AI applications in cancer research and care
NCI research is advancing the use of AI across the spectrum of cancer research and care, including mechanisms of cancer, cancer screening and diagnosis, drug discovery, cancer surveillance, and health care delivery.
Advancing fundamental knowledge of cancer biology
AI methods are being applied to advance knowledge about mechanisms of cancer initiation, progression, and metastasis. For example:
- The body of scientific literature provides a vast resource of information and knowledge on cancer. Artificial intelligence experts are taking advantage of large language models to develop new computational tools to improve knowledge extraction from research publications.
- As part of the collaboration between NCI and the Department of Energy, researchers are using AI to simulate the atomic behavior of the RAS protein, one of the most commonly mutated proteins in cancer. A better understanding of how RAS interacts with other proteins could help scientists find new avenues to target cancer-causing mutations in the RAS gene.
Expediting cancer screening, detection, and diagnosis
AI is helping to improve the speed, accuracy, and reliability of some cancer screening and detection methods. For example:
- The Food and Drug Administration has authorized the marketing of AI-based software to help pathologists identify areas of prostate biopsy images that may contain cancer.
- Medical images such as mammograms can be rapidly processed with the help of AI, allowing radiologists to focus their time on other tasks that require their technical judgement. NCI-supported research has shown that AI imaging algorithms not only improve breast cancer detection on mammography but can also help predict long-term risk of invasive breast cancers.
- NCI scientists are using AI to improve cervical and prostate cancer screening. One group of NCI researchers and their collaborators developed a deep learning approach for the automated detection of precancerous cervical lesions from digital images.
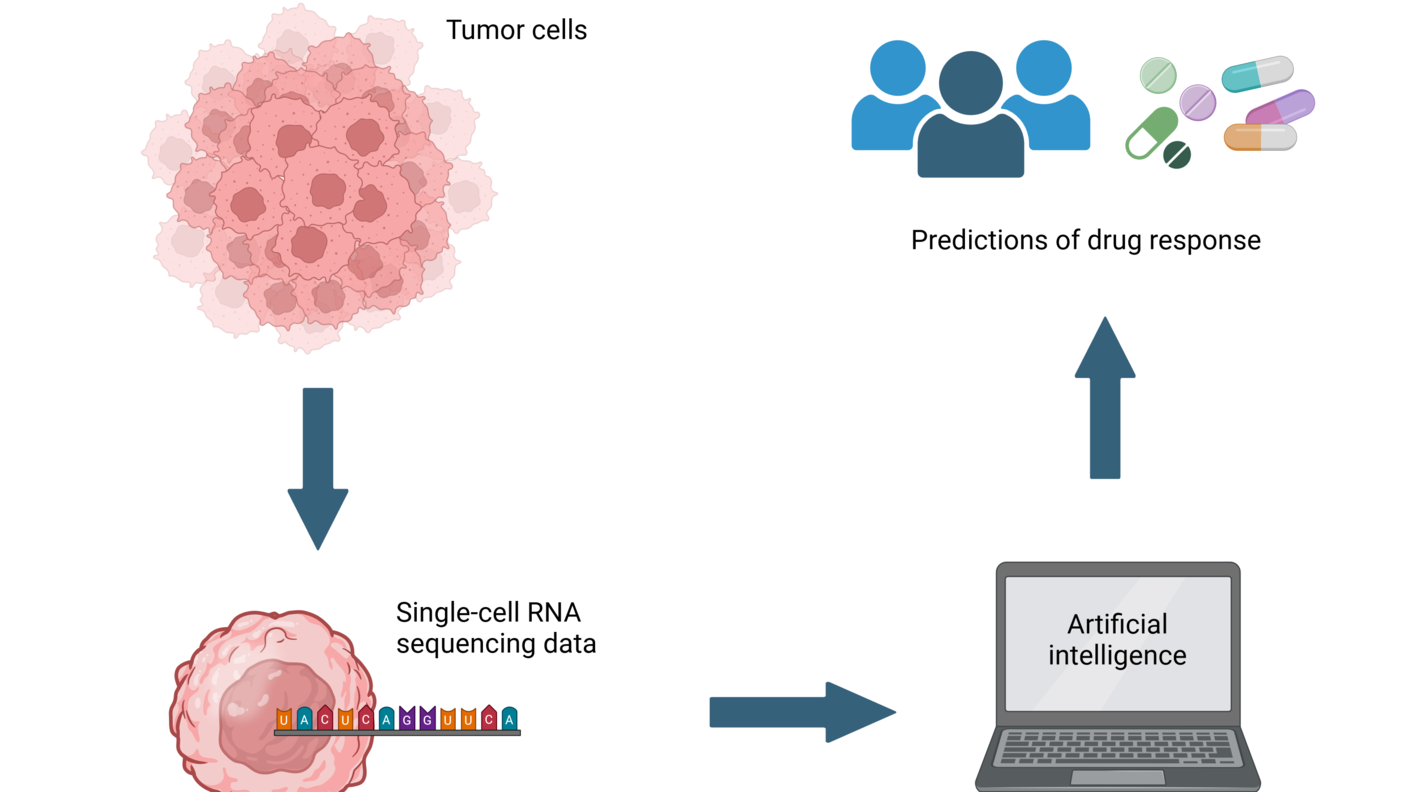
Accelerating cancer drug discovery
AI is being used in many ways to develop new treatments for cancer through novel approaches to drug discovery and design, drug repurposing, and predicting patient responses to treatment. For example:
- NCI researchers and their colleagues have employed AI to help predict how immune cells called T cells will respond to tumors. They used machine learning to find patterns within a large volume of human and mouse T-cell activation data and predict T-cell behavior with the aim of improving immunotherapies.
- New AI methods are helping researchers study the biological mechanisms of drug response. For example, NCI-supported researchers have developed predictive AI models that use deep learning to provide a map of common drug response pathways.
Facilitating precision cancer treatment
Precision oncology is an approach to cancer care in which information about a tumor, such as tumor biomarkers, are used to guide treatment. This form of cancer care often involves analyzing a large amount of data with advanced computational approaches to help physicians make decisions. For example:
- AI technology may help to expedite genetic subtyping of brain tumor tissue during a patient’s surgery, dramatically speeding up treatment decisions for these patients.
