Nivolumab and Relatlimab Combination Shows Promise in Advanced Melanoma
, by Linda Wang
People diagnosed with advanced melanoma may soon have a new treatment option that combines two immunotherapy drugs. In a large clinical trial, people with advanced melanoma treated with nivolumab (Opdivo) and a new drug called relatlimab—which targets a protein called LAG-3—lived longer without their cancer getting worse than those treated only with nivolumab.
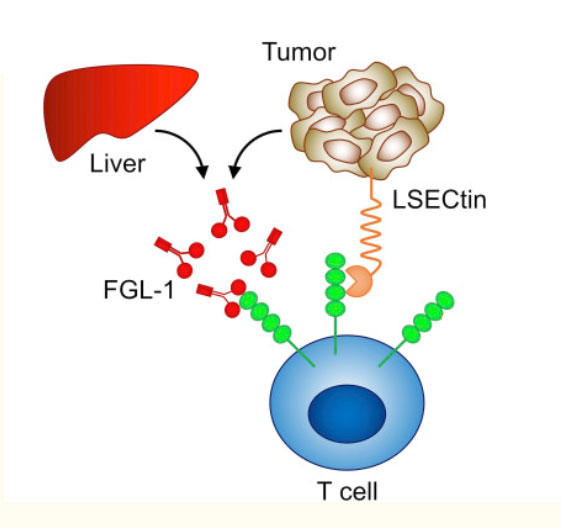
Both nivolumab and relatlimab are immune checkpoint inhibitors. By blocking immune checkpoint proteins, which generally keep the immune responses from being too strong, immune checkpoint inhibitors restore the natural ability of T cells to attack cancer cells.
Nivolumab blocks an immune checkpoint protein on T cells called PD-1 and is already broadly used to treat melanoma and many other cancer types. Other drugs that target LAG-3 have been developed, but relatlimab is the furthest along in clinical research. However, it has not yet been approved by the Food and Drug Administration for use outside of clinical trials.
The trial, called RELATIVITY-047, is the first large clinical study to show that targeting both LAG-3 and PD-1 can be an effective approach for treating patients with advanced melanoma, said the study’s lead investigator, Evan J. Lipson, M.D., of the Johns Hopkins Sidney Kimmel Comprehensive Cancer Center. He presented the findings on June 6 at the virtual annual meeting of the American Society of Clinical Oncology (ASCO). The study results were published January 6, 2022, by the New England Journal of Medicine.
“This is proof of principle that when you add other immune checkpoint inhibitors, you can activate antitumor immunity in ways that weren’t possible with just the existing [immune checkpoint inhibitors],” Dr. Lipson said.
The combination was tested as an initial, or first-line, treatment in the trial, which included more than 700 patients with advanced melanoma. Those who received both immunotherapy drugs lived for a median of approximately 10 months without their cancer getting worse, compared with about 4 and half months for those treated only with nivolumab.
Serious side effects were more frequent in patients treated with both drugs, and more of these patients stopped treatment early than those treated with nivolumab alone. Overall, the side effects were similar to those expected to be seen in patients being treated with checkpoint inhibitors, Dr. Lipson reported, but less common than is seen with other immunotherapy combinations.
“For melanoma patients, this is wonderful news,” said Sunandana Chandra, M.D., a medical oncologist at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, who was also involved in the trial. “This study shows us that there is potentially a new combination immunotherapy approach on the horizon that may be as efficacious, but with less toxicity.”
Two Drugs Are Better Than One
Immune checkpoint inhibitor combinations have already proven to be more effective than single agents in several cancers. For example, studies have shown that treatment with nivolumab and ipilimumab (Yervoy), which blocks an immune checkpoint protein known as CTLA-4, is more effective than nivolumab alone for melanoma that has spread to the brain.
Other studies have shown a similar benefit to combining immune checkpoint inhibitors for people with advanced non-small cell lung cancer. But the improved outcomes have generally come at the cost of more serious, and more frequent, side effects. Researchers have begun looking at other combinations to see if these can result in the same or better cancer-specific results, but with fewer side effects.
In the RELATIVITY-047 trial, 714 patients with previously untreated advanced melanoma were randomly assigned to receive either nivolumab plus relatlimab or nivolumab alone. Both drugs are given intravenously.
One year after beginning treatment, approximately 48% of patients who received both immunotherapy drugs were alive without their cancer getting worse, compared with 36% of patients treated with nivolumab alone.
Of the patients receiving nivolumab and relatlimab, 18.9% had serious side effects, including fatigue, rash, aching or painful joints, and diarrhea, compared with 9.7% of patients treated with nivolumab alone. The adverse events led 14.6% of the patients in the nivolumab and relatlimab group to stop treatment, compared with 6.7% of patients in the nivolumab alone group.
Comparing the combination of nivolumab and relatlimab with the combination of nivolumab and ipilimumab, “I think the big differentiator is in the reduced toxicities,” said Dario A. Vignali, Ph.D., who co-leads the Cancer Immunology and Immunotherapy Program at the University of Pittsburgh Medical Center Hillman Cancer Center.
The severe side effects associated with the combination of nivolumab and relatlimab are “way lower” than the severe side effects associated with the combination of nivolumab and ipilimumab, Dr. Vignali noted.
“Toxicities can be quite intolerable for a lot of patients, which reduces their quality of life. A third of patients [who receive] nivolumab plus ipilimumab have to discontinue treatment at some point, and that gives them very few [treatment] options,” said Dr. Vignali. “I think that [the] reduced toxicity is a game changer in many ways.”
Dr. Vignali was not involved in the RELATIVITY-047 trial but has served as a consultant to Bristol Myers Squibb, the company that developed relatlimab and funded the clinical trial.
Dr. Chandra agreed, saying that having a combination with fewer severe side effects “can really be practice changing.”
Waiting for More Data
James C. Yang, M.D., of NCI’s Center for Cancer Research, who was not involved in the trial, said the progression-free survival results are impressive but that it will be important to see if the combination of nivolumab and relatlimab can improve how long patients live overall.
“We need to get longer follow-up and the overall survival data to really lock down that this is a major advance and a major benefit,” said Dr. Yang, who added that it’s also important to determine whether this combination is effective against other cancers in addition to melanoma. “That will have a bigger impact on the whole cancer picture.”
Dr. Lipson said more data will be coming out from RELATIVITY-047 on overall response rate as well as overall survival.
In the meantime, the combination of nivolumab and relatlimab is already being tested as a treatment for people with less advanced melanoma. In a small study also presented at the ASCO meeting, the combination showed promising signs of effectiveness when given before and after surgery.
Relatlimab is just the tip of the iceberg when it comes to drugs that target LAG-3, Dr. Vignali said. “There are nearly a dozen additional LAG-3-targeting therapeutics in various stages of clinical trials, obviously further behind, but nevertheless creating a lot of potential therapeutics that might benefit patients,” he said.
Scientists are continuing to study combining immunotherapy with other treatments, including chemotherapy, targeted therapies, and other types of immune-based treatments.
All these combinations will give doctors more options when treating people with advanced melanoma and other cancers, Dr. Vignali continued. Which approach they choose will depend on a number of factors.
For some cancers, using only one immunotherapy drug may be the best choice because the side effects are just too much for a patient, or perhaps using two drugs “doesn’t show a lot of [added] benefit,” Dr. Vignali said. “But it wouldn't surprise me going forward if the majority of treatments for patients are combinations, simply because certain combinations will prove to be more effective.”