A common but poorly understood syndrome
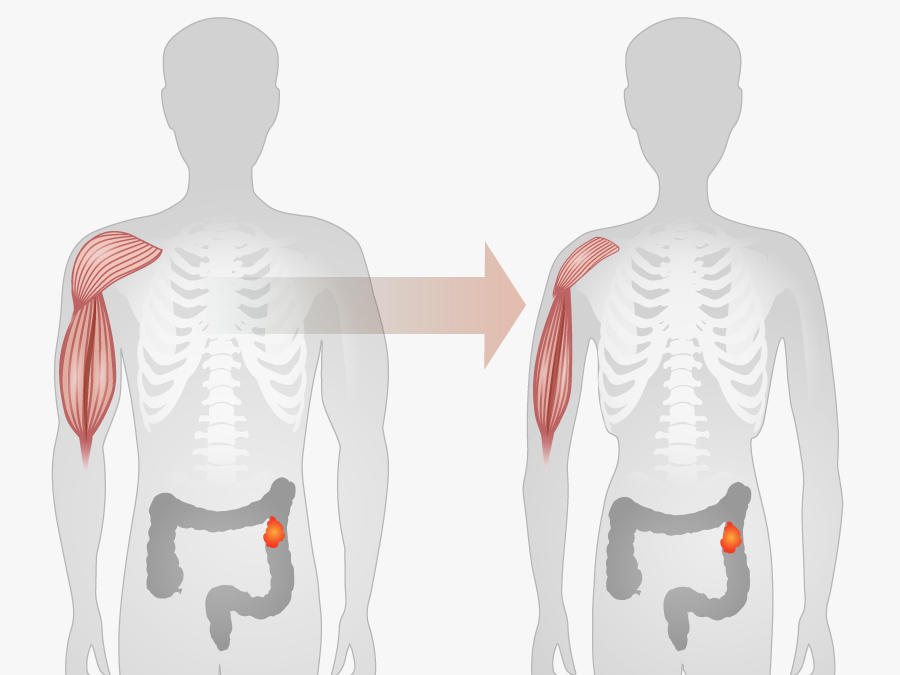
Cachexia is estimated to occur in up to 80% of people with advanced cancer, depending on the cancer type and how well they respond to cancer treatment. And it’s thought to directly cause up to 30% of cancer deaths, often because of heart or respiratory failure related to muscle loss.
In its most severe form, the physical deterioration that accompanies cachexia can leave a person not only weak and fatigued, but unable or unwilling to eat, and with alarming changes in appearance. For many, these problems can turn everyday life activities—a trip to the grocery store, meeting a friend for coffee, taking a bath—into Herculean tasks, if not outright impossibilities.
Mental health struggles often follow. As Felicia recounted at a meeting on cachexia held in late 2021 by the advocacy group Cancer Support Community, cachexia’s physical toll gave way to “worry, stress, anxiety, and mental anguish.”
And the mental health impact doesn’t stop there. For the family and loved ones of a person experiencing cachexia, witnessing this physical and mental decline can leave them feeling helpless and confused.
Unfortunately, there are no effective treatments for cachexia, explained Teresa Zimmers, Ph.D., of the Indiana University Simon Comprehensive Cancer Center, a leading expert on the condition.
In large part, that’s because for many years cachexia was understudied and, as a consequence, poorly understood. But that is changing, said Dr. Zimmers, the current president of the Cancer Cachexia Society. The number of researchers studying cancer-related cachexia is growing, bringing with them new information about how this wasting syndrome develops and clinical trials testing new treatments for it.
At least one drug has been shown in large studies to help people with cancer cachexia maintain lean muscle mass, which is critical for daily functioning and the ability to tolerate cancer treatments. And several other experimental drugs that showed remarkable effects against cachexia in mice are now being tested in human studies.
Researchers have also launched clinical trials to test exercise– and nutrition–based treatments for cancer cachexia.
