Ovarian, Fallopian Tube, and Primary Peritoneal Cancers Prevention (PDQ®)–Patient Version
What Is Prevention?
Cancer prevention is action taken to lower the chance of getting cancer. By preventing cancer, the number of new cases of cancer in a group or population is lowered. Hopefully, this will lower the number of deaths caused by cancer.
To prevent new cancers from starting, scientists look at risk factors and protective factors. Anything that increases your chance of developing cancer is called a cancer risk factor; anything that decreases your chance of developing cancer is called a cancer protective factor.
Some risk factors for cancer can be avoided, but many cannot. For example, both smoking and inheriting certain genes are risk factors for some types of cancer, but only smoking can be avoided. Regular exercise and a healthy diet may be protective factors for some types of cancer. Avoiding risk factors and increasing protective factors may lower your risk, but it does not mean that you will not get cancer.
Different ways to prevent cancer are being studied, including:
- Changing lifestyle or eating habits.
- Avoiding things known to cause cancer.
- Taking medicines to treat a precancerous condition or to keep cancer from starting.
General Information About Ovarian, Fallopian Tube, and Primary Peritoneal Cancers
Key Points
- Ovarian, fallopian tube, and primary peritoneal cancers are diseases in which malignant (cancer) cells form in the ovaries, fallopian tubes, or peritoneum.
- Ovarian cancer is the leading cause of death from cancer of the female reproductive system.
Ovarian, fallopian tube, and primary peritoneal cancers are diseases in which malignant (cancer) cells form in the ovaries, fallopian tubes, or peritoneum.
The ovaries are a pair of organs in the female reproductive system. They are in the pelvis, one on each side of the uterus (the hollow, pear-shaped organ where a fetus grows). Each ovary is about the size and shape of an almond. The ovaries make eggs and female hormones (chemicals that control the way certain cells or organs work in the body).
The fallopian tubes are a pair of long, slender tubes, one on each side of the uterus. Eggs pass from the ovaries, through the fallopian tubes, to the uterus. Cancer sometimes begins at the end of the fallopian tube near the ovary and spreads to the ovary.
The peritoneum is the tissue that lines the abdominal wall and covers organs in the abdomen. Primary peritoneal cancer is cancer that forms in the peritoneum and has not spread there from another part of the body. Cancer sometimes begins in the peritoneum and spreads to the ovary.
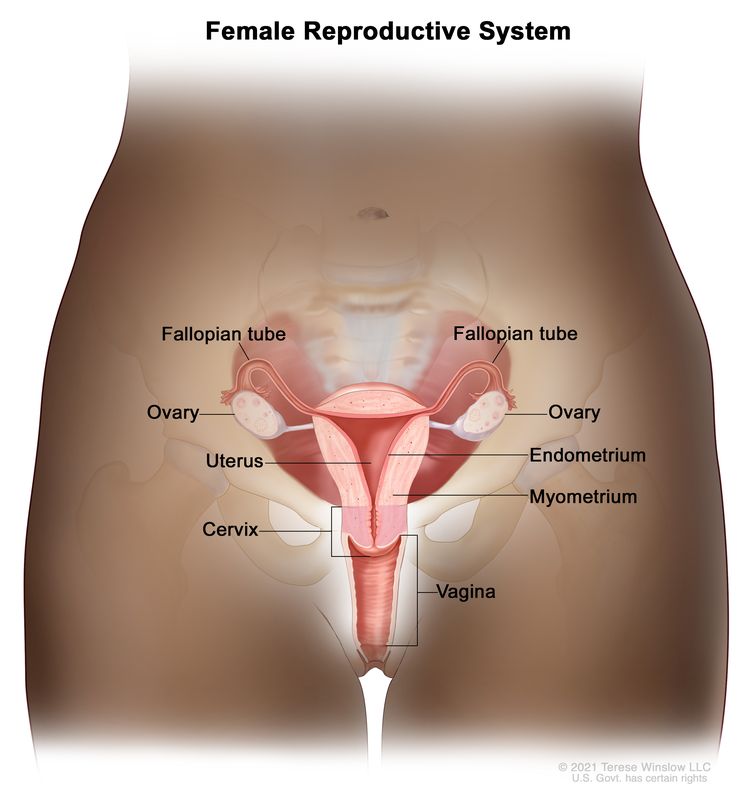
Ovarian cancer is the leading cause of death from cancer of the female reproductive system.
Ovarian cancer is most common in postmenopausal women. From 2012 to 2021, the number of new cases of ovarian cancer decreased slightly each year. There was also a slight decrease in the number of deaths from ovarian cancer each year from 2013 to 2022.
Women who have a family history of ovarian cancer and/or have inherited certain changes in genes, such as the BRCA1 or BRCA2 gene, have a higher risk than those who do not have a family history or who have not inherited these gene changes. For women with inherited risk, genetic counseling and genetic testing can be used to find out more about their likelihood of developing ovarian cancer.
It is hard to find ovarian cancer early. Early ovarian cancer may not cause any symptoms. When symptoms do appear, ovarian cancer is often advanced.
