Leadership & Expert Views - Cancer Currents Blog
Commentaries from scientific leaders at NCI and other institutions about cancer research topics, programs, and initiatives.
-
Prostate Cancer Prevention and Finasteride: A Conversation with NCI’s Dr. Howard Parnes
The Prostate Cancer Prevention Trial showed that finasteride can reduce the risk of prostate cancer, but might increase the risk of aggressive disease. NCI’s Howard Parnes talks about subsequent findings and what they mean for men aged 55 and older.
-
Ensuring a Smooth Leadership Transition at NCI
Doug Lowy, M.D., a long-time researcher and deputy director at NCI, addresses the cancer community as the institute's acting director, a position he previously held for more than 2 years.
-
Making Cancer Clinical Trials Available to More Patients
NCI is expanding eligibility criteria for its cancer clinical trials in the hope that more patients will join, leading to more rapid advances. The goal is to maximize the number of patients who are eligible to enroll while maintaining their safety.
-
Partnering with Small Business to Advance Innovation in Cancer Research and Care
NCI Director Dr. Norman Sharpless describes how NCI’s Small Business Innovation Research and Small Business Technology Transfer Programs act as “engines of innovation” and shares recommendations from a federal working group for strengthening the programs.
-
The HPV Vaccine: Increasing the Use of an Important Cancer Prevention Tool
Dr. Barbara Rimer, chair of the President’s Cancer Panel, summarizes the panel’s recent report on the HPV vaccine, which includes priorities and strategies to ensure that more people receive this critical cancer vaccine.
-
Plans for NCI’s Fiscal Year 2019 Budget
NCI Director Dr. Ned Sharpless describes important decisions NCI leaders have recently made with respect to the institute’s fiscal year 2019 budget, including those related to the Cancer Moonshot and early-stage investigators.
-
The Cancer Moonshot: Moving from Planning to Research
Dr. Dinah Singer, co-chair of the Cancer Moonshot Blue Ribbon Panel, provides an update to the cancer community on the science being supported under the Moonshot initiative.
-
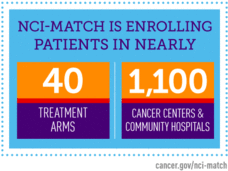
NCI-MATCH Update: More Labs, New Arms, and Initial Findings
NCI’s Dr. Lyndsay Harris provides an update on the NCI-MATCH trial, including the opening of new trial treatment arms and the addition of new laboratories to perform testing on tumor samples of prospective trial participants.
-
Reflections on My First Year as NCI Director
October 2018 marked Dr. Norman Sharpless’s one-year anniversary as NCI director. He reflects on noteworthy events from the past 12 months and his excitement about leading NCI at such a critical juncture in the history of cancer research.
-
Studying “Total Diet” and Its Impact on Health, Including Cancer Risk
Researchers are shifting how they assess diet and cancer risk, looking beyond the impact of individual foods or nutrients on health, and taking a more holistic approach, considering dietary patterns across the lifespan.
-
The R50 Research Specialist Award: Ensuring a Stable Cancer Research Workforce
With the increasing reliance on advanced technologies and the rise of big data in cancer research, NCI launched the R50 Research Specialist Award to help sustain a stable workforce of experienced scientists who can take fullest advantage of these tools and resources.
-
Hope and Challenge: The NCI Annual Plan and Budget Proposal for Fiscal Year 2020
NCI has released its Fiscal Year 2020 Annual Plan and Budget Proposal. The plan represents NCI’s best professional judgement on the optimum funding needed to make the most rapid progress against cancer.
-
NCI and FDA Address Common Concerns: A Win–Win for Making Progress against Cancer
NCI Director Dr. Norman Sharpless discusses efforts between NCI and FDA to advance progress against cancer, including initiatives to improve labeling of patient tumor and tissue samples and to address challenges presented by new tobacco products.
-
Integrating Geriatric Assessment into Cancer Care: A Conversation with Dr. Supriya Mohile
Dr. Supriya Mohile discusses the unique issues experienced by older adults with cancer and efforts to incorporate geriatric assessment into patient care, including the publication of recent ASCO clinical guidelines on geriatric cancer care.
-
Enhancements to NCI’s SEER Program Creating New Research Opportunities
NCI’s SEER program is expanding in size and operating a series of innovative pilot studies. As Dr. Lynne Penberthy explains, these studies are setting the stage for the routine collection of more clinical and genomic data that will help researchers better understand cancer and its impact.
-
Developing Biomarkers for Immunotherapy: A Conversation with Drs. Magdalena Thurin and Helen Chen
NCI is supporting a new research network to develop biomarkers for cancer immunotherapy. In this interview, NCI’s Dr. Helen Chen and Magdalena Thurin explain the networks’ structure and its goals.
-
The Opioid Epidemic and Cancer Pain Management: A Conversation with Dr. Judith Paice
Dr. Judith Paice, of the Cancer Pain Program at Northwestern University’s Feinberg School of Medicine, discusses the impacts of the opioid epidemic on cancer patients and how providers can address concerns about opioid misuse when managing cancer pain.
-
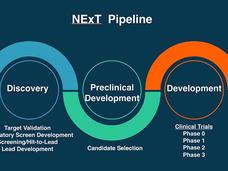
NExT: Advancing Promising Cancer Therapies from the Lab to Clinical Trials
The NCI Experimental Therapeutics (NExT) program works with researchers and top scientific experts to advance promising or novel cancer therapies from the earliest stages of research to human clinical trials.
-
Maximizing the Prospects for Progress Against Cancer
NCI Director Dr. Norman Sharpless highlights some of the important research findings from the 2018 American Society of Clinical Oncology annual meeting and discusses the rapid pace of progress in cancer research.
-
Cancer Control in American Indian and Alaska Native Populations: A Conversation with Dr. Shobha Srinivasan
American Indian and Alaska Native populations are disproportionately affected by certain cancers. In this interview, Dr. Shobha Srinivasan discusses some of these disparities and programs funded by NCI that are helping to address them.