“We are at the start of an exponential growth phase,” Dr. Zimmers said. “So while there remains much to be done to address this unmet need—including educating clinicians, patients, and caregivers, discovering mechanisms, and testing new and effective treatments—I think it's an auspicious time for cachexia research.”
Communicating about cancer cachexia
Cachexia is most common in people with advanced pancreatic and lung cancer, but it also frequently occurs in people with other types of cancer, including head and neck, colorectal, ovarian, and liver cancers.
The current definition of cancer cachexia is a loss of 5% or more of body weight over the preceding 6 months, accompanied by any of a handful of other symptoms, including fatigue and reduced strength.
But there is general agreement among cachexia researchers that this definition falls short, because it’s a crude measure of a complex physiologic and biologic condition. And diagnosing it is often complicated by the similar effects that cancer and its treatment can have on the body.
All of which makes communicating about cachexia difficult, explained Eric Roeland, M.D., an oncologist at the Oregon Health & Science University, who specializes in treating cancers in which cachexia is commonly seen.
Most patients “are completely unfamiliar with the term and what [cachexia] is,” Dr. Roeland said. And too often, he continued, oncologists erroneously view cachexia “as an end-stage complication of uncontrolled cancer.”
Richard Dunne, M.D., an oncologist at the University of Rochester’s Wilmot Cancer Institute, primarily sees patients with pancreatic and other gastrointestinal cancers. He often raises the prospect of cachexia—even if he doesn’t necessarily use that term—the first time he is seeing a newly diagnosed patient.
For patients who have lost weight in the months leading up to their diagnosis, “I want to get at why that might be happening,” Dr. Dunne explained, to help him determine whether cachexia might already have set in.
In addition to questions about weight loss and appetite, he also tries to determine whether patients have cachexia-related functional problems.
But, according to Jose Garcia, M.D., Ph.D., who studies cancer cachexia at the University of Washington, there aren’t broadly accepted ways of assessing function. A lot of cachexia-related research has focused on measuring changes in body mass, Dr. Garcia explained at the Cancer Support Community meeting. As a consequence, Dr. Garcia said, there’s “a lack of consensus on … the clinically important tools for assessing physical function.”
Along with a method commonly used by oncologists called the Karnofsky score, oncologists may use simple tools like seeing how well patients can climb a few stairs or walk on a slowly moving treadmill, Dr. Garcia explained.
Some oncologists have their own measures. Dr. Roeland, for example, will often test whether patients can stand up from a chair with their arms crossed or stand on one foot with their arms extended “to test length, strength, and balance,” something he adopted from his oncology nurse colleagues.
“Those are quick assessments,” he said. But they get at important functional abilities like balance and can be done within the strict time constraints oncologists and their staff often face, “where we’re seeing patients with complex problems in 20- to 30-minute blocks.”
Current treatment approaches
Once it’s been determined that cachexia has set in, what’s next?
Until recently, there were few formal recommendations on treating cachexia in people with cancer. In 2020, however, an expert committee convened by the American Society of Clinical Oncology, co-chaired by Dr. Roeland, released the first-ever professional guidelines on the topic.
Cachexia, the guidelines committee noted, is not synonymous with malnutrition. However, based on the committee’s review of evidence from published studies, their only specific recommendation for treating muscle and weight loss was “dietary counseling.” But even then, they wrote, evidence that dietary counseling and changes can counter or slow these problems “remains limited.”
Dr. Dunne said his approach entails “treating individual symptoms [that] we think might be contributing to a loss of weight or muscle mass.” That can include referrals to registered dieticians as well as to physical therapy.
Despite a lack of data from large clinical trials to guide the treatment of people with cachexia, a handful of hospitals have dedicated cancer cachexia programs. The staff that run these programs rely on the evidence from published studies of managing cancer cachexia and best practices developed at their institutions and others.
One of these programs is McGill University’s Cancer Nutrition Rehabilitation Program at Jewish General Hospital in Montreal.
McGill’s program includes dieticians, physiotherapists, a physician, and an advanced practice nurse—an array of expertise that can address the different problems and symptoms people with cachexia face, explained Monica Parmar, R.N., a program team member and clinical researcher.
