Childhood Ependymoma
Ependymoma is a rare type of tumor that starts in the brain or spinal cord. The brain controls all body functions, such as breathing, heart rate, memory and learning, emotion, and senses. The spinal cord is made up of bundles of nerve fibers that carry messages between the brain and the rest of the body. Together, the brain and spinal cord make up the central nervous system (CNS).
Ependymomas start when cells called ependymal cells grow without control. Ependymal cells line the ventricles and passageways in the brain and spinal cord and make cerebrospinal fluid (CSF), which acts as a cushion to protect the brain and spinal cord from injury. Ependymomas can spread when the CSF carries ependymoma cells to other parts of the CNS. Ependymomas rarely spread outside the CNS.
Children and adults can get ependymoma, but it is more common in young children. This type of tumor accounts for about 9% of all childhood brain and spinal cord tumors, affecting about 200 children per year in the United States.
Types of childhood ependymoma
There are different types of ependymomas depending on where the tumor is located. Three main types of ependymoma are seen in children:
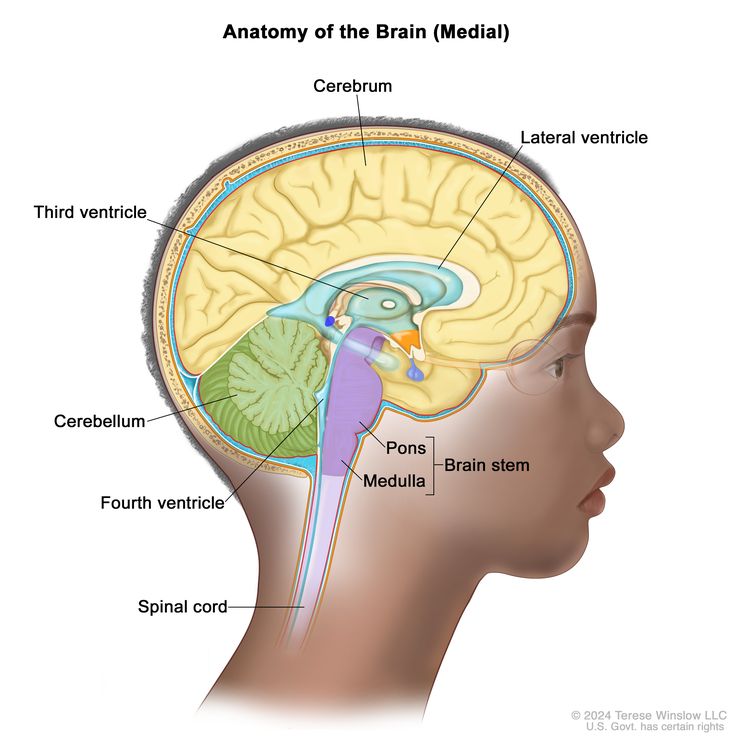
- Posterior fossa (infratentorial) ependymomas form in the lower part of the brain near the middle of the back of the head. In children, most ependymomas arise in this area of the brain and affect the cerebellum and brain stem.
- The cerebellum is the lower, back part of the brain (near the middle of the back of the head). The cerebellum controls movement, balance, and posture.
- The brain stem, located in the lowest part of the brain (just above the back of the neck), connects the brain to the spinal cord. The brain stem controls vital functions, such as breathing, heart rate, blood pressure, and the nerves and muscles used in seeing, hearing, walking, talking, and eating.
- Supratentorial ependymomas form at the top of the head and affect the cerebrum. Ependymoma in this area of the brain is less common in children.
- The cerebrum is the largest part of the brain, at the top of the head. The cerebrum controls thinking, learning, problem-solving, emotions, speech, reading, writing, and voluntary movement.
- Spinal cord ependymomas are rare in children. Most spinal cord ependymomas in children are a type called myxopapillary ependymomas, which usually occur in the lower part of the spine.