- NCI-funded researchers have developed an AI model that can predict survival outcomes for patients with invasive, nonmetastatic breast cancer using digital pathology slide images.
- AI methods can also be used to better integrate multiple data types from patients to improve clinical decision making. For example, NCI-funded researchers have developed AI methods to combine histopathology data and molecular data to predict outcomes from patients with brain cancer that perform better than models using one data type.
Improving cancer surveillance
Cancer surveillance is the ongoing collection of patient information and cancer statistics. AI methods are being used to accelerate information extraction for surveillance reporting and to identify patterns in population-level cancer data. For example:
- A collaboration between NCI and the Department of Energy called Modeling Outcomes Using Surveillance Data and Scalable Artificial Intelligence for Cancer (MOSSAIC) is using AI approaches to submit data to NCI’s Surveillance, Epidemiology, and End Results (SEER) program more quickly. As part of this effort, scientists developed AI algorithms to extract tumor features automatically from unstructured clinical text, saving thousands of hours of manual processing time. This process will help researchers better understand how new diagnostic methods, treatments, and other factors affect patient outcomes.
- NCI-supported researchers are developing deep learning algorithms trained on population-level disease data to predict a person’s risk of pancreatic cancer, paving the way for early detection.
- Large language models for electronic health record surveillance are helping researchers better understand social determinants of health that may be critical for preventing, detecting, and treating cancer.
Improving access to cancer care
AI tools could also help make high-quality care accessible to more patients, even those who live far from cancer specialists or in low-resource settings, potentially helping to reduce cancer health disparities.
- With the emergence of chatbots, cancer researchers are probing whether this technology can be used in cancer care. Some studies have suggested that chatbots could help provide patients with tailored cancer information and even help draft physician responses to patient questions.
Challenges and opportunities for AI in cancer research
AI presents an unprecedented opportunity for rapid advances in understanding of cancer biology and optimization of patient care. However, if data used to train AI models are not appropriately diverse and representative of the broader population, these models can perpetuate medical bias. There is a need for broadly accepted and adopted standards for development of AI and machine learning models to mitigate bias and ensure reproducibility.
There is also a need for further randomized clinical trials to validate applications of AI and machine learning technologies in clinical practice. And advancing explainable artificial intelligence will be essential to integrate AI and machine learning technologies into clinical workflows.
NCI is committed to supporting research aimed at addressing these challenges and advancing the development of AI methods that will accelerate the effort to end cancer as we know it.