Other PDQ summaries containing information related to ovarian, fallopian tube, and primary peritoneal cancers include:
Ovarian, Fallopian Tube, and Primary Peritoneal Cancers Prevention
Key Points
- Avoiding risk factors and increasing protective factors may help prevent cancer.
- The following are risk factors for ovarian, fallopian tube, and primary peritoneal cancers:
- Family history of ovarian, fallopian tube, and primary peritoneal cancers
- Inherited risk
- Hormone replacement therapy
- Weight and height
- Endometriosis
- The following are protective factors for ovarian, fallopian tube, and primary peritoneal cancers:
- Oral contraceptives
- Tubal ligation
- Giving birth
- Salpingectomy
- Breastfeeding
- Risk-reducing salpingo-oophorectomy
- It is not clear whether the following affect the risk of ovarian, fallopian tube, and primary peritoneal cancers:
- Diet
- Alcohol
- Aspirin and nonsteroidal anti-inflammatory drugs
- Talc
- Infertility treatment
- Cancer prevention clinical trials are used to study ways to prevent cancer.
- New ways to prevent ovarian, fallopian tube, and primary peritoneal cancers are being studied in clinical trials.
Avoiding risk factors and increasing protective factors may help prevent cancer.
Avoiding cancer risk factors may help prevent certain cancers. Risk factors include smoking, having overweight, and not getting enough exercise. Increasing protective factors such as quitting smoking and exercising may also help prevent some cancers. Talk to your doctor or other health care professional about how you might lower your risk of cancer.
The following are risk factors for ovarian, fallopian tube, and primary peritoneal cancers:
Family history of ovarian, fallopian tube, and primary peritoneal cancers
A woman whose mother or sister had ovarian cancer has an increased risk of ovarian cancer. A woman with two or more relatives with ovarian cancer also has an increased risk of ovarian cancer.
Inherited risk
The risk of ovarian cancer is increased in women who have inherited certain changes in genes, such as the BRCA1 or BRCA2 gene.
The risk of ovarian cancer is also increased in women who have certain inherited syndromes, such as:
- Familial site-specific ovarian cancer syndrome.
- Familial breast/ovarian cancer syndrome.
- Hereditary nonpolyposis colorectal cancer (HNPCC; also called Lynch syndrome).
Hormone replacement therapy
There is a slightly increased risk of ovarian cancer in women who are taking hormone replacement therapy (HRT) after menopause. There is also an increased risk of ovarian cancer in women who have recently used HRT, even if they used it for less than 5 years. The risk of ovarian cancer is the same for HRT with estrogen only or with combined estrogen and progestin. HRT taken through the skin may have a lower risk than HRT taken by mouth. When HRT is stopped, the risk of ovarian cancer decreases over time. The risk of ovarian cancer is not affected by the age of the woman when taking HRT.
Weight and height
Having excess body weight or obesity is linked to an increased risk of ovarian cancer. Obesity is also linked to an increased risk of death from ovarian cancer. Being tall may also be linked to a slight increase in the risk of ovarian cancer.
Endometriosis
Women who have endometriosis have an increased risk of ovarian cancer.
The following are protective factors for ovarian, fallopian tube, and primary peritoneal cancers:
Oral contraceptives
Taking oral contraceptives ("the pill") lowers the risk of ovarian cancer. The longer oral contraceptives are used, the lower the risk may be. The decrease in risk may last up to 30 years after a woman has stopped taking oral contraceptives.
Taking oral contraceptives increases the risk of blood clots. This risk is higher in women who also smoke.
Tubal ligation
The risk of ovarian cancer is decreased in women who have a tubal ligation (surgery to close both fallopian tubes).
Giving birth
Women who have given birth have a decreased risk of ovarian cancer compared to women who have not given birth. Giving birth to more than one child further decreases the risk of ovarian cancer.
Salpingectomy
Some studies have shown that salpingectomy (surgery to remove one or both fallopian tubes) is linked to a decreased risk of ovarian cancer. When both fallopian tubes are removed, the risk of ovarian cancer is lowered more than when one fallopian tube is removed.
Breastfeeding
Breastfeeding is linked to a decreased risk of ovarian cancer. The longer a woman breastfeeds, the lower her risk of ovarian cancer. Women who breastfeed for at least 8 to 10 months have the greatest decrease in risk of ovarian cancer.
Risk-reducing salpingo-oophorectomy
Some women who have a high risk of ovarian cancer may choose to have a risk-reducing salpingo-oophorectomy (surgery to remove the fallopian tubes and ovaries when there are no signs of cancer). This includes women who have inherited certain changes in the BRCA1 or BRCA2 gene or have an inherited syndrome.
It is very important to have a cancer risk assessment and counseling before making this decision. These and other factors may be discussed:
- Infertility.
- Early menopause: The drop in estrogen levels caused by removing the ovaries can cause early menopause. Symptoms of menopause can worsen and include: These symptoms may not be the same in all women. Hormone replacement therapy (HRT) may be used to lessen these symptoms.
- Hot flashes.
- Night sweats.
- Trouble sleeping.
- Mood changes.
- Decreased sex drive.
- Heart disease.
