Could CryoEM structures of neurofibromin lead the way to better therapeutic approaches for Neurofibromatosis type 1?
, by Dom Esposito
Dom Esposito has directed the Protein Expression Laboratory of the NCI’s Frederick National Lab since 2011, and currently leads the RAS Reagents Core, which provides DNA cloning, protein expression and purification, and qualified cell lines to the NCI RAS Initiative. He earned his Ph.D. in Biochemistry and Biophysics at Johns Hopkins University and did postdoctoral work in the Laboratory of Molecular Biology at the National Institute of Diabetes and Digestive and Kidney Diseases.
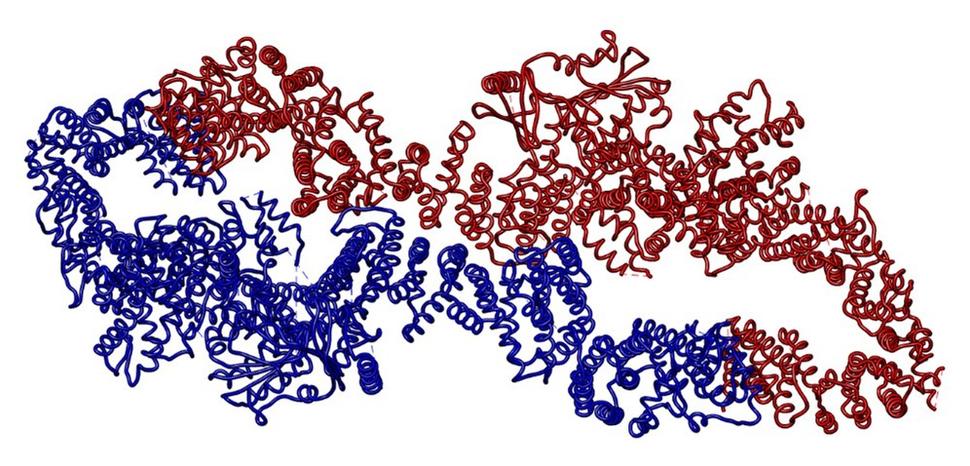
Recently, three groups (https://pubmed.ncbi.nlm.nih.gov/35353986/, https://pubmed.ncbi.nlm.nih.gov/34887559/, https://pubmed.ncbi.nlm.nih.gov/34707296/) reported detailed molecular structures of the neurofibromin protein responsible for the genetic disorder Neurofibromatosis type 1 (NF1). This disease impacts 1 in 2,000 adults and leads to neurological issues as well as an increased risk for benign and malignant nerve and skin tumors. The neurofibromin protein is a GTPase activating protein (GAP) responsible for turning off RAS activity, and defects in this large 2818 amino acid protein lead to uncontrolled signaling through the RAS pathway. It has been unclear how so many individual mutations throughout the sequence of this protein can lead to disease, but hints emerged from a 2019 study from the NCI RAS Initiative (https://pubmed.ncbi.nlm.nih.gov/31836666/) demonstrating that Neurofibromin is found exclusively in a dimeric state and seemed to be highly sensitive to mutations that perturb its structure. The recent structures offer a clearer explanation for the impact of mutations by highlighting a complex set of helical repeats throughout the protein. Mutations that disrupt these helical regions throughout the core of the protein seem likely to impact the overall protein structure and the positioning of the GAP domain, and may lead to overall protein destabilization or altered activity. In addition, the structures reveal that the protein exists in two states—an “autoinhibited” state which cannot bind to RAS, and an “active” state in which RAS binding can occur. How these states transition and the impact of mutations on that transition event remain to be explored, but the structures provide a framework for additional experiments which could lead to the development of new therapeutic approaches.
NF1 is a debilitating disease, and even in cases where malignant tumors are not present, the impact of benign cutaneous neurofibromas (cNF) can lead to severe pain, disfigurement, and emotional and social distress. The limited therapeutic options for NF1 patients have in part been due to a poor understanding of the disease’s basic biology and the role of mutations in neurofibromin activity. A number of groups have been working to improve research into these areas, including the Neurofibromatosis Therapeutic Acceleration Program (NTAP) at the Johns Hopkins University. NTAP provides grant funding for academic, government, and private sector researchers investigating preclinical and clinical aspects of NF1, and has recently requested applications for funding for new research into biology and therapeutic development for cNFs in NF1 disease (learn more about the opportunity HERE. Letters of intent are due August by 5th, and full applications are due by September 2nd). The new structural data will hopefully enable researchers to identify novel therapeutic approaches for these patients.