Potential New Cancer Therapy Could Target Tumors Two Ways
, by NCI Staff
A team of researchers has developed a potential new therapy that may work in two distinct ways to attack tumors, according to findings from a new study.
The potential therapy targets a protein called TNFR2. This protein is present at elevated levels on many tumor cells as well as on a type of immune cell that can prevent a cancer patient’s immune system from attacking tumors.
Using newly created TNFR2-targeting antibodies they developed, the researchers showed that the antibodies could kill ovarian cancer cells that express TNFR2, as well as the immune cells that suppress the immune response to tumors, called regulatory T cells or T-reg cells.
Importantly, explained the study’s lead investigator, Denise Faustman, M.D., Ph.D., of Massachusetts General Hospital and Harvard Medical School, T-reg cells collected from the tumor site in women with ovarian cancer were more sensitive to the antibodies than T-reg cells from the blood of healthy donors. This is also the first study to identify TNFR2 as an oncogene in ovarian cancer, she added.
"What this means is that we may be able to target the tumor and only the 'bad' host T cells that are suppressing the immune response. We should therefore be able to more completely preserve the overall immune system," Dr. Faustman said.
The team's findings were published January 17 in Science Signaling.
Killing Two Birds with One Stone
Researchers have known for some time that T-reg cells in the tumor microenvironment—the normal cells, molecules, and blood vessels that surround and feed tumor cells—express high levels of TNFR2, Dr. Faustman said. TNFR2 is also present on some other types of immune cells that help tumors evade an immune response.
More recently, Dr. Faustman's team found that ovarian cancer cells aberrantly express TNFR2 and that activation of this receptor promotes tumor cell growth.
Other recent studies have shown aberrant expression of TNFR2 on an array of tumor cell types, “as a presumed oncogene,” she said, including colon and kidney cancer, multiple myeloma, Hodgkin lymphoma, and a type of non-Hodgkin lymphoma.
These findings suggest that TNFR2 could be an important target for cancer therapies, wrote Xin Chen, M.D., Ph.D., and Joost Oppenheim, M.D., of NCI's Center for Cancer Research (CCR) in a commentary on the new study. Blocking TNFR2 "may have the effect of killing two birds with one stone: boosting antitumor immune responses and directly killing tumor cells," they wrote.
More Potent Killing of Tumor Site T-Reg Cells
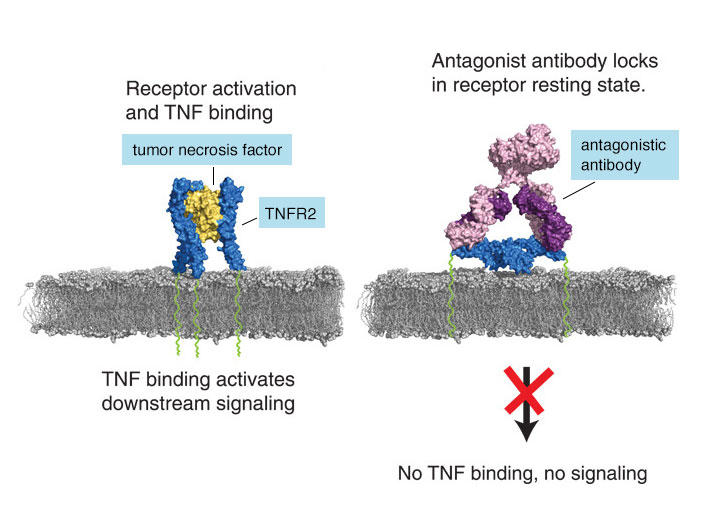
The two TNFR2 antibodies developed by Dr. Faustman's team are "antagonistic" antibodies: they attach to TNFR2 and lock the receptor in an inactive state.
Normally, TNFR2 is activated when it latches on to tumor necrosis factor (TNF), a substance produced by some white blood cells. Activation triggers signals inside the cell that cause cells to divide and increase in number (proliferate).
The researchers found that the antagonistic TNFR2 antibodies eliminated T-reg cells from blood samples of healthy donors by preventing the cells from being activated and proliferating, even in the presence of high concentrations of TNF. The antibodies do not directly kill T-reg cells but rather, the cells die after their proliferation is halted, Dr. Faustman explained.
The team then showed that the TNFR2 antibodies more potently killed T-reg cells isolated from fluid surrounding metastatic ovarian cancer tumor sites than T-reg cells from healthy blood donors. The antagonistic antibodies also directly killed cells from a laboratory-grown ovarian cancer cell line that expresses TNFR2 on the cell surface.
These findings, said Dr. Faustman, suggest that "we can now selectively target the T-reg cells that express TNFR2 and are found predominantly in the tumor microenvironment." And although other members of the TNF receptor family are found on many different cell types in the body, she said, "the beauty of TNFR2 is that it's expressed at high levels in the tumor and the tumor environment but is barely expressed anywhere else in the body."
As a result, antibodies that block TNFR2 may be more specific and less toxic than antibodies that block other TNF receptors or antibodies that act as immune checkpoint inhibitors and are already used to treat cancer.
"For years, people have worked hard to find a way to selectively kill the bad T cells at the tumor site, and this is the first demonstration" that this may be possible, said Dr. Faustman.
Promise as Cancer Therapies
The work by Dr. Faustman and colleagues "clearly shows that TNFR2 antagonists have promise as cancer therapies," wrote Drs. Chen and Oppenheim.
"To achieve an optimal antitumor effect, the TNFR2 antagonist may have to be used in conjunction with other therapeutics," they continued. "TNFR2 antagonists have the capacity to eliminate T-reg activity and thus may further enhance the efficacy of current immunotherapeutics. In addition, suppression of tumor cell survival by the TNFR2 antagonist may improve the outcome of chemotherapy or radiotherapy in cancer patients."
"We're about 2 years away from the clinic," Dr. Faustman said. "Our primary goals are to show the efficacy of these antibodies in fresh human tumor samples and figure out how to design the best clinical trial."