- The spinal cord is the column of nerve tissue that runs from the brain stem down the center of the back. Spinal cord nerves carry messages between the brain and the rest of the body, such as a message from the brain to cause muscles to move or a message from the skin to the brain to feel touch.
Causes and risk factors for childhood ependymoma
Childhood ependymoma is caused by certain changes to the way ependymal cells function, especially how they grow and divide into new cells. The exact cause of these cell changes is unknown. Learn more about how cancer develops at What Is Cancer?
A risk factor is anything that increases the chance of getting a disease. Children with an inherited condition called neurofibromatosis type 2 (NF2) may have an increased risk of developing ependymoma along the optic pathway. Not every child with this risk factor will develop ependymoma. And it will develop in some children who don't have a known risk factor.
Symptoms of childhood ependymoma
Symptoms of ependymoma depend on the child's age and where the tumor has formed. It's important to check with your child's doctor if your child has any of the symptoms below.
Symptoms of posterior fossa ependymoma in children can include:
- buildup of spinal fluid in the brain that may cause tiredness, vomiting, eyes that stay looking down, irritability, slowed development, or increased size of the head
- loss of balance or trouble walking
- neck pain
- loss of function of the nerves in the back of the brain
Symptoms of supratentorial ependymomas in children can include:
- seizures
- frequent headaches
- blurry vision
- nausea and vomiting
- changes in movement and sensation
Symptoms of spinal cord ependymomas in children can include:
- neck or back pain
- neck weakness or stiffness
- weakness in one or both legs
- trouble urinating
- a change in bowel function
These symptoms may be caused by problems other than an ependymoma. The only way to know is to see your child's doctor.
Tests to diagnose childhood ependymoma
If your child has symptoms that suggest an ependymoma, their doctor will need to find out if these are due to ependymoma or some other problem. The doctor will ask when the symptoms started and how often your child has been having them. They will also ask about your child's personal and family medical history and do a physical exam, including a neurologic exam. Depending on these results, they may recommend other tests. If your child is diagnosed with an ependymoma, the results of these tests will help you and your child's doctor plan treatment.
The tests used to diagnose ependymoma in children may include:
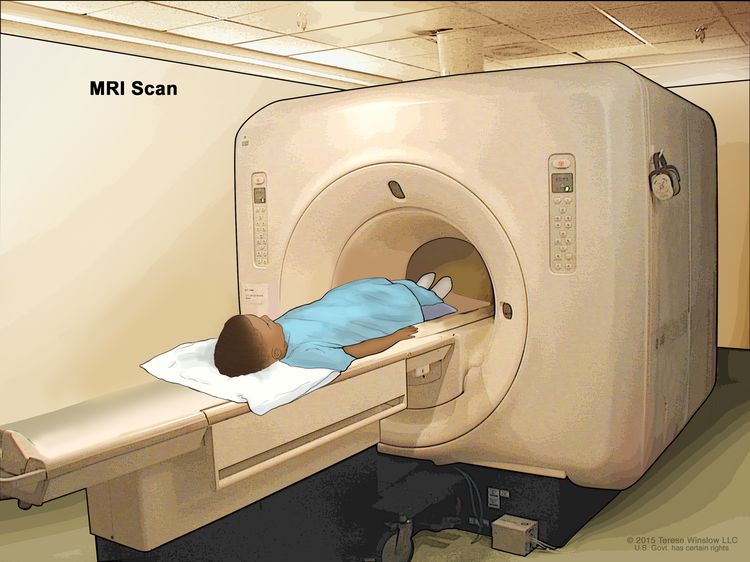
Magnetic resonance imaging (MRI) with or without gadolinium
MRI uses a magnet, radio waves, and a computer to make a series of detailed pictures of areas inside the brain and spinal cord. A substance called gadolinium is injected into a vein and travels through the bloodstream. The gadolinium may collect around the cancer cells so they show up brighter in the picture. This procedure is also called nuclear magnetic resonance imaging (NMRI).
