The Childhood Cancer Data Initiative Molecular Characterization Initiative
What is molecular characterization?
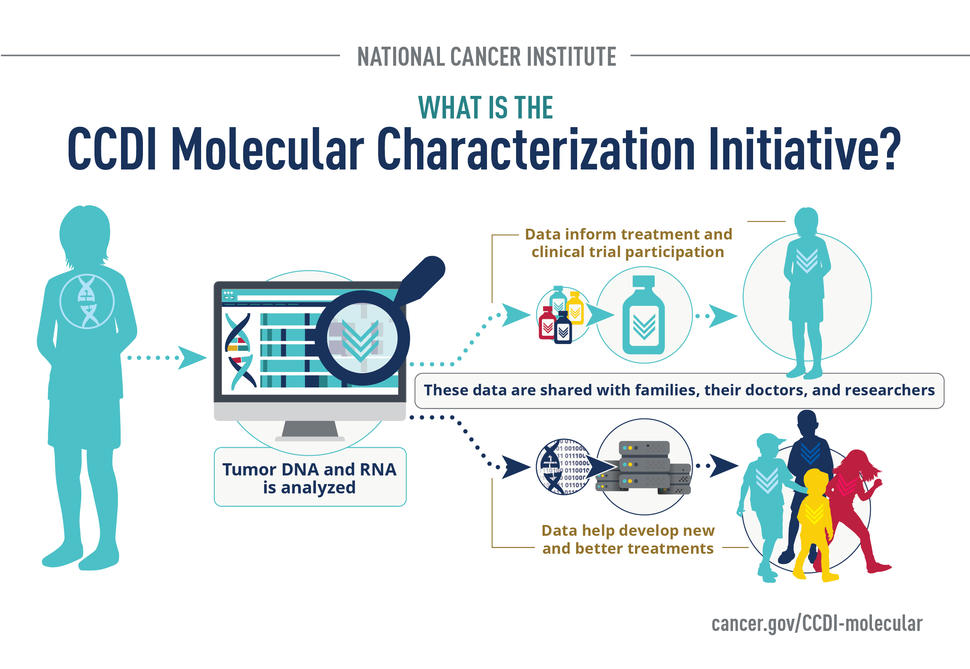
Molecular characterization tests look at the DNA and RNA in a tumor and in blood. Looking at cancer at a molecular level helps to provide a precise diagnosis and tell doctors more about what might be causing or driving an individual cancer.
It also helps determine the most effective treatment and inform what clinical trials someone may be able to participate in.
What is the CCDI Molecular Characterization Initiative?
The Molecular Characterization Initiative is part of NCI’s Childhood Cancer Data Initiative (CCDI) and is a national collaboration between the childhood cancer community, including the Children’s Oncology Group, advocates, pediatric oncologists, researchers, data scientists, children and adolescent and young adults (AYAs) with cancer, and families. It provides state-of-the-art molecular characterization at the time of diagnosis that helps participants and doctors select the best and most appropriate treatment.
How it works
A specialized and accredited lab analyzes tumor and blood samples that may have already been provided for initial diagnosis to learn about a participant’s cancer at its most basic, molecular level. Results will be shared with them and their doctors to inform treatment options and potential clinical trial participation.
Later, these data will be made accessible to researchers for future studies. Data that are shared with researchers have no personal information and cannot be connected directly to participants.
Participation in the initiative is voluntary and the costs to participate are covered by the program.
Helping advance childhood cancer research
These data will also enable researchers to create better clinical trials, learn about the origins and drivers of childhood cancer, and make faster progress in the development of new and better treatments, especially for childhood cancers with limited effective treatment options.
Who can participate in the Molecular Characterization Initiative?
Currently, children and AYAs who are newly diagnosed with a central nervous system tumor, soft tissue sarcoma, high-risk neuroblastoma, or rare tumor, and who are receiving care at a hospital affiliated with the Children’s Oncology Group are eligible to participate in the initiative.
As of February 2025, the initiative has enrolled more than 6,000 participants.
To join at this time, a child or AYA must:
- be newly diagnosed (have not had any treatments for their cancer yet)
- be 25 years old or younger
- be diagnosed with a central nervous system tumor (tumors of the brain and spine), soft tissue sarcoma, high-risk neuroblastoma, or rare tumor* (childhood cancers with a low number of patients, which have been hard to study and understand)
- get cancer care from a hospital affiliated with the Children’s Oncology Group
If a child or AYA meets the above criteria, they can get more information on how to participate by talking with their Children's Oncology Group–affiliated doctor.
Enrollment will soon open to children and AYAs with Ewing sarcoma. The initiative will continue to expand to children and AYAs outside of Children’s Oncology Group–affiliated hospitals, those with other childhood cancers, and those whose cancer has returned.
*Rare tumors include adrenocortical, colorectal, nasopharyngeal, thyroid, and any other (extra-cranial) carcinomas, as well as desmoplastic small round cell tumors, gastrointestinal or gonadal stromal tumors, melanoma, neuroendocrine tumors, pancreatoblastoma, pleuropulmonary blastoma, and retinoblastoma.
Where can these data be accessed?
Search for initiative data in the CCDI Hub Explore Dashboard using the study title, "Molecular Characterization Initiative."
Controlled-access initiative data can be accessed through the database of Genotypes and Phenotypes (dbGaP) using accession number phs002790. Note that a Data Access Request must be submitted.
Open-access hematoxylin and eosin stained images and their manifests are available to explore and visualize in the Imaging Data Commons. These images are in Digital Imaging and Communications in Medicine format.
For more information about Molecular Characterization Initiative testing and data, visit the CCDI Hub.
How will personal health information be protected?
Privacy is extremely important to NCI, and personal information will only be shared with participants and their medical team.
Data from the Molecular Characterization Initiative and from any clinical trials will have personal health information removed before it is made accessible to researchers. There will be no way for researchers or anyone to connect the data to participants directly.
Where can you learn more about molecular characterization?
Talk with a doctor
To participate in the Molecular Characterization Initiative, an eligible child or AYA must be receiving care at a Children's Oncology Group–affiliated hospital. To check a hospital's affiliation, visit the group's institution locator page.
If a child or AYA is not eligible for the initiative at this time, they can talk with their doctor or oncology team about molecular characterization. They can also discuss clinical trials that might be studying new ways to treat specific types of cancer. Additional clinical trial information is available on trials.cancer.gov.
Reach out to NCI's Cancer Information Service
Please contact NCI’s Cancer Information Service via chat, email, or phone (both in English and Spanish) with additional questions. Information specialists can help find information on cancer in children and AYAs, clinical trials, and relevant community resources.