Childhood Gastrointestinal Stromal Tumors (PDQ®)–Patient Version
What is childhood gastrointestinal stromal tumor?
Childhood gastrointestinal stromal tumor (GIST) is a cancer that forms in the tissues of the wall of the stomach or intestines. Childhood GIST usually occurs in the stomach. It is most common in girls and typically appears in the teen years.
The gastrointestinal (GI) tract is part of the body's digestive system. It helps digest food and takes nutrients (vitamins, minerals, carbohydrates, fats, proteins, and water) from food so they can be used by the body. The GI tract is made up of the:
- stomach
- small intestine
- large intestine (colon)
GISTs usually begin in cells in the tissues of the wall of the stomach or intestines that help food move along the digestive tract. 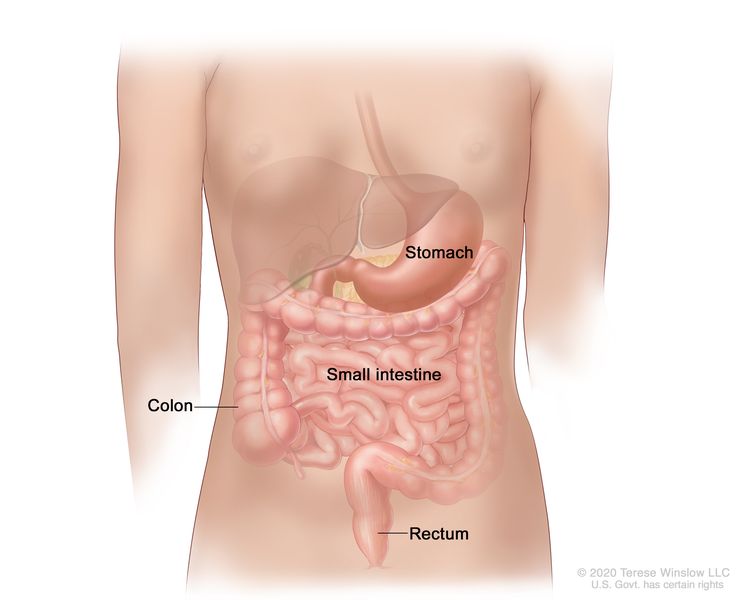
GISTs in children are not the same as GISTs in adults. Children should be seen at centers that specialize in treating GISTs in children and adolescents.
Causes and risk factors for childhood gastrointestinal stromal tumor
Childhood GIST is caused by certain changes to the way the cells in the wall of the stomach and intestines function, especially how they grow and divide into new cells. Often, the exact cause of these changes is unknown. Learn more about how cancer develops at What Is Cancer?
A risk factor is anything that increases the chance of getting a disease. Not every child with a risk factor will develop a GIST. And it will develop in some children who don't have a known risk factor.
GIST may occur as part of the following syndromes:
Children with neurofibromatosis type 1 (NF1) may also have an increased risk.
Talk with your child's doctor if you think your child may be at risk.
Symptoms of childhood gastrointestinal stromal tumor
The symptoms of childhood GIST may not appear until the tumor grows bigger. It's important to check with your child's doctor if your child has:
- anemia, which causes tiredness, dizziness, fast or irregular heartbeat, shortness of breath, or pale skin
- blood (either bright red or very dark) in the stool
- a lump in the abdomen
- a blockage in the intestine, which causes cramping pain in the abdomen, nausea, vomiting, diarrhea, constipation, and swelling of the abdomen
These symptoms may be caused by problems other than a GIST. The only way to know is for your child to see a doctor.
Tests to diagnose childhood gastrointestinal stromal tumor
If your child has symptoms that suggest a stomach or intestinal tumor, the doctor will need to find out if these are due to cancer or another problem. The doctor will ask when the symptoms started and how often your child has been having them. They will also ask about your child's personal and family medical history and do a physical exam. Depending on these results, they may recommend other tests. If your child is diagnosed with a GIST, the results of these tests will help plan treatment.
The tests used to diagnose GIST may include:
Gene testing
Gene testing analyzes cells or tissues from the tumor to look for changes in the KIT, PDGFRA, and SDH genes. Knowing whether there are changes in these genes can help diagnose GIST and plan treatment.
Magnetic resonance imaging (MRI)
MRI uses a magnet, radio waves, and a computer to make a series of detailed pictures of areas in the body. This procedure is also called nuclear magnetic resonance imaging (NMRI).
CT scan
CT scan (CAT scan) uses a computer linked to an x-ray machine to make a series of detailed pictures of areas inside the body. The pictures are taken from different angles and are used to create 3-D views of tissues and organs. A dye may be injected into a vein or swallowed to help the organs or tissues show up more clearly. This procedure is also called computed tomography, computerized tomography, or computerized axial tomography. Learn more about Computed Tomography (CT) Scans and Cancer.