- Vaginal dryness.
- Frequent urination.
- Osteoporosis (decreased bone density).
- Risk of ovarian cancer in the peritoneum: Women who have had a risk-reducing salpingo-oophorectomy continue to have a small risk of ovarian cancer in the peritoneum (thin layer of tissue that lines the inside of the abdomen). This is rare, but may occur if ovarian cancer cells had already spread to the peritoneum before the surgery or if some ovarian tissue remains after surgery.
It is not clear whether the following affect the risk of ovarian, fallopian tube, and primary peritoneal cancers:
Diet
Studies of dietary factors have not found a strong link to ovarian cancer.
Alcohol
Studies have not shown a link between drinking alcohol and the risk of ovarian cancer.
Aspirin and nonsteroidal anti-inflammatory drugs
Some studies of aspirin and nonsteroidal anti-inflammatory drugs (NSAIDs) have found a decreased risk of ovarian cancer and others have not.
Talc
Studies of women who used talcum powder (talc) dusted on the perineum (the area between the vagina and the anus) have not found clear evidence of an increased risk of ovarian cancer.
Infertility treatment
Overall, studies in women using fertility drugs have not found clear evidence of an increased risk of ovarian cancer. The risk of invasive ovarian cancer may be higher in women who do not get pregnant after taking fertility drugs.
Cancer prevention clinical trials are used to study ways to prevent cancer.
Cancer prevention clinical trials are used to study ways to lower the risk of developing certain types of cancer. Some cancer prevention trials include healthy people who may or may not have an increased risk of cancer. Other prevention trials include people who have had cancer and are trying to prevent recurrence or a second cancer.
The purpose of some cancer prevention clinical trials is to find out whether actions people take can prevent cancer. These may include eating fruits and vegetables, exercising, quitting smoking, or taking certain medicines, vitamins, minerals, or food supplements.
New ways to prevent ovarian, fallopian tube, and primary peritoneal cancers are being studied in clinical trials.
Information about clinical trials supported by NCI can be found on NCI’s clinical trials search webpage. Clinical trials supported by other organizations can be found on the ClinicalTrials.gov website.
About This PDQ Summary
About PDQ
Physician Data Query (PDQ) is the National Cancer Institute's (NCI's) comprehensive cancer information database. The PDQ database contains summaries of the latest published information on cancer prevention, detection, genetics, treatment, supportive care, and complementary and alternative medicine. Most summaries come in two versions. The health professional versions have detailed information written in technical language. The patient versions are written in easy-to-understand, nontechnical language. Both versions have cancer information that is accurate and up to date and most versions are also available in Spanish.
PDQ is a service of the NCI. The NCI is part of the National Institutes of Health (NIH). NIH is the federal government’s center of biomedical research. The PDQ summaries are based on an independent review of the medical literature. They are not policy statements of the NCI or the NIH.
Purpose of This Summary
This PDQ cancer information summary has current information about ovarian, fallopian tube, and primary peritoneal cancers prevention. It is meant to inform and help patients, families, and caregivers. It does not give formal guidelines or recommendations for making decisions about health care.
Reviewers and Updates
Editorial Boards write the PDQ cancer information summaries and keep them up to date. These Boards are made up of experts in cancer treatment and other specialties related to cancer. The summaries are reviewed regularly and changes are made when there is new information. The date on each summary ("Updated") is the date of the most recent change.
The information in this patient summary was taken from the health professional version, which is reviewed regularly and updated as needed, by the PDQ Screening and Prevention Editorial Board.
Clinical Trial Information
A clinical trial is a study to answer a scientific question, such as whether one treatment is better than another. Trials are based on past studies and what has been learned in the laboratory. Each trial answers certain scientific questions in order to find new and better ways to help cancer patients. During treatment clinical trials, information is collected about the effects of a new treatment and how well it works. If a clinical trial shows that a new treatment is better than one currently being used, the new treatment may become "standard." Patients may want to think about taking part in a clinical trial. Some clinical trials are open only to patients who have not started treatment.
Clinical trials can be found online at NCI's website. For more information, call the Cancer Information Service (CIS), NCI's contact center, at 1-800-4-CANCER (1-800-422-6237).
Permission to Use This Summary
PDQ is a registered trademark. The content of PDQ documents can be used freely as text. It cannot be identified as an NCI PDQ cancer information summary unless the whole summary is shown and it is updated regularly. However, a user would be allowed to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks in the following way: [include excerpt from the summary].”
The best way to cite this PDQ summary is:
PDQ® Screening and Prevention Editorial Board. PDQ Ovarian, Fallopian Tube, and Primary Peritoneal Cancers Prevention. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/ovarian/patient/ovarian-prevention-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389375]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use in the PDQ summaries only. If you want to use an image from a PDQ summary and you are not using the whole summary, you must get permission from the owner. It cannot be given by the National Cancer Institute. Information about using the images in this summary, along with many other images related to cancer can be found in Visuals Online. Visuals Online is a collection of more than 3,000 scientific images.
Disclaimer
The information in these summaries should not be used to make decisions about insurance reimbursement. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s E-mail Us.