Novel Strategy Isolates Immune Cells in the Blood that Recognize Melanoma
, by NCI Staff
Patients with metastatic melanoma have experienced dramatic tumor regressions in early clinical trials of adoptive cell transfer—a technique in which immune cells are collected from patients, expanded in the laboratory, and infused back into patients to attack the cancer. However, to date, these immune cells have been harvested only from solid tumor tissue, a process that requires invasive surgery and is not always possible.
Now, scientists in NCI’s Center for Cancer Research (CCR) have developed a novel strategy for identifying tumor-reactive and mutation-specific immune cells circulating in the blood of patients with melanoma.
Because it does not require surgery and can identify T cells that recognize proteins specific to each patient’s tumor, the research team believes the findings, published February 22 in Nature Medicine, may expand studies of adoptive cell transfer and other personalized immunotherapies to a greater number of patients.
Fishing for “Neoantigen-Reactive” T Cells
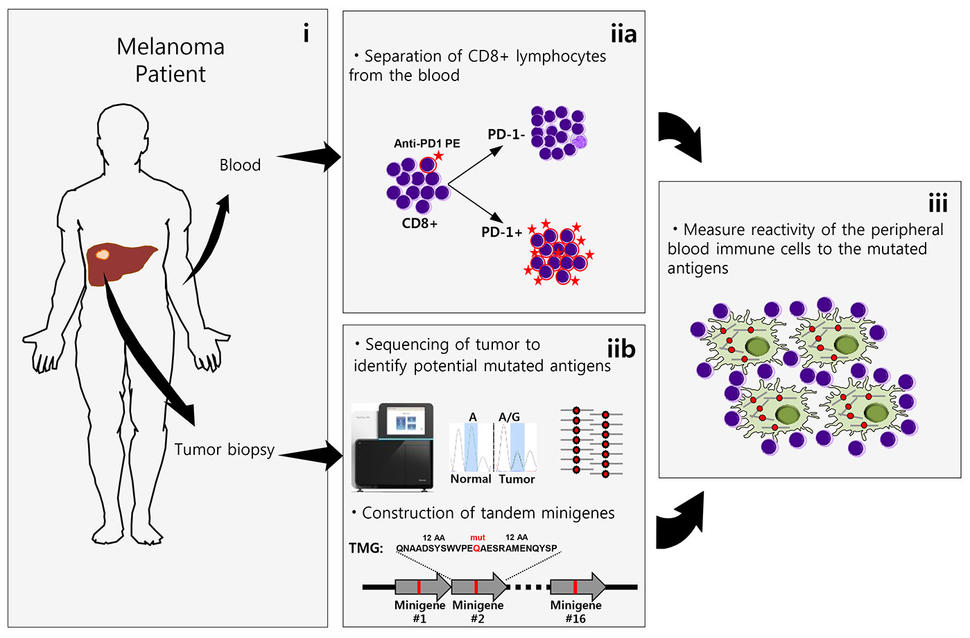
The researchers, led by Alena Gros, Ph.D., and Steven A. Rosenberg, M.D., Ph.D., of CCR’s Surgery Branch, studied the blood from five patients with stage 4 melanoma who later underwent adoptive cell transfer using immune cells collected from their tumors—called tumor infiltrating lymphocytes (TILs).
They focused on a type of immune cell, CD8+ T cells, that express the protein PD-1 on their surfaces (CD8+PD-1+ cells), which represent a small fraction of the peripheral blood CD8+ cells. In a previous study, the team showed that a subset of CD8+PD-1+ TILs can reliably recognize and attack tumor cells.
In the current study, the researchers used a novel personalized screening approach to identify immune cells that recognized mutated proteins, or neoantigens, produced by specific mutations in the patients’ tumors.
The screening method involves identifying genetic mutations in each patient’s tumor samples and creating a library of laboratory constructs, called minigenes, that correspond to these mutations. Immune cells collected from the patient’s blood are then exposed to the proteins produced by the minigene library to identify the immune cells that react to specific neoantigens. Using this approach, the researchers could find neoantigen-reactive cells in this subgroup of CD8+PD-1+ T cells in blood samples of four of the five patients. CD8+ T cells that did not express PD-1 did not recognize any neoantigens.
In the one patient whose peripheral blood did not have any neoantigen-reactive T cells, additional experiments showed that the CD8+PD-1+ cells could still recognize the tumor cells.
“A tumor can express multiple antigens that can be recognized by immune cells; some are [the result of] mutations, and others that are known to be expressed preferentially by the tumor and not by normal cells,” Dr. Gros explained. “Our results show that the CD8+PD-1+ lymphocytes in the blood of melanoma patients contain some that recognize neoantigens as well as other tumor antigens, and thus selection of this population can identify the personalized immune cells that recognize the patient’s tumor.”
Expanding Immunotherapy Opportunities
“We have known for a while that tumor-reactive cells can be isolated from the tumor, but our work now describes a new way to identify these cells and enrich for them straight out of the blood,” Dr. Gros added. “The main advantage [of this technique] is that it overcomes the need for having to perform invasive surgery to study these cells and to potentially use them therapeutically.”
The CCR team is looking at using tumor-specific T cells from peripheral blood samples in adoptive cell transfer. They are also studying a procedure that isolates a tumor-specific receptor on such T cells and then engineers other immune cells from the same patient to express that receptor. A set of experiments within the current study showed that both options could produce a batch of immune cells that recognize tumor tissue.
The researchers are also studying whether they can extend this approach to other tumor types, Dr. Gros said, including patients with gastrointestinal cancer.
For example, in an ongoing study of patients with advanced gastrointestinal cancers, of the 26 patients studied to date, 23 have TILs that recognize specific mutations in their tumors. They are currently evaluating whether these cells can also be detected in patients’ peripheral blood, Dr. Gros said.