Screening for Many Cancers with One Test: Uncertainty Abounds
, by Philip Castle, Ph.D., M.P.H.
One of the exciting areas of cancer prevention research is the development of noninvasive tests that may have the potential to easily and accurately determine whether and where in the body a person has early-stage cancer. And not just for a single cancer but for many cancers. Over the last several years, remarkable progress has been made on this front.
There are many multi-cancer early detection (MCED) tests in development intended to simultaneously screen for multiple types of cancer in otherwise healthy people. Someday, if MCED tests prove to be clinically beneficial, they may be ordered alongside tests for cholesterol or liver function.
On its face, a test that requires a simple blood draw to tell a symptom-free person whether they may have a cancer and where it is in their body seems like an amazing advance. This is particularly true for cancers like pancreatic, ovarian, and brain cancers, for which no screening tests exist and that typically are not diagnosed until they are advanced, very difficult to treat, and highly lethal.
However, as with much of medicine and public health, it’s not that simple. Despite their tremendous promise, much about MCED tests is still unknown and many unanswered questions remain, such as:
- Who should undergo an MCED test, and which one would be best for you?
- What happens if subsequent testing doesn’t find cancer in a person who tested positive on an MCED? What is their future risk of cancer?
- Will people who receive an MCED test then forgo proven cancer screening tests in the future?
- Is there the potential for MCED tests to exacerbate racial, ethnic, and socioeconomic disparities in cancer outcomes?
- Do they work equally well for everyone?
And then there is the most important question: Do these tests actually reduce deaths from cancer, which is the ultimate goal of cancer screening? Finding cancer is not enough if a person’s chance of dying from that cancer does not change.
Simply put, there is still a substantial level of uncertainty and many unknowns surrounding these tests. And it’s clear that more robust research on MCED tests is needed to understand how best to maximize their benefits and minimize their potential harms.
As the leader of the national cancer program, NCI is one of the few institutions that can carry out the large-scale studies needed to determine MCED tests’ potential role in improving public health.
How do MCED tests work? Do they work well?
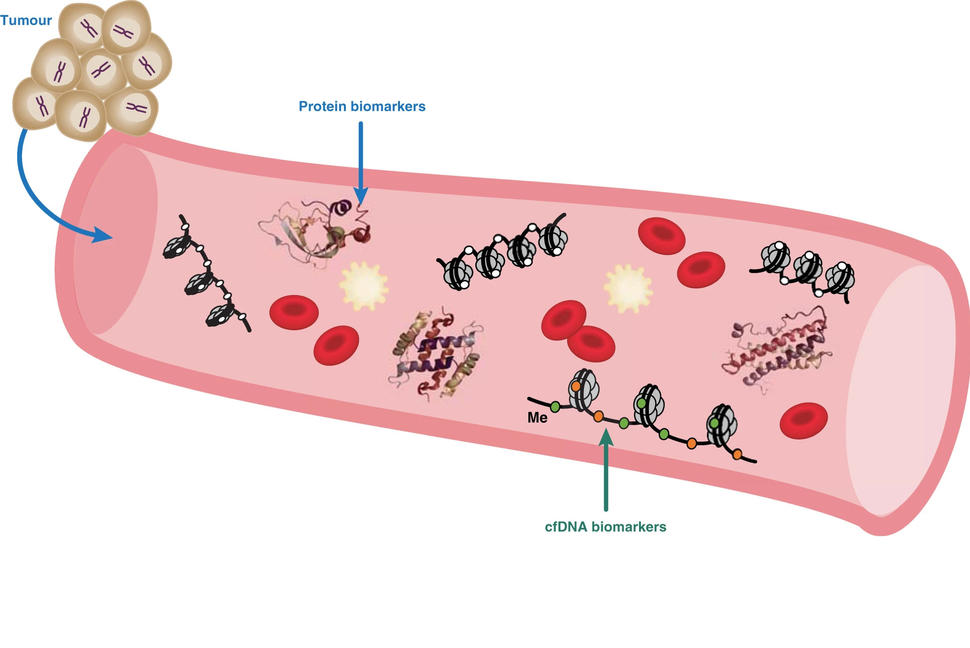
MCED tests are a type of test known as a liquid biopsy. Liquid biopsies currently cleared by the Food and Drug Administration (FDA) are used in people who have already been diagnosed with cancer.