Patient assessments include analysis of body composition (e.g., lean muscle mass, body mass index), nutritional status, cachexia-related symptoms and how those symptoms affect their quality of life, and functional testing, such as a 6-minute walk test on a treadmill.
“Our goal as a team is to really align with the patient,” Ms. Parmar explained.
That means focusing their strength– and endurance–based work with patients on things that will “allow them to participate more actively in their daily life in a way that's meaningful to them,” she said. “For some people, it could be that I want to pick up my grandchild … or I want to be able to continue walking my dog.”
Participation in the program, Ms. Parmar said, has shown to improve people’s quality of life.
At the Shirley Ryan AbilityLab in Chicago, Ishan Roy, M.D., Ph.D., runs a physical rehabilitation program for people with cancer, with a research emphasis on cancer cachexia.
Dr. Roy’s work with patients and his research revolves around the concept of “treating exercise like medicine,” he explained. That includes assessing how exercise can be adapted to a person’s specific physical abilities, he continued, and trying to learn how “exercise can be more precisely delivered so that it can be combined with medical therapies or nutritional changes.”
For many patients with cachexia, he uses gait training—that is, exercises that can improve walking and related movements. Improving core strength is another area of focus. Both are driven by findings from studies linking improved mobility with an increased ability to engage in daily activities, he said.
But much of his work with patients “is being driven by my clinical judgment,” he acknowledged. “We just don't have a lot of evidence [to know] what works best.”
What causes cancer cachexia?
The clinical and emotional fallout experienced by people suffering from cancer cachexia is mirrored in many ways by the frustration among researchers with the limited progress in developing effective treatments for it.
Finding those treatments, of course, requires understanding what causes cachexia in the first place. And researchers have nailed down a general outline of the biologic changes that cause the body to start to waste away.
To begin, the widespread inflammation that often accompanies cancer is thought to be the primary instigating and sustaining force behind cachexia, explained Joanna Watson, Ph.D., of NCI’s Division of Cancer Biology.
The inflammation causes multiple biologic behaviors to go awry, including disruptions in how muscle and fat cells make and use energy, known as metabolism.
“Factors produced by the tumor and the ‘host,’ or patient … disrupt the metabolic balance,” Dr. Watson said. This disruption creates an environment where muscle and fat cells break down far faster than they can be replenished.
Studies have also identified another feature of cancer cachexia, a “cross-talk” among tumor cells and cells in fat, muscle, the brain, and other parts of the body—the molecular equivalent of a vicious neighborhood gossip circle.
Much of this cross-talk involves chemical messengers called cytokines that are released mainly by immune cells, but also by other types of cells, explained Denis Guttridge, Ph.D., of the Medical University of South Carolina, who has spent decades studying cachexia’s underlying biology.
Inflammation influences both the levels and behavior of many of these cytokines, Dr. Guttridge explained, which has ripple effects throughout the body that can cause skeletal muscle to wither (sarcopenia) and dampen the desire to eat (anorexia), further robbing the body of the fuel it needs.
What is clear at this point, he continued, is that different cytokines “are each contributing to the pathology [of cachexia] in different ways."
Fortunately, Dr. Zimmers said, cachexia researchers are beginning to untangle this cross-talk.
Studies by her team, for example, have demonstrated that the cytokine Interleukin-6 is a central player in a cycle of tumor growth and cachexia through cross-talk among tumors, fat and muscle.
Other studies have shown that “bone is a repository of certain factors that can induce inflammation and muscle loss,” she said. Among these are a protein produced by bone called RANKL, which may contribute to muscle loss in ovarian cancer, and a protein called lipocalin 2, which may fuel cachexia in pancreatic cancer.
Yet other studies have singled out other likely contributors to cachexia in people with cancer, including malfunctioning mitochondria and the deterioration of nerves that support skeletal muscle fibers.
The preponderance of the evidence suggests, Dr. Guttridge said, that in any given patient with cancer, different players are likely working on their own and in combination to generate the metabolic chaos that underlies cachexia.