PET scan
A PET scan (positron emission tomography scan) uses a small amount of radioactive sugar (also called radioactive glucose) that is injected into a vein. The PET scanner rotates around the body and makes pictures of where sugar is being used by the body. Cancer cells show up brighter in the pictures because they are more active and take up more sugar than normal cells do.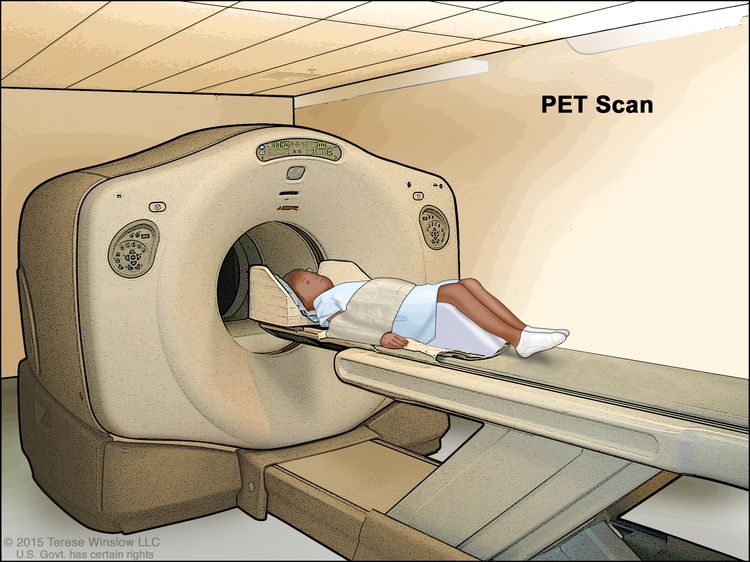
X-ray
X-ray is a type of radiation that can go through the body and make pictures of areas inside the body, such as the abdomen or the area where the tumor formed.
Biopsy
Biopsy is the removal of a sample of tissue from the tumor so that a pathologist can view it under a microscope to check for cancer. The following types of biopsies may be used to check for GIST:
- Fine-needle aspiration uses a thin needle to remove tissue from the tumor.
- Endoscopy looks at organs and tissues inside the body to check for abnormal areas. An endoscope is inserted through an incision (cut) in the skin or opening in the body, such as the mouth or anus. An endoscope is a thin, tube-like instrument with a light and a lens for viewing. It may also have a tool to remove tissue or lymph node samples, which are checked under a microscope for cancer.
The following laboratory test may be done to study the tissue samples:
- Immunohistochemistry uses antibodies to check for certain antigens (markers) in a sample of a patient's cells or tissue. The antibodies are usually linked to an enzyme or a fluorescent dye. After the antibodies bind to a specific antigen in the tissue sample, the enzyme or dye is activated, and the antigen can then be seen under a microscope. This type of test looks for the enzyme SDH in the patient's tissue. When SDH is not present, it is called SDH-deficient GIST. Knowing whether the cancer is SDH-deficient can help plan treatment.
Getting a second opinion
You may want to get a second opinion to confirm your child's cancer diagnosis and treatment plan. If you seek a second opinion, you will need to get medical test results and reports from the first doctor to share with the second doctor. The second doctor will review the pathology report, slides, and scans. They may agree with the first doctor, suggest changes to the treatment plan, or provide more information about your child's cancer.
To learn more about choosing a doctor and getting a second opinion, visit Finding Cancer Care. You can contact NCI's Cancer Information Service via chat, email, or phone (both in English and Spanish) for help finding a doctor or hospital that can provide a second opinion. For questions you might want to ask at your child's appointments, visit Questions to Ask Your Doctor About Cancer.
Who treats children with gastrointestinal stromal tumor?
A pediatric oncologist, a doctor who specializes in treating children with cancer, oversees the treatment of GIST. The pediatric oncologist works with other health care providers who are experts in treating children with cancer and who specialize in certain areas of medicine. Other specialists may include:
Treatment of childhood gastrointestinal stromal tumor
There are different types of treatment for children and adolescents with GIST. You and your child's care team will work together to decide treatment. Many factors will be considered, such as your child's overall health, whether the tumor has changes to the KIT, PDGFRA, or SDH genes, and whether the cancer is newly diagnosed or has come back.
Your child's treatment plan will include information about the cancer, the goals of treatment, treatment options, and the possible side effects. It will be helpful to talk with your child's care team before treatment begins about what to expect. For help every step of the way, visit our booklet, Children with Cancer: A Guide for Parents.
Types of treatment your child might have include:
- Children with a GIST that has changes in the KIT or PDGFRA gene are treated with targeted therapy. Targeted therapy uses drugs or other substances to block the action of specific enzymes, proteins, or other molecules involved in the growth and spread of cancer cells. Imatinib and sunitinib are targeted therapies approved for adults with GIST and may be used in children and adolescents. Learn more about Targeted Therapy to Treat Cancer.
- Children with a GIST that is SDH-deficient are treated with surgery to remove the tumor. More surgery may be needed if an intestinal blockage or bleeding occurs.
If the cancer comes back after treatment, your child's doctor will talk with you about what to expect and possible next steps. There might be treatment options that may shrink the cancer or control its growth. If there are no treatments, your child can receive care to control symptoms from cancer so they can be as comfortable as possible.
