What is SELECT?
SELECT stands for the Selenium and Vitamin E Cancer Prevention Trial. SELECT was a clinical trial to see if one or both of these substances could help prevent prostate cancer when taken as dietary supplements. The trial was funded primarily by NCI and developed and carried out by SWOG, an international network of research institutions. Enrollment for the trial began in 2001 and ended in 2004. More than 400 sites in the United States, Puerto Rico, and Canada took part in the study. Over 35,000 men, age 50 and older at the start of the trial, participated in SELECT (1).
What were the initial results of SELECT as published in 2008?
SELECT was initially planned for a minimum of seven years and a maximum of 12 years of participants taking supplements, plus follow-up observation after the men finished taking their supplements. However, the independent Data and Safety Monitoring Committee (DSMC) for the trial met on September 15, 2008, to review SELECT study data and found that selenium and vitamin E, taken alone or together did not prevent prostate cancer. Based on their recommendation, with SWOG and NCI agreement, SELECT participants were told in October 2008 to stop taking their study supplements..
Although there were no statistically significant differences in the rates of prostate cancer between the four groups in the trial, there were more cases of prostate cancer in men taking only vitamin E. Statistical significance describes a mathematical measure of how sure one can be that a difference seen is not due to chance.
In the 2008 report, there were also more new cases of diabetes in men taking only selenium compared with men taking placebo. This finding was also not statistically significant and did not prove an increased risk from selenium. Initial results of SELECT were published online in JAMA-Express on December 9, 2008, and appeared in the first print edition of JAMA in January 2009 (2). There was no difference in the incidence of lung or colorectal cancers, all cancers combined, all deaths combined, or the overall incidence of cardiovascular events between the study groups.
What were the updated results of SELECT published in 2011?
The data published in 2011 include 18 months of additional follow-up information on the participants through July 5, 2011. During this time, SELECT men were no longer taking study supplements. These additional data provide an average of seven years of information on the participants: 5.5 years taking study supplements plus 1.5 years of observation or follow-up.
The additional data show that the men who took vitamin E alone had a 17 percent relative increase in numbers of prostate cancers compared to men on placebo. This difference in prostate cancer incidence between the vitamin E only group and the placebos only group is now statistically significant, and not likely to be due to chance.
Men taking selenium alone, or vitamin E and selenium, were also more likely to develop prostate cancer than men taking placebo, but those increases were smaller and are not statistically significant and may be due to chance. Updated results of SELECT were published in JAMA on October 12, 2011 (3).
Do we know why an increased risk for prostate cancer was found in 2011 vs. 2008?
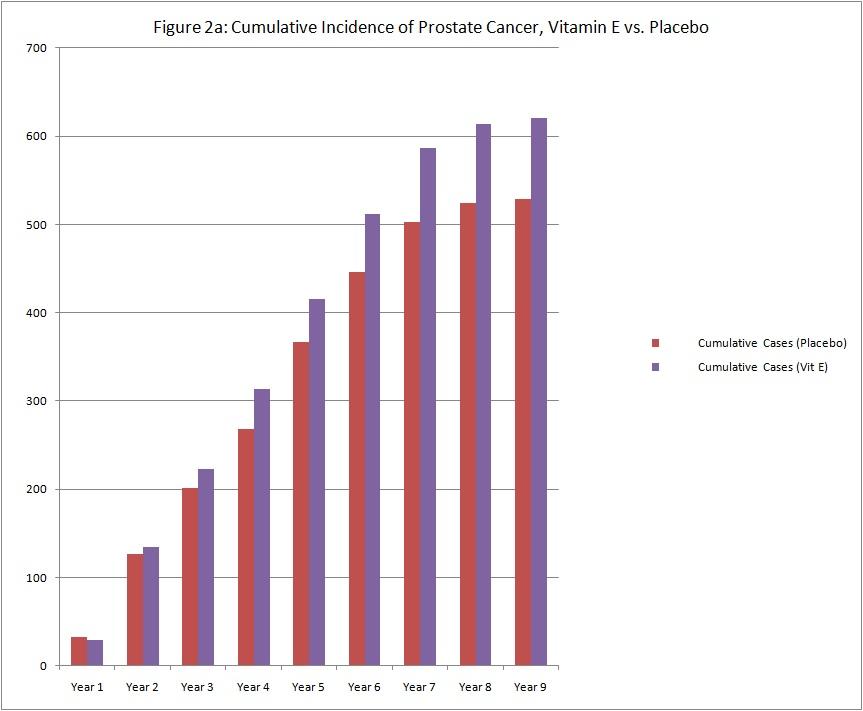
As with many medications, the actions of vitamin E may last long after the last pill is taken. The differences in prostate cancer incidence between the vitamin E group and the placebo group began to emerge at about the third year of study supplementation (see graph below of cumulative cases).
