Trial Confirms CAR T-Cell Therapy Benefits People with Aggressive Lymphomas
, by Sharon Reynolds
CAR T-cell therapy, a type of personalized immunotherapy, can help cure some people with aggressive non-Hodgkin lymphoma (NHL). That’s according to updated results from a large randomized phase 3 clinical trial of the CAR T-cell therapy axicabtagene ciloleucel (Yescarta).
Most people diagnosed with diffuse large B-cell lymphoma, the most common form of aggressive NHL, will be cured by initial treatment with chemotherapy. But for those who aren’t, the prospects of a cure have been uncertain at best, even with additional grueling chemotherapy and stem cell transplant.
Now, new data from the ZUMA-7 clinical trial strongly suggests that axi-cel, as this CAR T-cell therapy is often called, can offer real hope for this latter group of patients.
The study investigators presented their findings June 5 at the American Society of Clinical Oncology (ASCO) annual meeting. They estimate that about 55% of patients would still be alive 4 years after receiving axi-cel, compared with 46% of those who initially received the standard treatment for relapsed disease.
“This is the first trial in nearly 30 years to significantly improve overall survival for patients with large B-cell lymphoma that is not responding to initial treatment or has relapsed,” said Dr. Jason Westin, M.D., of the University of Texas MD Anderson Cancer Center, who helped lead the study.
And with patients being followed for almost 4 years, those still alive have likely been cured, he added.
Based on these results, axi-cel is now the preferred treatment for people whose diffuse large B-cell lymphoma has recurred quickly or proven resistant to standard initial treatment, explained Christopher Melani, M.D., of NCI’s Center for Cancer Research. And for future studies, axi-cel “is now the benchmark upon which to improve with future treatments,” Dr. Melani explained.
A clear improvement in survival
Until recently, the only treatment with the possibility of eliminating recurrent or treatment-resistant diffuse large B-cell lymphoma was more high-dose chemotherapy, followed by an autologous stem cell transplant. This is where a patient’s healthy blood-forming stem cells are collected from the blood or bone marrow before treatment and then given back to the patient after high-dose intensive chemotherapy.
However, only about half of patients are healthy enough to endure this treatment. And less than a quarter of those who can tolerate it will be cured.
This landscape began to change in 2021, when the results of two large clinical trials showed that CAR T cells could improve the time that people with treatment-resistant or recurrent large B-cell lymphoma lived without their cancer getting worse.
Earlier findings from the ZUMA-7 trial, which began recruiting patients in 2018, led the Food and Drug Administration (FDA) to approve axi-cel in 2022 for adults with diffuse large B-cell lymphoma that wasn’t eliminated by their initial treatment or that returned within 12 months of initial chemotherapy. That approval was based on improvements in a measure called event-free survival, which includes the length of time it takes after treatment for the NHL to begin getting worse. But whether axi-cell would also improve how long patients lived overall wasn’t yet clear.
ZUMA-7 enrolled people deemed healthy enough to undergo additional high-dose chemotherapy and, potentially, an autologous stem cell transplant. This combination is currently considered the standard of care for treatment-resistant or relapsed diffuse large B-cell lymphoma.
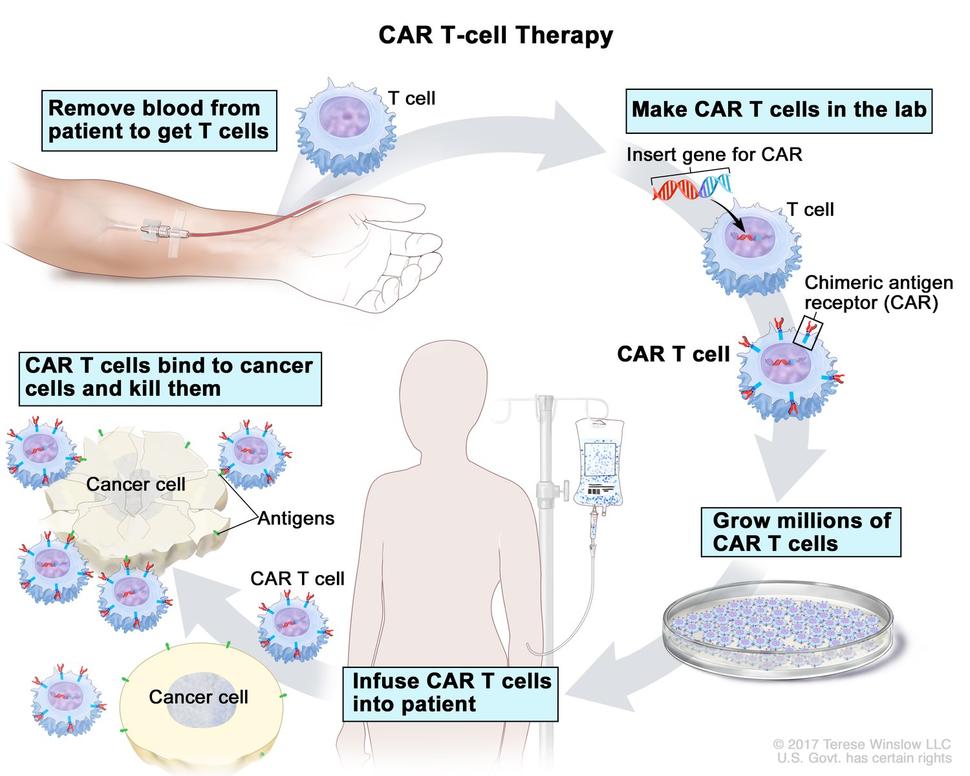
Like all currently approved CAR T-cell therapies, axi-cel is truly personalized, in that it uses a patient’s own immune cells to create the treatment.