Biopsy
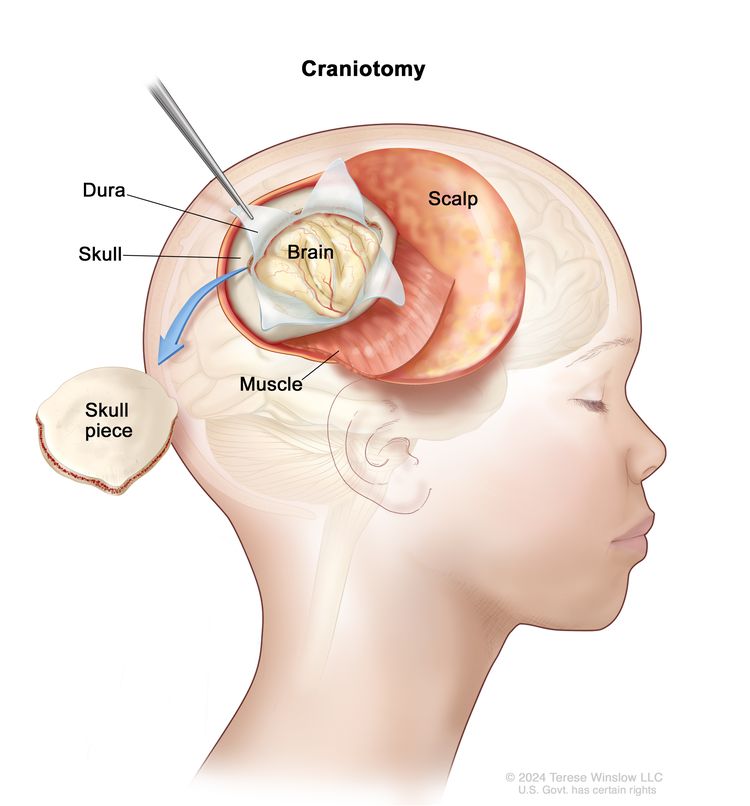
If the diagnostic tests show there may be a brain tumor, a biopsy can be done by removing part of the skull or making a small hole in the skull and using a needle or surgical device to remove a sample of the brain tissue. Sometimes, when a needle is used, it is guided by a computer to remove the tissue sample. A pathologist views the tissue under a microscope to look for cancer cells and determine the grade of the tumor. If cancer cells are found, the doctor will remove as much tumor as safely possible during the same surgery.
The following laboratory tests may be done on the tissue that was removed during the biopsy:
- Immunohistochemistry uses antibodies to check for certain antigens (markers) in a sample of a patient's tissue. The antibodies are usually linked to an enzyme or a fluorescent dye. After the antibodies bind to a specific antigen in the tissue sample, the enzyme or dye is activated, and the antigen can then be seen under a microscope. This type of test is used to help diagnose cancer and to help tell one type of cancer from another type.
- Next-generation sequencing uses computers to piece together DNA or RNA fragments in order to sequence a person or other organism's entire DNA, large segments of DNA or RNA, or the DNA in specific types of cells from a sample of tissue. Next-generation sequencing can also identify changes in certain areas of the genome or in specific genes. There are many different types of next-generation sequencing methods, including whole-genome sequencing, whole-exome sequencing, multigene panel testing, and transcriptome sequencing. Next-generation sequencing may help researchers understand the cause of certain diseases, such as cancer. Also called massively parallel sequencing and NGS.
The Molecular Characterization Initiative offers free molecular testing to children, adolescents, and young adults with certain types of newly diagnosed cancer. The program is offered through NCI's Childhood Cancer Data Initiative. To learn more, visit About the Molecular Characterization Initiative.
