Abemaciclib Approval Expands Initial Treatment Options for Advanced Breast Cancer
, by NCI Staff
The Food and Drug Administration (FDA) has expanded the approved uses for the drug abemaciclib (Verzenio) in women with breast cancer.
On February 26, the agency approved the targeted therapy as an initial, or first-line, treatment in postmenopausal women with advanced or metastatic breast cancer whose tumors are hormone receptor (HR)-positive and HER2-negative.
Under the approval, abemaciclib must be used in combination with an aromatase inhibitor, a type of hormone therapy.
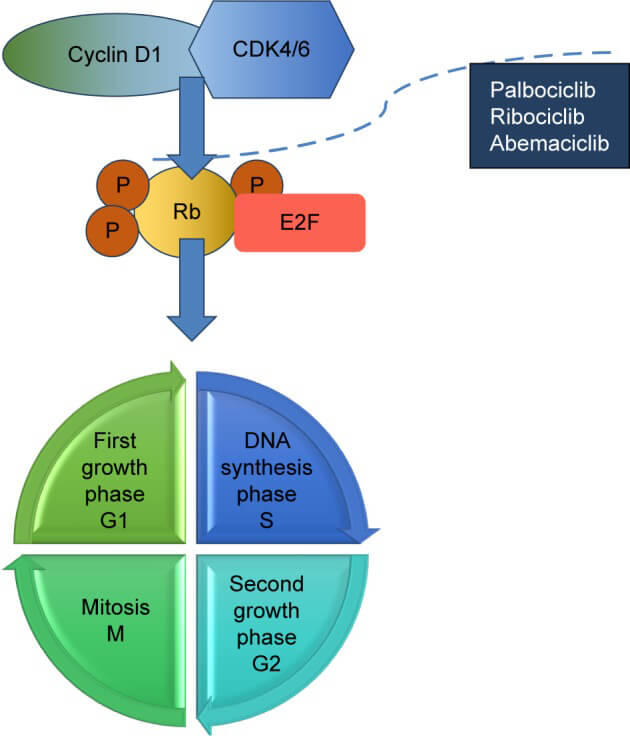
Abemaciclib is one of a class of targeted cancer therapies that block the activity of CDK4 and CDK6 proteins (CDK4/6). These proteins can serve as dual threats, fueling the progression of breast tumors and helping to make them resistant to commonly used treatments like aromatase inhibitors.
Two other CDK4/6 inhibitors, ribociclib (Kisqali) and palbociclib (Ibrance), were previously approved as first-line treatments in postmenopausal women with HR-positive, HER2-negative metastatic breast cancer.
In the phase 3 clinical trial that led to the approval of abemaciclib, called MONARCH 3, the drug substantially increased how long women lived without their disease progressing (progression-free survival).
“The magnitude of the effect, when compared with the use of an aromatase inhibitor alone, is similar to the effect observed with the previously approved CDK4/6 inhibitors,” explained Alexandra Zimmer, M.D., of the Women’s Malignancies Branch in NCI’s Center for Cancer Research.
The approval, Dr. Zimmer continued, “makes abemaciclib another option for first-line use in these patients.”
Improved Progression-Free Survival, but Some Side Effects
FDA initially approved abemaciclib in October 2017 for women with HR-positive, HER2-negative advanced breast cancer whose disease had returned after treatment with aromatase inhibitors or other hormone therapies.
MONARCH 3 enrolled nearly 500 postmenopausal women with HR-positive, HER2-negative advanced breast cancer who had not previously received treatment for advanced cancer. Trial participants were randomly assigned to receive abemaciclib or a placebo, together with one of two aromatase inhibitors, anastrozole (Arimidex) or letrozole (Femara). The trial was funded by Eli Lilly, which manufactures abemaciclib.
In addition to experiencing improved progression-free survival, more women treated with abemaciclib and an aromatase inhibitor experienced at least some reduction in the size of their tumors than women treated only with an aromatase inhibitor: 59% versus 44%.
Approximately 20% of women in the trial treated with abemaciclib had to stop treatment because of side effects, and more than 40% had to have the dose of the drug reduced.
As with other studies involving abemaciclib, the most frequent serious side effect for those receiving the drug was diarrhea, which occurred in approximately 80% of patients. This side effect was usually manageable, the trial leaders reported, with most cases of diarrhea treated with commonly used medications and/or reductions in the abemaciclib dose.
Low-grade fatigue and nausea were also common in women treated with abemaciclib.
Which CDK4/6 Inhibitor for Which Patient?
The approved CDK4/6 inhibitors “have changed the way we manage hormone receptor-positive breast cancer,” said Andres Forero, M.D., who directs the Breast Cancer Program at the University of Alabama at Birmingham School of Medicine.
A big challenge for oncologists at this point, Dr. Forero continued, is deciding which CDK4/6 inhibitor is the best option for a given patient. “That’s the million-dollar question,” he said.
Despite sharing the same molecular target in cancer cells, each of the three drugs has different side effects, Dr. Zimmer said. And those differences may help to identify which patients might be best off receiving which drug.
For example, serious diarrhea is more common with abemaciclib than either palbociclib or ribociclib, she noted. Meanwhile, a substantial drop in white blood cells (neutropenia) is more common in patients treated with palbociclib, and patients treated with ribociclib are more likely to experience a type of change in heart rhythm called QT prolongation.
Dr. Forero agreed that other health issues that could be exacerbated by each respective drug can help to drive treatment decisions for individual patients.
What’s Next for CDK4/6 Inhibitors?
Among the larger questions now, Dr. Zimmer said, is whether women being treated with a CDK4/6 inhibitor whose cancer begins to progress should continue the treatment along with the addition of another therapy.
Another question is whether CDK4/6 inhibitors might improve outcomes in women with early-stage cancer. All three FDA-approved CDK4/6 inhibitors, in fact, are being tested in large phase 3 clinical trials of women with early-stage HR-positive, HER2-negative breast cancer, both as a presurgical (neoadjuvant) and a post-surgical (adjuvant) treatment.
Other trials are also testing CDK4/6 inhibitors in women with HER2-positive breast cancer, using the drugs in combination with HER2-targeted therapies like trastuzumab (Herceptin) and lapatinib (Tykerb), Dr. Forero noted.
“I think the next big hit for these drugs will be in HER2-positive cancers,” he said.