Michael Espey, Ph.D., of NCI’s Division of Cancer Treatment and Diagnosis, agreed. Results from many studies “point to the need to consider cancer cachexia as a ‘systems-level’ problem,” Dr. Espey said.
Each new research finding helps researchers better understand the systems fueling cachexia, Dr. Guttridge said.
“We are getting smarter and understanding the role each of these factors [plays] in … cachexia,” he continued. And that might eventually translate into developing therapies that target specific drivers of cachexia in individual patients, he added, along the lines of the many cancer therapies that target specific genetic alterations in patients’ tumors.
A bullseye on GDF15 and GFRAL
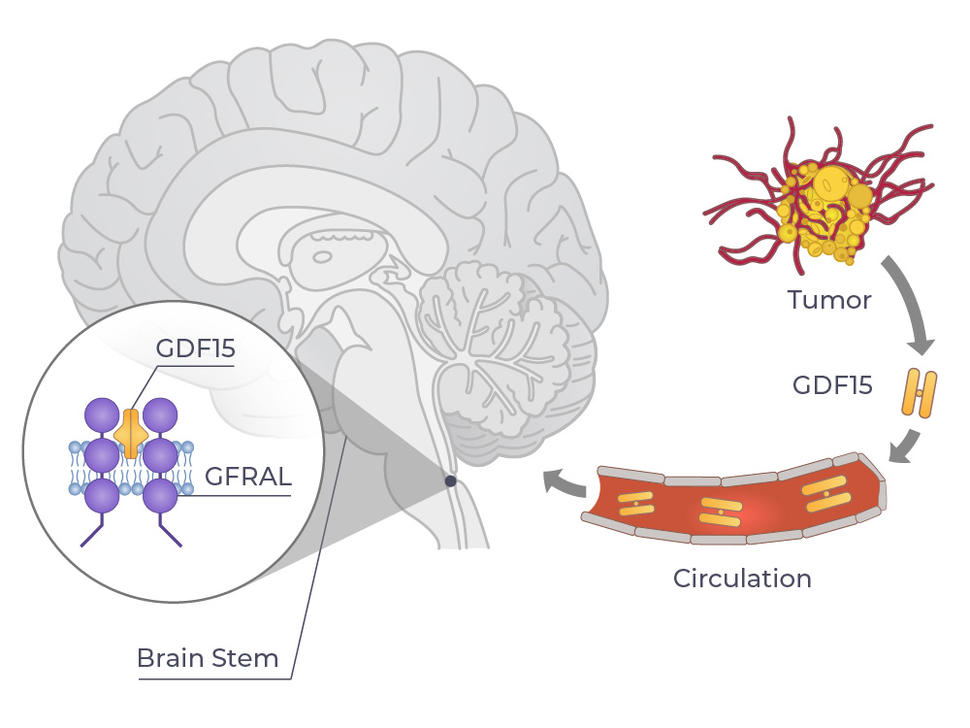
That more precise targeting is already beginning to happen. One potential cachexia driver of great interest to researchers is a cytokine called GDF15.
In the early 2010s, several studies pegged GDF15—which is produced by cells in several different organs, including the liver, kidneys, and intestines—as a direct contributor to a person’s desire to eat. By later in the decade, other research demonstrated how: GDF15 binds to a protein called GFRAL, which is produced by neurons in a part of the brain involved in appetite control.
Then, a study published in July 2020 unleashed a ripple of excitement through the cachexia research community. In mice with cancer cachexia, the study found, an experimental GFRAL-targeted drug could completely reverse wasting.
But there was also a surprising finding: The treatment helped cachectic mice gain weight even if their food intake was minimal. The results, researchers believe, suggest that the communication pathways in cells affected by GDF15 and GFRAL do more than just influence eating behavior.
Within a short period, human clinical trials of drugs that target GDF15 or GFRAL were launched.
One trial involves a drug called NGM120, a version of the GRFAL-targeted agent that showed such remarkable effects in mice. Another, PF-06946860, which blocks GDF15, is also being tested in a small clinical trial of people with advanced forms of several cancers who have signs of anorexia and elevated blood levels of GDF15.