Lumbar puncture
Lumbar puncture is a procedure used to collect cerebrospinal fluid (CSF) from the spinal column. This is done by placing a needle between two bones in the spine and into the lining around the spinal cord to remove a sample of CSF. The sample of CSF is checked under a microscope for signs of tumor cells. The sample may also be checked for the amounts of protein and glucose. 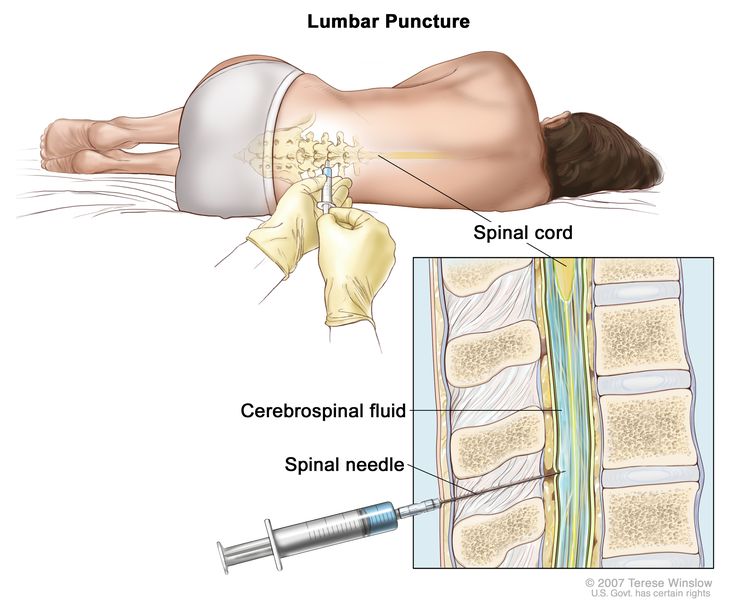
Getting a second opinion
You may want to get a second opinion to confirm your child's diagnosis and treatment plan. If you seek a second opinion, you will need to get medical test results and reports from the first doctor to share with the second doctor. The second doctor will review the pathology report, slides, and scans. This doctor may agree with the first doctor, suggest changes to the treatment plan, or provide more information about your child's cancer.
To learn more about choosing a doctor and getting a second opinion, see Finding Cancer Care. You can contact NCI's Cancer Information Service via chat, email, or phone (both in English and Spanish) for help finding a doctor or hospital that can provide a second opinion. For questions you might want to ask at your child's appointments, see Questions to Ask Your Doctor About Cancer.
Stages and tumor grades of childhood ependymoma
Staging is the process of learning the extent of the cancer in the body and is often used to help plan treatment and make a prognosis. There is no staging system used for childhood ependymoma, but it is given a tumor grade. Tumor grading is based on the World Health Organization (WHO) criteria.
Tumor grade describes how abnormal the cancer cells look under a microscope, how quickly the tumor is likely to grow and spread, and how likely the tumor is to come back after treatment. The WHO criteria also classifies ependymomas by their location in the brain or spinal cord (see the section on Types of childhood ependymoma) and the tumor's molecular or genetic features.
Low-grade (grade I) cancer cells look more like normal cells than high-grade (grades II and III) cancer cells. Grade I cancer cells also tend to grow and spread more slowly than grade II and III cancer cells.
Childhood ependymoma often comes back after treatment, sometimes as long as 15 years after the initial treatment. The tumor commonly comes back at the original cancer site, although it can also spread to areas near the original site. It is rare for ependymoma to spread to areas far from the original cancer site.
Types of treatment for childhood ependymoma
Who treats children with ependymoma?
A pediatric oncologist, a doctor who specializes in treating children with cancer, oversees treatment for childhood ependymoma. The pediatric oncologist works with other health care providers who are experts in treating children with brain tumors and also specialize in other areas of medicine. Other specialists may include:
There are different types of treatment for children and adolescents with ependymoma. You and your child's care team will work together to decide treatment. Many factors will be considered, such as where the cancer is located, your child's age and overall health, and the type and grade of the ependymoma.
Your child's treatment plan will include information about the cancer, the goals of treatment, treatment options, and the possible side effects. It will be helpful to talk with your child's care team before treatment begins about what to expect. For help every step of the way, see our booklet, Children with Cancer: A Guide for Parents.