The observation that the risk of prostate cancer has continued to increase suggests that vitamin E may have long-term effects on prostate cancer risk.
There continued to be no differences in the incidence of lung or colorectal cancers, all cancers combined, all deaths combined, or the overall incidence of cardiovascular events between the study groups. Although initial results showed that more men taking selenium were diagnosed with diabetes, longer follow-up showed no increased risk of this disease.
What does a 17 percent increased risk of prostate cancer mean?
In SELECT, this measure means that there were 17 percent more prostate cancer cases diagnosed in the group of men assigned to take 400 International Units (IU) of vitamin E (and no selenium) daily compared to the men taking two placebos (no vitamin E and no selenium) after an average of seven years -- 5.5 of the years on supplements followed by 1.5 years not taking supplements.
In the men in SELECT who took only placebos, after seven years, 65 prostate cancer cases were diagnosed for every 1,000 men. For the men assigned to take vitamin E only, for every 1,000 men, there were 76 cases of prostate cancer diagnosed (11 additional cases of prostate cancer per 1,000 men over seven years).
What does a 17 percent increased risk of prostate cancer mean to men who take vitamin E but who were not SELECT participants?
The incidence rate of prostate cancer in the placebo arm of SELECT is similar to the incidence rate for men in the U.S. age 65 and older (4). Thus, the estimate of increased risk for men of SELECT age taking vitamin E supplements of 400 IUs daily would be about the same as for the men on SELECT taking vitamin E only (i.e. about 17 percent).
In other words, if 1,000 men similar to those on SELECT were followed with annual physician visits, one would expect 65 of them to be diagnosed with prostate cancer over a seven year period. If these same men took 400 IUs of vitamin E daily for 5.5 years, researchers would expect 76 of them to be diagnosed with prostate cancer over a seven year period (i.e., 11 additional cases over seven years).
Why didn't the vitamin E supplements prevent prostate cancer as expected? Why did the men taking vitamin E have more prostate cancers?
There are many reasons why the vitamin E supplements may not have prevented prostate cancer. Two of the most likely reasons look back at the Alpha-Tocopherol Beta Carotene (ATBC) Cancer Prevention trial, a study designed to test vitamin E and beta carotene for lung cancer prevention in smokers (10). In the ATBC, a reduction in prostate cancer incidence was observed, but this secondary finding may have been due to chance, as the study was not designed to determine prostate cancer risk. Another possible reason that men in ATBC had a reduction in prostate cancer incidence, while men on SELECT did not, is that the dose of vitamin E used in SELECT (400 IU/day) was higher than the dose used in the ATBC (50 mg/day). Researchers sometimes talk about a "U-shaped response curve" where very low or very high blood levels of a nutrient are harmful but more moderate levels are beneficial; while the ATBC dose may have been preventive, the SELECT dose may have been too large to have a prevention benefit.
Did the researchers find any difference in the effect of the supplements based on the level of vitamin E or selenium in participants when they joined the trial?
Yes. In an analysis published in 2014, men who had high levels of selenium at the start of the trial, as assessed by measures of selenium in their toenail clippings, had almost double the chance of developing a high-grade prostate cancer if they took the selenium supplement compared to men with low levels of selenium at the start of the trial. This finding was unexpected, as previous studies had shown that men with low levels of selenium had an increased risk of prostate cancer that was reduced with supplements (11, 12). Additionally, men with low levels of selenium at the start of the trial had double the chance of developing a high-grade prostate cancer if they took the vitamin E supplement.
