Bladder Cancer Screening
Screening is looking for cancer before a person has any symptoms. This can help find cancer at an early stage. When abnormal tissue or cancer is found early, it may be easier to treat.
Tests to screen for bladder cancer
There is no standard screening test for bladder cancer in people at average risk. When enough evidence has been collected to show that a screening test is safe, accurate, and useful for people at average risk of bladder cancer, it will become a standard test. However, certain tests may be used to screen for bladder cancer in people who have had bladder cancer in the past or who are at an increased risk of developing it.
Hematuria test
Hematuria is red blood cells in the urine. It may be caused by cancer or by other conditions. A hematuria test is used to check for blood in a sample of urine by viewing it under a microscope or using a special test strip. The test may be repeated over time.
Urine cytology
Urine cytology is a lab test in which a sample of urine is checked under a microscope for abnormal cells.
Urine tumor marker tests
Urinary tumor markers are substances found in the urine that are either made by bladder cancer cells or that the body makes in response to bladder cancer. For this test, a sample of urine is checked in the lab to detect the presence of these substances. Urine tumor marker tests may also be used to help diagnose some types of bladder cancer.
Cystoscopy
Cystoscopy is a procedure to look inside the bladder and urethra to check for abnormal areas. A cystoscope (a thin, lighted tube) is inserted through the urethra into the bladder. Tissue samples may be taken for biopsy. 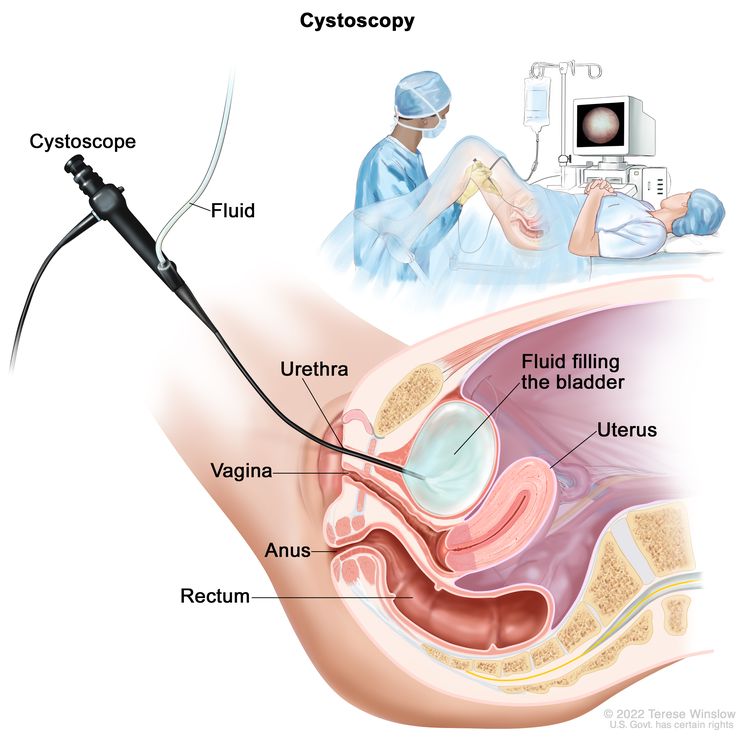
Risks of bladder cancer screening
Screening tests have benefits and risks. Talk with your doctor about whether bladder cancer screening is right for you.
Potential risks of harm from bladder cancer screening include false-positive and false-negative test results:
- False-positive test results. Screening test results may appear to be abnormal even though no cancer is present. A false-positive test result (one that shows there is cancer when there isn't) can cause anxiety and is usually followed by more tests (such as cystoscopy or other invasive procedures), which also have risks. False-positive results often occur with hematuria testing; blood in the urine is usually caused by conditions other than cancer.
- False-negative test results. Screening test results may appear to be normal even though bladder cancer is present. A person who receives a false-negative test result (one that shows there is no cancer when there is) may delay seeking medical care even if there are symptoms.