Clinical trials
For some children, joining a clinical trial may be an option. There are different types of clinical trials for childhood cancer. For example, a treatment trial tests new treatments or new ways of using current treatments. Supportive care and palliative care trials look at ways to improve quality of life, especially for those who have side effects from cancer and its treatment.
You can use the clinical trial search to find NCI-supported cancer clinical trials accepting participants. The search allows you to filter trials based on the type of cancer, your child's age, and where the trials are being done. Clinical trials supported by other organizations can be found on the ClinicalTrials.gov website.
Learn more about clinical trials, including how to find and join one, at Clinical Trials Information for Patients and Caregivers.
Side effects and late effects of treatment
Cancer treatments can cause side effects. Which side effects your child might have depends on the type of treatment they receive, the dose, and how their body reacts. Talk with your child's treatment team about which side effects to look for and ways to manage them.
To learn more about side effects that begin during treatment for cancer, visit Side Effects.
Problems from cancer that begin 6 months or later after treatment and continue for months or years are called late effects. Late effects of cancer treatment may include:
- physical problems
- changes in mood, feelings, thinking, learning, or memory
- second cancers (new types of cancer) or other conditions
Some late effects may be treated or controlled. It is important to talk with your child's doctors about the possible late effects caused by some treatments. Learn more about Late Effects of Treatment for Childhood Cancer.
Follow-up care
As your child goes through treatment, they will have follow-up tests or check-ups. Some tests that were done to diagnose or stage the cancer may be repeated to see how well the treatment is working. Decisions about whether to continue, change, or stop treatment may be based on the results of these tests.
Some of the tests will continue to be done from time to time after treatment has ended. The results of these tests can show if your child's condition has changed or if the cancer has recurred (come back).
To learn more about follow-up tests, visit Tests to diagnose childhood gastrointestinal stromal tumor.
Coping with your child's cancer
When your child has cancer, every member of the family needs support. Taking care of yourself during this difficult time is important. Reach out to your child's treatment team and to people in your family and community for support. To learn more, visit Support for Families: Childhood Cancer and the booklet Children with Cancer: A Guide for Parents.
Related resources
For more childhood cancer information and other general cancer resources, visit:
About This PDQ Summary
About PDQ
Physician Data Query (PDQ) is the National Cancer Institute's (NCI's) comprehensive cancer information database. The PDQ database contains summaries of the latest published information on cancer prevention, detection, genetics, treatment, supportive care, and complementary and alternative medicine. Most summaries come in two versions. The health professional versions have detailed information written in technical language. The patient versions are written in easy-to-understand, nontechnical language. Both versions have cancer information that is accurate and up to date and most versions are also available in Spanish.
PDQ is a service of the NCI. The NCI is part of the National Institutes of Health (NIH). NIH is the federal government’s center of biomedical research. The PDQ summaries are based on an independent review of the medical literature. They are not policy statements of the NCI or the NIH.
Purpose of This Summary
This PDQ cancer information summary has current information about the treatment of childhood gastrointestinal stromal tumors. It is meant to inform and help patients, families, and caregivers. It does not give formal guidelines or recommendations for making decisions about health care.
Reviewers and Updates
Editorial Boards write the PDQ cancer information summaries and keep them up to date. These Boards are made up of experts in cancer treatment and other specialties related to cancer. The summaries are reviewed regularly and changes are made when there is new information. The date on each summary ("Updated") is the date of the most recent change.
The information in this patient summary was taken from the health professional version, which is reviewed regularly and updated as needed, by the PDQ Pediatric Treatment Editorial Board.
Clinical Trial Information
A clinical trial is a study to answer a scientific question, such as whether one treatment is better than another. Trials are based on past studies and what has been learned in the laboratory. Each trial answers certain scientific questions in order to find new and better ways to help cancer patients. During treatment clinical trials, information is collected about the effects of a new treatment and how well it works. If a clinical trial shows that a new treatment is better than one currently being used, the new treatment may become "standard." Patients may want to think about taking part in a clinical trial. Some clinical trials are open only to patients who have not started treatment.
Clinical trials can be found online at NCI's website. For more information, call the Cancer Information Service (CIS), NCI's contact center, at 1-800-4-CANCER (1-800-422-6237).
Permission to Use This Summary
PDQ is a registered trademark. The content of PDQ documents can be used freely as text. It cannot be identified as an NCI PDQ cancer information summary unless the whole summary is shown and it is updated regularly. However, a user would be allowed to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks in the following way: [include excerpt from the summary].”
The best way to cite this PDQ summary is:
PDQ® Pediatric Treatment Editorial Board. PDQ Childhood Gastrointestinal Stromal Tumors. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/soft-tissue-sarcoma/patient/child-gist-treatment-pdq. Accessed <MM/DD/YYYY>.
Images in this summary are used with permission of the author(s), artist, and/or publisher for use in the PDQ summaries only. If you want to use an image from a PDQ summary and you are not using the whole summary, you must get permission from the owner. It cannot be given by the National Cancer Institute. Information about using the images in this summary, along with many other images related to cancer can be found in Visuals Online. Visuals Online is a collection of more than 3,000 scientific images.
Disclaimer
The information in these summaries should not be used to make decisions about insurance reimbursement. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s E-mail Us.