Do the new findings about the men’s selenium status help to explain the overall trial results better?
Yes. In 2011, data showed that men taking vitamin E alone had a significantly increased risk of prostate cancer, but men taking vitamin E plus selenium did not. If men had low selenium levels at the start of the trial, the selenium supplement may have counteracted a negative effect of the vitamin E supplement.
Should men take vitamin E or selenium supplements for cancer prevention?
No. Scientists do not understand how these supplements really work and more importantly, the interactions that these supplements have together or with foods, drugs, or other supplements. There are no clinical trials that show a benefit from taking vitamin E or selenium to reduce the risk of prostate cancer or any other cancer or heart disease (2, 3, 5-9). While the men in SELECT who took both vitamin E and selenium did not have a statistically significant increase in their risk for prostate cancer, they also did not have a reduced risk of prostate cancer or any other cancer or heart disease. SELECT researchers were surprised by the findings in the men who took both vitamin E and selenium, and while the 2014 analysis suggests possible reasons for the findings, the mechanism remains unclear.
Why did SELECT use a dose of 400 IU of vitamin E? What form of vitamin E was used?
At the time SELECT was being designed, trials to test vitamin E in other common diseases were also under way. These trials were studying 400 IU to 660 IU of vitamin E to prevent macular degeneration, Alzheimer's disease, or heart disease. Preliminary evidence suggested the outcomes would be favorable. SELECT researchers matched the doses in these trials to ensure data at the 400 IU dose was available if using the supplement for other diseases became common.
The form of vitamin E given was dl-alpha tocopheryl acetate, the form of vitamin E found to reduce prostate cancer incidence in the ATBC.
Did the African American men in SELECT have any different outcomes compared to the rest of the participants?
Probably not, but the number of African American men in the trial is too small to be certain that separate analyses would be statistically sound. In SELECT, African American men were permitted to join the trial at a younger age than men of other races (at age 50 vs. 55) because of their increased risk of prostate cancer. About 12 percent of the participants (4,314 of 34,888) identified themselves as African-American, and they were represented equally across the study arms (as would be expected in a randomized trial). Although this percentage reflects the proportional representation of African Americans in the United States, the number of men is not large enough to do thorough, separate statistical analyses of each SELECT study endpoint. However, SELECT researchers did look at cancer rates and other measures by race and did not see any differences in the response to the supplements.
Who got which supplement?
SELECT was a randomized, controlled clinical trial. The men who participated took two capsules a day. They were randomly assigned (that is, assigned by chance) to receive:
- Selenium and vitamin E
- Selenium and a placebo
- Vitamin E and a placebo, or
- Two placebos. Two placebos were used in the trial: one looked like a selenium capsule; the other looked like a vitamin E capsule. Each placebo contained only inactive ingredients. Neither the participants nor the researchers knew who received the selenium and vitamin E, or the placebos, a process known as blinding or masking. 23.
Who was eligible to participate in SELECT? Were there restrictions on eligibility?
African-American men had to be age 50 or older to participate, and men of other races and ethnicities had to be 55 or older. The age for eligibility was lower for African-American men because, on average, they develop prostate cancer at an earlier age and have an overall increased risk of developing prostate cancer.
Many diseases, including prostate cancer, occur more frequently in older persons. The risk of developing prostate cancer increases with age. More than 90 percent of prostate cancer cases occur in men age 55 or older (4).
What happened if a participant developed prostate cancer while involved in SELECT?
Participants diagnosed with prostate cancer during the study underwent treatment within their community, based upon community treatment standards. These participants continued to be followed by the SELECT study staff. Costs for diagnosis and treatment of prostate problems, prostate cancer, or other medical conditions during the study were charged to the participant in the same way as if he were not part of the trial. A participant's insurance should pay for diagnosis and treatment according to the plan's policies. If the participant has no insurance, social services may be available at the local level to cover costs for diagnosis and treatment.