The types of treatment your child might have include:
Surgery
Surgery to remove the tumor and some of the healthy tissue around it is usually the first treatment for childhood ependymoma. Surgery may be done to obtain a biopsy sample to confirm the diagnosis (see Tests to diagnose childhood ependymoma), relieve symptoms caused by the tumor pressing on the brain or spinal cord, and to remove as much of the tumor as possible.
An MRI is often done after the tumor is removed to find out whether any tumor remains. If tumor remains, a second surgery to remove as much of the remaining tumor as possible may be done.
Some children may be given chemotherapy or radiation therapy after surgery to kill any cancer cells that are left. Treatment given after surgery, to lower the risk that the cancer will come back, is called adjuvant therapy.
Radiation therapy
Radiation therapy is a cancer treatment that uses high-energy x-rays or other types of radiation to kill cancer cells or keep them from growing. Ependymoma is treated with external beam radiation therapy. This type of therapy uses a machine outside the body to send radiation toward the area of the body with cancer. Radiation therapy may be given alone or with other treatments, such as chemotherapy.
Certain ways of giving external beam radiation therapy can help keep radiation from damaging nearby healthy tissue. These include:
- Conformal radiation therapy uses a computer to make a 3-dimensional (3-D) picture of the tumor and shapes the radiation beams to fit the tumor.
- Intensity-modulated radiation therapy (IMRT) is a type of 3-D radiation therapy that uses a computer to make pictures of the size and shape of the tumor. Thin beams of radiation of different intensities (strengths) are aimed at the tumor from many angles.
- Proton-beam radiation therapy is a type of high-energy radiation therapy. A radiation therapy machine aims streams of protons (tiny, invisible, positively-charged particles) at the cancer cells to kill them.
- Stereotactic radiosurgery uses a rigid head frame attached to the skull to keep the head still during the radiation treatment. A machine aims a single large dose of radiation directly at the tumor. This procedure does not involve surgery. It is also called stereotaxic radiosurgery, radiosurgery, and radiation surgery.
Younger children who receive radiation therapy to the brain have a higher risk of problems with growth and development than older children. 3-D conformal radiation therapy and proton-beam therapy are being studied in young children to see if it decreases the effects of radiation on growth and development.
Learn more about External Beam Radiation Therapy for Cancer and Radiation Therapy Side Effects.
Chemotherapy
Chemotherapy (also called chemo) uses drugs to stop the growth of cancer cells. Chemotherapy either kills the cancer cells or stops them from dividing. Chemotherapy may be given alone or with other types of treatment, such as radiation therapy.
For ependymoma, chemotherapy is injected into a vein. When given this way, the drugs enter the bloodstream to reach cancer cells throughout the body. Chemotherapy that may be used alone or in combination includes:
Other chemotherapy drugs not listed here may also be used.
Learn more at Chemotherapy to Treat Cancer.
Clinical trials
A treatment clinical trial is a research study meant to help improve current treatments or obtain information on new treatments for people with cancer. Because cancer in children is rare, taking part in a clinical trial should be considered.
Find clinical trials for people with ependymoma at Ependymoma Clinical Trials, or use our clinical trial search to find NCI-supported cancer clinical trials that are accepting patients. You can search for trials based on the type of cancer, the age of the patient, and where the trials are being done. Some clinical trials are open only to patients who have not started treatment. Clinical trials supported by other organizations can be found on the ClinicalTrials.gov website.
Learn more at Clinical Trials Information for Patients and Caregivers.
Treatment of childhood ependymoma in the brain or brain stem
Treatment of newly diagnosed childhood ependymoma in the brain or brain stem includes surgery.
After surgery, the plan for further treatment depends on:
- the ependymoma subtype
- whether any cancer cells remain after surgery
- whether the cancer has spread to other parts of the brain or spinal cord
- your child's age
When the tumor is completely removed and cancer cells have not spread, treatment may include radiation therapy.