In people with cancer, FDA-cleared liquid biopsies identify specific biological signals in DNA, RNA, or proteins released by cancer cells into the blood. That information can then guide what specific, targeted cancer therapies or immunotherapies patients should receive.
MCED tests also attempt to look in blood for biological signals, or biomarkers, that are released by cancer cells or induced by their presence. However, MCED tests differ from currently FDA-cleared liquid biopsies in two ways.
First, MCED tests are intended for use as cancer screening test in symptom-free individuals, as opposed to a test in people known to have cancer. And second, MCED tests simultaneously assess multiple signals to determine whether there is a strong likelihood that a person has cancer and, in most cases, identify where in the body that tumor is located.
Companies with MCED tests currently under study claim they can detect anywhere from two or three cancer types up to as many as 50.
With one or two exceptions, MCED studies performed to date used previously collected blood samples from groups of people known to have cancer or not have cancer. And thus far, these limited data show that they can indeed detect multiple types of cancer. However, their ability to find cancer when it is present—known as their sensitivity—varies by test and for each cancer type.
Those same studies have consistently found that MCED tests are better at detecting advanced, or later-stage, cancers than earlier-stage cancers, the latter of which are more easily treated and sometimes cured. They also found that the tests had high specificity, that is, they are highly unlikely to produce a positive result for people without cancer.
More questions than answers
When it comes to screening healthy people to see if they have a disease, it’s extremely important to show that potential benefits of that screening outweigh any potential harms.
As I’ve already said, the most important benefit of a cancer screening test is that it reduces the number of deaths from that cancer when used broadly in the general population. Achieving that aim raises a critical point: the screening test itself is just the beginning, particularly if the test result suggests that cancer is present. Many steps must then occur to confirm that result and, if necessary, treat the cancer.
It’s those postscreening steps where many of the potential harms of screening can arise. Of those potential harms, two strike me as the most significant.
One is a false-positive screening test result that leads to invasive and expensive procedures to ultimately find no cancer is present. The second is the diagnosis of slow-growing cancers that may never cause symptoms, a phenomenon called overdiagnosis. And overdiagnosis, unfortunately, can sometimes lead to overtreatment.
There are also potential societal harms, including the unknown financial impact of widespread MCED test use before it’s known whether they reduce deaths from cancer.
Planning important studies of MCED tests
The bottom line is: We need more research on MCED tests.
That’s why NCI has been working with leading experts at FDA and outside of the government on screening and early detection, diagnostic testing, and other relevant fields, to identify the priority areas of need.
That analysis has led to the identification of several research priorities.
For example, more studies on the underlying basic and clinical science of MCED tests are needed. That includes studies of how MCED tests perform for specific cancers and in different patient populations, and the biology and natural history of the biomarkers used in these tests.
We also need to be able to validate the performance of the MCED tests under development. Along those lines, NCI is supporting a project led by the Alliance for Clinical Trials in Oncology to collect blood samples from people with and without cancer. Those samples will be used to independently confirm the performance of the MCED test(s) being considered for use in clinical trials.
Along those lines, we appreciate the need to perform large clinical trials to help answer some of the most pressing questions about MCED tests. Designing such trials, however, is a difficult and complex undertaking.
That’s why NCI sponsored an October 2021 workshop to get feedback from research leaders on how such trials should be designed including: what should be the primary measures (or endpoints), how to evaluate multiple MCED tests at the same time, the diagnostic work-up of positive results, and the care of those diagnosed with cancer. NCI is now using that feedback to guide the design of randomized clinical trials of MCED tests.
Researchers in the United Kingdom recently launched a large trial of a single MCED test. There is no immediate timeline for launching a large NCI-supported trial in the United States, although our goal is to launch one that includes multiple MCED tests in the next 18 to 24 months.
Yes, MCED tests hold tremendous promise. And it might be frustrating to some to hear that, despite their promise, we should take the time to learn more about them before they are widely used.
But that is a responsibility and role that NCI is committed to fulfilling. In doing so, we can help to ensure that the years of research and innovation that led to the development of MCED tests can save as many lives as possible.