Why didn't the selenium supplement in SELECT prevent prostate cancer?
Researchers don't know why. There are several possible explanations why selenium supplements did not prevent prostate cancer in men on SELECT. These reasons include:
- The findings of reduced prostate cancer incidence associated with selenium supplementation in the Nutritional Prevention of cancer (NPC) study may not have been correct and selenium may not affect prostate cancer risk.
- The participants in the NPC study were deficient in selenium compared to the men in SELECT who were not selected because of a likely selenium-deficiency; it may be that selenium only reduces the risk of prostate cancer in selenium-deficient men and not in the general population.
- The supplements given to the men in SELECT may have exceeded the best dose to prevent prostate cancer.
- The formulation of selenium used in the NPC trial (high-selenium yeast) may have been more active than the l-selenomethionine used in SELECT and this may have prevented researchers from seeing a cancer prevention effect. Arguing against this was the fact that early tests showed that, for the selenium yeast, the amount of selenium per dose varied in the NPC trial – this was why this form of selenium was not used in SELECT. Additionally, the inorganic compounds present in the yeast can be toxic and can lead to lower body reserves of selenium. The SELECT biorepository data and samples will help to support research that may answer this question.
References
- Lippman SM, Goodman PJ, Klein EA, et al. Designing the selenium and vitamin E cancer prevention trial. JNCI 2005; 97(2):94-102.
- Lippman SM, Klein EA, Goodman PJ, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers. JAMA 2009; 301(1). Published online December 9, 2008. Print edition January 2009.
- EA Klein, IM Thompson, CM Tangen, et al. Vitamin E and the Risk of Prostate Cancer: Results of The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2011; 306(14) 1549-1556.
- Howlader N, Noone AM, Krapcho M, et al (eds). SEER Cancer Statistics Review, 1975-2008, National Cancer Institute. Bethesda, MD, http://seer.cancer.gov/csr/1975_2008, based on November 2010 SEER data submission, posted to the SEER web site, 2011.
- Heinonen OP, Albanes D, Virtamo J, et al. Prostate cancer and supplementation with alpha-tocopherol and beta-carotene: Incidence and mortality in a controlled trial. JNCI 1998; 90(6):440-446.
- Kristal AR, Darke AK, Morris S, et al. Baseline selenium status and effects of selenium and vitamin E supplementation on prostate cancer risk. J Natl Cancer Inst 2014.
- Clark LC, Combs GF Jr., Turnbull BW, et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial: Nutritional Prevention of Cancer Study Group. JAMA 1996; 276(24):1957-1963.
- Duffield-Lillico AJ, Reid ME, Turnbull BW, et al. Baseline characteristics and the effect of selenium supplementation on cancer incidence in a randomized clinical trial: A summary report of the Nutritional Prevention of Cancer Trial. Cancer Epidemiology, Biomarkers & Prevention 2002; 11(7):630-639.
- Yusuf S, Dagenais G, Pogue J, et al. Vitamin E supplementation and cardiovascular events in high risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. New England Journal of Medicine. 2000;342:154-60.
- Sesso HD, Buring JE, Christen WG, et al. Vitamins E and C in the prevention of cardiovascular disease in men: the Physicians' Health Study II randomized controlled trial. JAMA. 2008; 300(18):2123-33.
- Lee IM, Cook NR, Gaziano JM, et al. Vitamin E in the primary prevention of cardiovascular disease and cancer: the Women's Health Study: a randomized controlled trial. JAMA. 2005; 294(1):56-65.
- Lonn E, Bosch J, Yusuf S, et al. Effects of long-term vitamin E supplementation on cardiovascular events and cancer: A randomized controlled trial. JAMA 2005; 293(11):1338-1347.
- Miller ER III, Pastor-Barriuso R, Dalal D, et al. Meta-analysis: High-dosage vitamin E supplementation may increase all-cause mortality. Annals of Internal Medicine 2005; 142(1):37-46.