About NCI-COG Pediatric MATCH
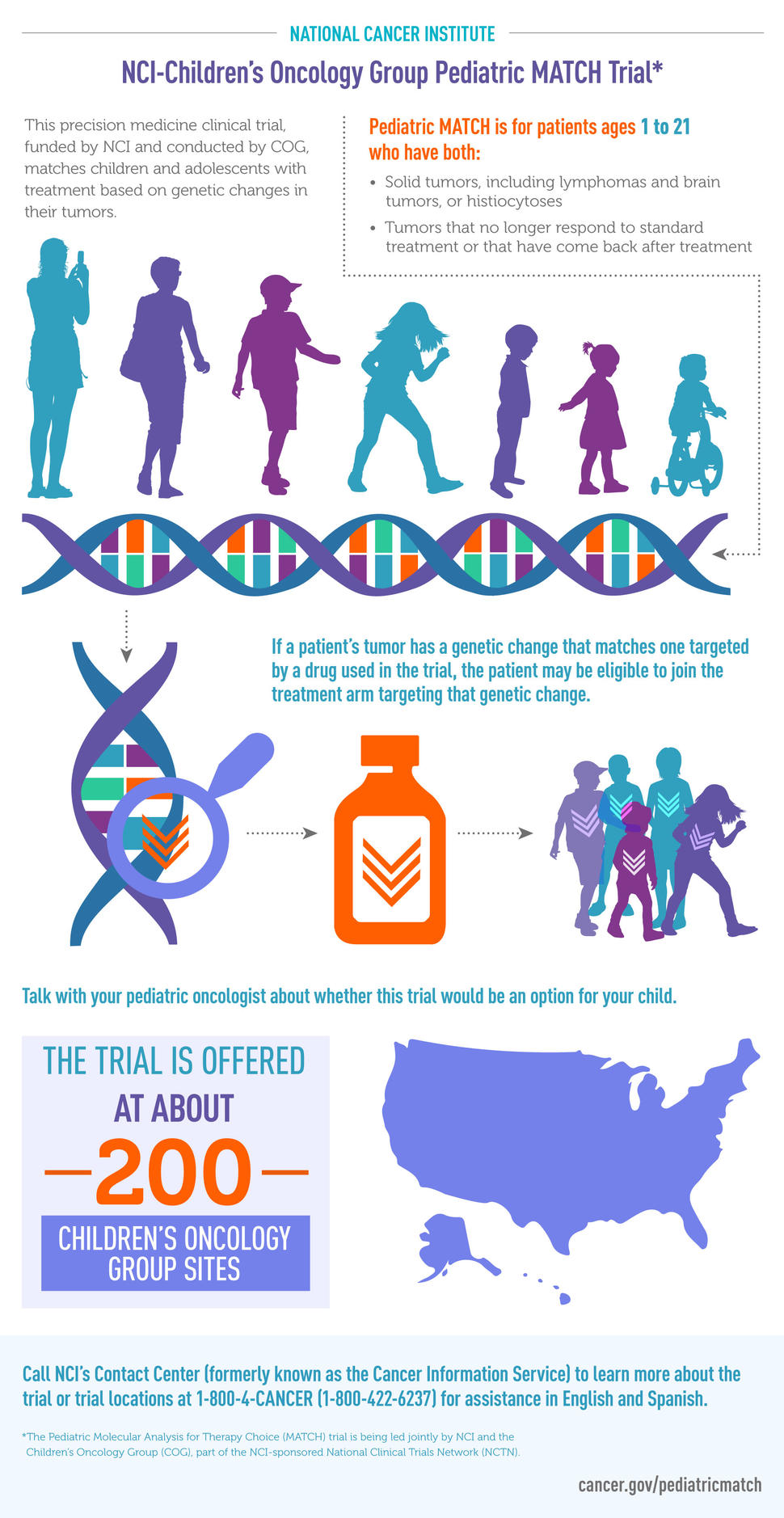
NCI-COG Pediatric MATCH is an cancer treatment clinical trial for children, teens, and young adults, aged 1 to 21 years, that is testing the use of precision medicine for childhood cancers. In this trial, young people with solid tumors that are not responding to treatment are assigned to an experimental treatment based on the genetic changes found in their tumors rather than on their type of cancer or cancer site. The genetic changes are found through genomic sequencing, which is a laboratory method used to determine the genetic makeup of cancer cells.
If you are interested in having your child take part in Pediatric MATCH, talk with their oncologist. The oncologist will decide if their tumor is a candidate for testing. For your child to take part in the study, their tumor would need to contain certain genetic changes that are targeted in the study. They would also need to meet other eligibility criteria.
If your child enrolls in the study, they will receive the experimental treatment as long as their tumors are stable or getting smaller.
Types of cancers treated in Pediatric MATCH
Pediatric MATCH is for young people with advanced solid tumors—including non-Hodgkin lymphomas, brain tumors, and histiocytoses—whose cancer has gotten worse while on treatment or has come back after treatment.
Where Pediatric MATCH is taking place
Pediatric MATCH is taking place at about 200 children’s hospitals, university medical centers, and cancer centers in the United States and Australia that are part of the Children’s Oncology Group (COG). For your child to take part in the study, their oncologist must be at one of the study sites. To find a site, visit the Children’s Oncology Group institution locator page.
Goals of Pediatric MATCH
Pediatric MATCH researchers want to find out whether it is effective to treat cancer in children, teens, and young adults by targeting certain genetic changes in their tumors with specific targeted drugs, no matter the type of cancer or cancer site.
The study will also help doctors and researchers learn more about how often children, teens, and young adults with cancer have genetic changes that can be targeted by certain drugs. It will also help them learn more about the genetic changes in childhood cancers that get worse or come back after standard treatment. The knowledge gained will help lead to better treatments for young people with cancer.
Pediatric MATCH treatment arms
Pediatric MATCH is winding down, most treatment arms have closed, and some results have already been published. Right now, there is one arm still accepting young people that is testing ensartinib in tumors with changes in ALK or ROS1.
Pediatric MATCH Results
NCI-COG Pediatric MATCH enrolled 1371 children, teens, and young adults on 13 different treatment arms. Follow-up will continue for all patients receiving treatment for a total of 5 years from enrollment.
Researchers continue to publish papers about the findings for each arm. These papers describe the findings in detail, including the results of each of treatment, genomic analyses, and what was learned from this information.
Trial costs
Study drugs are provided by the drug companies and are free of charge for those in the study.
Unless informed that certain tests are being done at no charge, you or your health insurance plan will need to cover all other costs, including the cost of tests, procedures, or medicines to manage any side effects of the treatment. Patients will not be paid to take part in this study.
Other clinical trial options
If your child is not eligible for Pediatric MATCH, you can talk with their oncologist about other clinical trials that may be an option. If you want to search for trials on your own, visit NCI’s clinical trials search page. If you would like help with your search, NCI’s cancer information specialists can assist you in English and Spanish. You can contact NCI's Cancer Information Service at 1-800-4-CANCER (1-800-422-6237) or via NCI's LiveHelp service.
Pediatric MATCH trial collaborators
The study was developed and is led jointly by the NCI and the Children’s Oncology Group, part of the NCI-sponsored National Clinical Trials Network.
Learn more about NCI-COG Pediatric MATCH
To learn more about Pediatric MATCH, parents should start by speaking with their children’s doctors or healthcare team. More details about Pediatric MATCH are available in the protocol summary.