Lessons learned from anamorelin
There are no approved treatments for cancer cachexia in the United States. But in 2021, Japanese health officials approved a drug called anamorelin to treat people with the condition.
Anamorelin works by mimicking the activity of a hormone called ghrelin that is released by cells in the stomach. Like GDF15 and GFRAL, ghrelin modulates appetite, traveling from the stomach to a tiny compartment in the middle of the brain and activating another hormone that stimulates the desire to eat.
Perhaps surprisingly, ghrelin levels don’t appear to be substantially lower in people with cachexia than in otherwise healthy people. Instead, ghrelin’s function seems to be impaired in people with cachexia. So by behaving like a ghrelin impostor, anamorelin is intended to ramp up a person with cachexia’s otherwise suppressed appetite.
In a series of large international clinical trials conducted in the 2010s, treatment with anamorelin helped people with advanced lung cancer and cachexia modestly increase lean body mass (by a median of 2 pounds) and increased their appetite and how much they ate.
From a regulatory perspective, however, there was a problem. The clinical trials were designed to determine whether treatment with anamorelin could meet two primary measures, or endpoints, in patients: an improvement in their lean body mass and an improvement in their hand–grip strength.
Although the drug succeeded on the first, it failed on the second.
Helsinn, the Switzerland-based manufacturer of anamorelin, initially applied for the drug’s approval to treat cachexia in the European Union. But regulators there rejected the application, citing the drug’s failure to improve hand–grip strength and concluding that the increase in lean body mass was not meaningful for patients. It’s unclear whether the company ever sought approval in the United States.
The use of hand–grip strength as a clinical trial endpoint is largely a holdover from earlier cachexia studies, Dr. Zimmers said, in which it served as a proxy for improvements in strength and physical function. But there’s general agreement among clinicians who work with patients that hand–grip strength isn’t a telling measure of improvement in either one.
Other functional measures, such as whether somebody can stand up from a chair, can potentially provide more meaningful information, Dr. Roeland said. But given the outsized impact cachexia has on people’s quality of life, he believes studies should use measures that are based on whether patients say they feel better.
“We have multiple supportive-care drugs that were approved [by FDA] based on what patients tell us,” he said. These include drugs for nausea/vomiting, pain, and shortness of breath, all of which were assessed in clinical trials based on patients’ responses to questionnaires.
Helsinn is currently conducting two new large clinical trials of anamorelin to treat people with advanced lung cancer and cachexia. In both trials, the primary endpoints are weight gain and improvement in anorexia-related symptoms, both as reported by patients.
Tackling cachexia with nutrition and exercise
In addition to the anamorelin trials and those testing the GDF15/GFRAL targeted drugs, other trials are testing combinations of nutrition and/or exercise to treat cachexia.
The only ongoing large trial, called MENAC, is being conducted primarily in Europe, with a few participating hospitals in the United States. Patients in the trial are either getting standard treatment for their cancer or standard treatment plus a home-based exercise regimen, nutritional supplements, and dietary guidance, along with a daily anti-inflammatory drug.
Dr. Dunne led a similar but smaller trial to see if a home-based exercise program of walking and resistance training could improve participants’ daily functioning and quality of life. Initial results from the study won’t be available until late in 2022, he explained. He’s also leading an ongoing trial that added a nutritional supplement to the exercise program.
Results from the different ongoing clinical trials may eventually help improve the treatment of cachexia. In the meantime, clinicians and people experiencing cachexia continue to do their best to deal with it.
One positive sign, Dr. Roy said, is that oncologists appear to be more attuned to the severe problems cachexia can pose for their patients.
He is also affiliated with Northwestern University’s Lurie Comprehensive Cancer Center. As more oncologists at Lurie have learned about what his AbilityLab program offers, Dr. Roy continued, they are referring increasing numbers of patients with cachexia to him.
“I'll tell you that people are excited to have this opportunity,” he said.