Basic and Translational Research
Researchers with the OHSU CSBC Center determined mechanisms of treatment-resistance related to cell plasticity in triple-negative breast cancer (TNBC) using systems approaches. Additionally, the computational approaches that they have developed for their CSBC research are being used in the Serial Measurements of Molecular and Architectural Responses to Therapy (SMMART) program to guide the treatment of patients with TNBC.
By integrating computational modeling and metabolomic profiling, Dr. Stacey Finley et al. with the USC CSBC revealed insights into the metabolic interactions between colorectal cancer cells and cancer-associated fibroblasts. Their systems biology approach can be used to preclinically develop cancer therapy strategies.
Using mathematical and experimental models, Dr. Peter Sorger et al. revealed mechanisms of adaptive rewiring that contribute to drug resistance in mutant BRAF melanoma. These findings could help enable the development of strategies to overcome tumor resistance to therapeutic inhibitors.
Technology Development
By combining single-cell transcriptomics with dynamic cell mass measurements, Dr. Scott Manalis et al. with the MIT CSBC Center developed a new tool to determine therapeutic susceptibility in multiple myeloma cells. This approach is currently being tested in the clinic to identify cells that underlie minimal residual disease in leukemia and used in Travera’s 2-day Rapid Therapy Guidance test.
Investigators at the Yale CSBC Center developed a method that enables studies of cell migration at the single-cell level. Using this platform, they identified a migratory phenotype in glioblastoma cells that predicts patient outcomes.
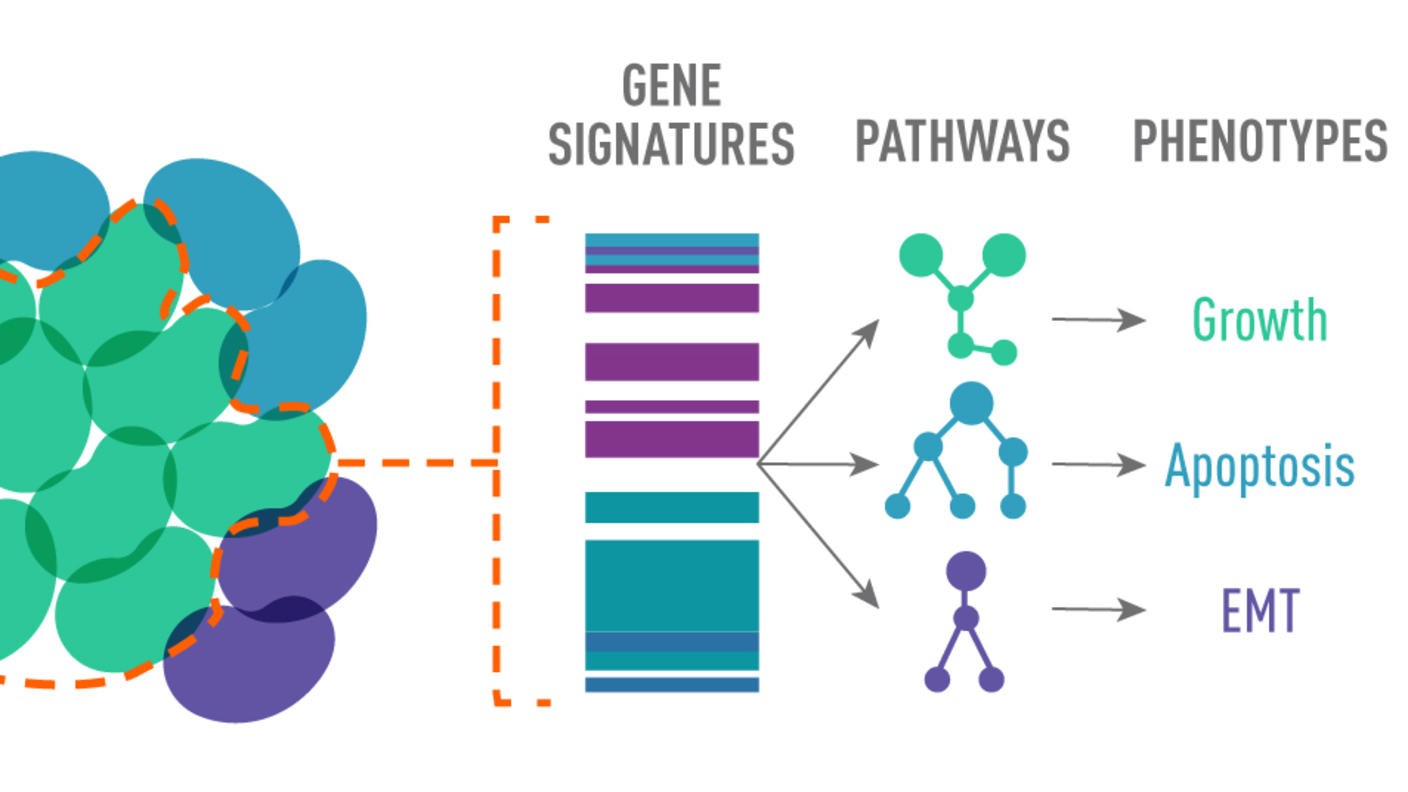
Dr. Andrea Califano et al. at the Columbia CSBC Center developed publicly available software for identifying and targeting master regulators across tumor types. These computational tools have been licensed to DarwinHealth to facilitate their use for precision medicine by the pharmaceutical industry.