When part of the tumor remains after surgery, but cancer cells have not spread, treatment may include:
- a second surgery to remove as much of the remaining tumor as possible
- radiation therapy
- chemotherapy before radiation therapy
When cancer cells have spread within the brain and spinal cord, treatment may include:
- radiation therapy to the brain and spinal cord
- chemotherapy
Treatment for children younger than 1 year of age may include:
- chemotherapy
- a clinical trial of 3-dimensional (3-D) conformal radiation therapy or proton-beam radiation therapy
Radiation therapy is not given to children until they are older than 1 year.
To learn more about these treatments, see Types of treatment for childhood ependymoma.
Treatment of childhood spinal cord ependymoma
Treatment of newly diagnosed childhood myxopapillary spinal ependymoma (grade 2) is surgery. Sometimes radiation therapy is given after surgery.
Treatment of newly diagnosed childhood nonmyxopapillary spinal ependymoma is surgery. Sometimes radiation therapy is given after surgery for children with grade 2 or grade 3 tumors.
To learn more about these treatments, see Types of treatment for childhood ependymoma.
Treatment of recurrent childhood ependymoma
Treatment of recurrent childhood ependymoma may include:
- surgery
- external beam radiation therapy
- chemotherapy
To learn more about these treatments, see Types of treatment for childhood ependymoma.
Prognostic factors for childhood ependymoma
If your child has been diagnosed with ependymoma, you likely have questions about how serious the cancer is and your child's chances of survival. The likely outcome or course of a disease is called prognosis. Your child's prognosis depends on many factors, including:
- where the tumor has formed in the central nervous system (CNS)
- whether there are certain changes in the genes or chromosomes of the cancer cells
- whether the cancer was completely removed by surgery; the prognosis is better if the cancer can be completely removed
- the type and grade of ependymoma
- your child's age when the tumor was diagnosed
- whether the cancer has spread to other parts of the brain or spinal cord; the prognosis is better if the cancer has not spread
- whether the tumor has just been diagnosed or has come back
Prognosis also depends on whether radiation therapy was given, the type and treatment dose, and whether chemotherapy alone was given.
No two people are alike, and responses to treatment can vary greatly. Your child's cancer care team is in the best position to talk with you about your child's prognosis.
Side effects and late effects of cancer treatment
Cancer treatments can cause side effects. Which side effects your child might have depends on the type of treatment they receive, the dose, and how their body reacts. Talk with your child’s treatment team about which side effects to look for and ways to manage them.
To learn more about side effects that begin during treatment for cancer, visit Side Effects.
Problems from cancer treatment that begin 6 months or later after treatment and continue for months or years are called late effects. Late effects of treatment for childhood ependymoma may include:
- physical problems, including problems with:
- changes in mood, feelings, thinking, learning, or memory
- second cancers (new types of cancer), such as thyroid cancer or brain cancer
Some late effects may be treated or controlled. It is important to talk with your child's doctors about the effects cancer treatment can have on your child. Learn more about Late Effects of Treatment for Childhood Cancer.
Follow-up care
As your child goes through treatment, they will have follow-up tests or check-ups. Some of the tests that were done to diagnose the cancer may be repeated to see how well the treatment is working. Decisions about whether to continue, change, or stop treatment may be based on the results of these tests.
Childhood ependymoma may come back as long as 15 years or more after initial treatment. So once your child finishes treatment, they will continue to have certain tests from time to time. The results of these tests can show if your child's condition has changed or if the cancer has come back.
Your child may receive an MRI of the brain and spinal cord at the following intervals:
- every 3 to 4 months for the first 2 to 3 years
- every 6 months at 4 to 5 years after treatment
- once a year at more than 5 years after treatment
Coping with your child's cancer
When a child has cancer, every member of the family needs support. Taking care of yourself during this difficult time is important. Reach out to your child's treatment team and to people in your family and community for support. To learn more, see Support for Families: Childhood Cancer and the booklet Children with Cancer: A Guide for Parents.
Related resources
For more childhood cancer information and other general cancer resources, see: