologists) analyze stained slides to look for abnormal cells, is used to find precancers before they progress to cancer. But these approaches are not ideal. For example, Pap cytology tests are time consuming, not very sensitive, and prone to false-positive findings.
Dual-stain testing has emerged as a way to more accurately predict the chance that a woman with a positive HPV test has precancerous cervical changes. The test measures the presence of two proteins, p16 and Ki-67, in cervical samples. In two previous studies, Dr. Wentzensen and his colleagues found that women who had a negative result on a dual-stain test had a low risk of developing cervical precancer in the following five years and that fewer women test positive for dual-stain compared to Pap cytology. In March 2020, the manual dual-stain cytology test was approved by the U.S. Food and Drug Administration (FDA) for women who have received a positive result on a primary HPV screening.
The manual dual-stain test has a subjective component, in that a cytotechnologist must look at the slide to determine the results. In the new study, the investigators wanted to see if a fully automated dual-stain test could match or exceed the performance of the manual approach. In collaboration with Niels Grabe, Ph.D., and Bernd Lahrmann, Ph.D., of the Steinbeis Transfer Center for Medical Systems Biology, which is associated with the University of Heidelberg, they developed a whole-slide imaging platform that, after being trained with deep learning, could determine if any cervical cells were stained for both p16 and Ki-67. They compared this method with both conventional Pap cytology and manual dual-stain testing in samples from a total of 4,253 people participating in one of three epidemiological studies of HPV-positive cervical and anal precancers at Kaiser Permanente Northern California and the University of Oklahoma.
The researchers found that the AI-based dual-stain test had a lower rate of positive tests than both Pap cytology and manual dual-stain, with better sensitivity (the ability to correctly identify precancers) and substantially higher specificity (the ability to correctly identify those without precancers) than Pap cytology. AI-based dual-stain reduced referral to colposcopy by about a third compared with Pap (approximately 42% vs. 60%). The testing method was also robust, showing comparable performance in anal cytology.
In short, the automated test surpassed the performance of the current standard, Pap cytology, reducing the number of false positive results and substantially reducing referral to unnecessary colposcopy procedures. The results also support further evaluation of the test as an option for anal cancer screening. The researchers note that their approach has clear clinical application, and through cloud-based implementation, it would be globally accessible. Other applications of the platform include assisted evaluation, second opinion, and quality control.
Because the manual dual-stain test has only recently received FDA approval for screening of women who have HPV-positive test results, its use is just getting started. Additional regulatory approval will be needed to allow for screening of HPV-positive women with a fully automated dual-stain test. The researchers say that their findings serve as an important example for introducing digital pathology and deep learning into clinical practice, and their approach has the potential to substantially improve cervical cancer screening, affecting millions of women testing HPV-positive each year.
About the National Cancer Institute (NCI): NCI leads the National Cancer Program and NIH’s efforts to dramatically reduce the prevalence of cancer and improve the lives of cancer patients and their families, through research into prevention and cancer biology, the development of new interventions, and the training and mentoring of new researchers. For more information about cancer, please visit the NCI website at cancer.gov or call NCI’s contact center, the Cancer Information Service, at 1-800-4-CANCER (1-800-422-6237).
About the National Institutes of Health (NIH): NIH, the nation's medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit nih.gov.