Mapping Cell States and Networks
Researchers with the Stanford CSBC Center developed a machine learning framework, which was used to generate an atlas of cell states and ecosystems across tumors. They further showed that different tumor ecosystems have specific biological and clinical responses.
Using sequencing approaches, Dr. Christina Leslie et al. with the MSKCC CSBC Center generated an atlas of T cell dysfunctional states in cancer and infection. This resource can be used to enable studies of immunity and cancer.
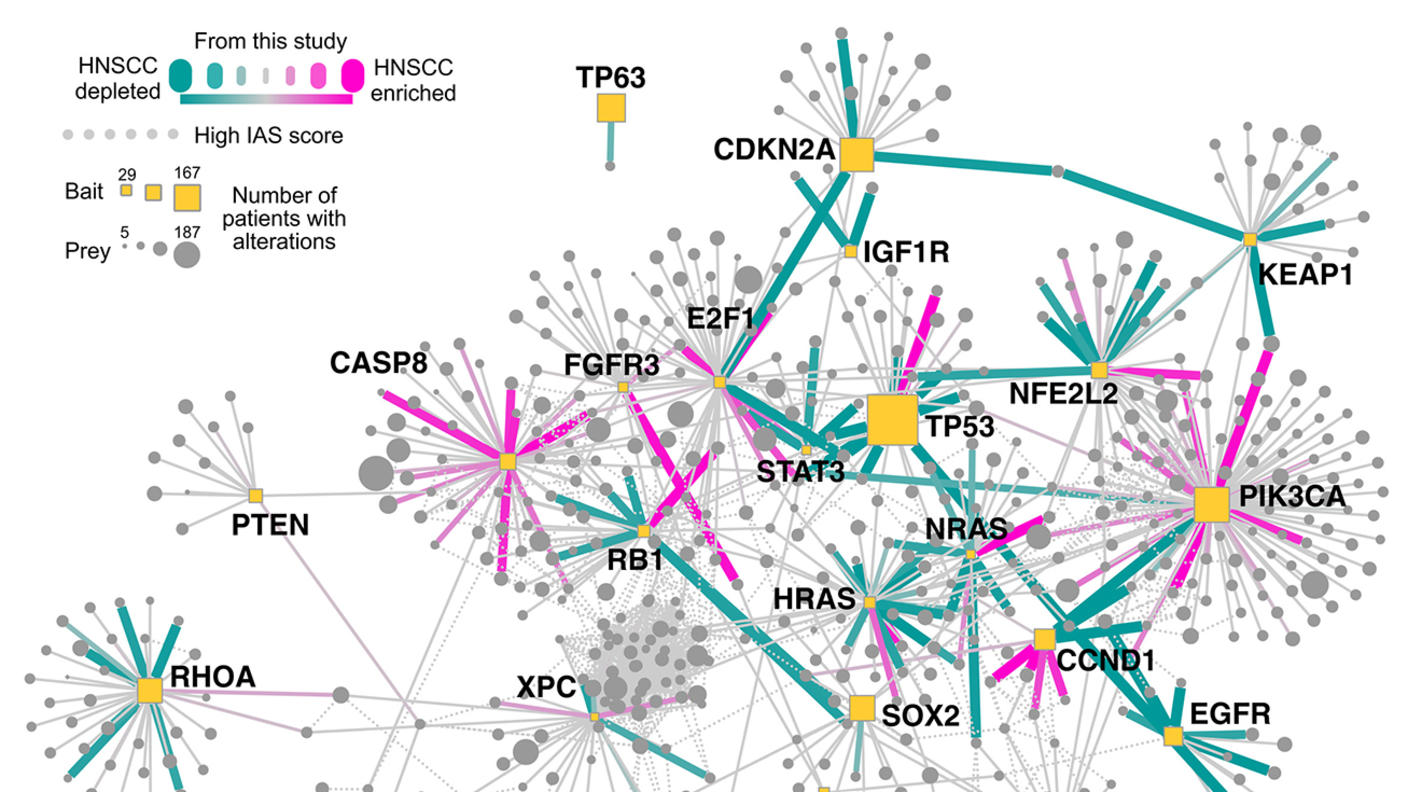
Researchers with the St. Jude Children’s Research Hospital CSBC Project revealed pharmacotypes (i.e., variability in drug sensitivity) across the genomic landscape of pediatric acute lymphoblastic leukemia. The findings also provided new insights into the pharmacological basis of variability in treatment outcomes for a childhood cancer.