In the ZUMA-7 trial, which was funded by Kite Pharma, axi-cel’s manufacturer, researchers randomly assigned 180 people to receive axi-cel and 179 people to receive standard treatment. Differences between the groups emerged quickly. While 94% of people in the axi-cel group received their CAR T cells, only 36% of those in the standard-treatment group were able to have a stem cell transplant because in most cases their disease continued to progress despite chemotherapy, Dr. Westin explained at the ASCO meeting.
Because of this rapid disease progression, 57% of the participants in the standard-treatment group went on to receive CAR T-cell therapy (in most cases with axi-cel) after their disease got worse.
The substantial improvement in how long people in the axi-cel group lived held up even though so many in the standard treatment group rapidly switched to CAR T-cell therapy, Dr. Westin stressed.
Axi-cel was originally approved by the FDA in 2017 as a third or later-line treatment, meaning it could be given only to people whose disease continued to get worse or came back after two or more forms of therapy, explained Dr. Melani. But the ZUMA-7 findings indicate that waiting that long to use axi-cel is no longer the best treatment approach. “Patients who got chemotherapy first showed worse survival, even when most crossed over to receive CAR T cells,” he said. “This study puts axi-cel as the preferred second-line option.”
CAR T-cell therapies, including axi-cel, can have serious long-term side effects that are different from those seen after chemotherapy. In the short term, these include a dangerous immune system reaction called cytokine release syndrome, neurological effects, and the reactivation of dormant viruses in the body. In the long term, they can include immunosuppression, which may in turn lead to second cancers.
At the time of the most recent data analysis, 82 patients in the axi-cel group had died, compared with 95 in the standard care group. Seven people in the axi-cel group died of side effects related to treatment, compared with two in the standard care group. However, since the analysis of the ZUMA-7 data that led to the most recent FDA approval, no new deaths related to treatment with axi-cel had occurred.
Issues of equity and access
Even as CAR T-cell therapy has proven to be effective for more and more people with blood cancers like NHL, lack of access has plagued this treatment option.
“That’s kind of the kicker [for CAR T-cell therapy],” Dr. Melani said. “Not everyone is going to be able to get it.”
One of the biggest issues around CAR T-cell therapies is the cost. Between their production and the extensive hospitalization and monitoring required, “this [treatment] can cost upwards of half a million dollars or more,” Dr. Melani explained.
“We must bring the costs [down] to … affordable levels,” said Asher Chanan-Khan, M.D., from the Mayo Clinic Cancer Center, speaking at the ASCO meeting. Some university hospitals have been producing CAR T cells in-house, explained Dr. Melani, which could potentially cut the price down substantially.
But there are also other aspects of CAR T-cell therapy that affect equitable access to this treatment, explained Leyla Shune, M.D., from the University of Kansas Cancer Center, who discussed issues related to equity and CAR T-cell therapy at the ASCO meeting.
Managing the unique—and often dangerous—side effects of CAR T-cell therapy requires extensive training and practice. So CAR T-cell therapies can currently only be given at certain specialty centers—which can often be hundreds of miles away from many patients’ homes, Dr. Shune explained. And because of the chemotherapy given before receiving CAR T-cell therapies and the recovery time required after receiving it, patients may need to take weeks or months off work.
“I live in a state where we only have two CAR T centers, and my patients have to travel [up to] 8 hours to get to me,” said Dr. Shune. In the future, to provide more equitable access to CAR T-cell treatment, patient assistance programs may need to include services like travel assistance and subsidized lodging, she explained.
Other options in the pipeline
For now, CAR T cells look to be a game changer for many people with relapsed or resistant diffuse large B-cell lymphoma. But they’re not the only game in town.
Clinical trials are also testing another type of immunotherapy called bispecific antibodies, or BiTEs, in people with relapsed or treatment-resistant diffuse large B-cell lymphoma, explained Dr. Melani. These drugs simultaneously attach to immune cells and cancer cells, enabling the immune cells to easily find and destroy the cancer cells by bringing them closer together.
Two BiTEs, glofitamab (Columvi) and epcoritamab (Epkinly), have recently received accelerated approval as third or later-line treatments for diffuse large B-cell lymphoma, and are being tested as earlier treatments in other trials.
Unlike CAR T-cell therapies, BiTEs are “off-the-shelf” drugs, meaning they don’t have to be manufactured for each patient. That feature will likely make them more accessible to patients because they can be given at smaller hospitals, where most people with cancer are treated, Dr. Melani explained.
Other clinical trials, such as the ViPOR and ViPOR-P studies being conducted at the NIH Clinical Center, are testing cocktails of targeted drugs selected to hit known vulnerabilities in lymphoma cells.
“It’s an exciting time in [treating] aggressive lymphoma, with lots of newer therapies that are helping patients live longer,” said Dr. Melani.