Checking In on Cancer Checkpoint Inhibitors
, by NCI Staff
Earlier this year, former U.S. President Jimmy Carter announced that he was undergoing treatment for advanced melanoma that included the immunotherapy drug pembrolizumab (Keytruda®)—part of a class of drugs known as checkpoint inhibitors. He recently reported that his most recent MRI scan did not reveal any signs of the original cancer or any new tumors.
In this interview, James Gulley, M.D., Ph.D., head of the Immunotherapy Section in the Genitourinary Malignancy Branch of NCI's Center for Cancer Research and director of the Center's Medical Oncology Service, discusses checkpoint inhibitors, their impact on patient care, and future directions for these therapies.
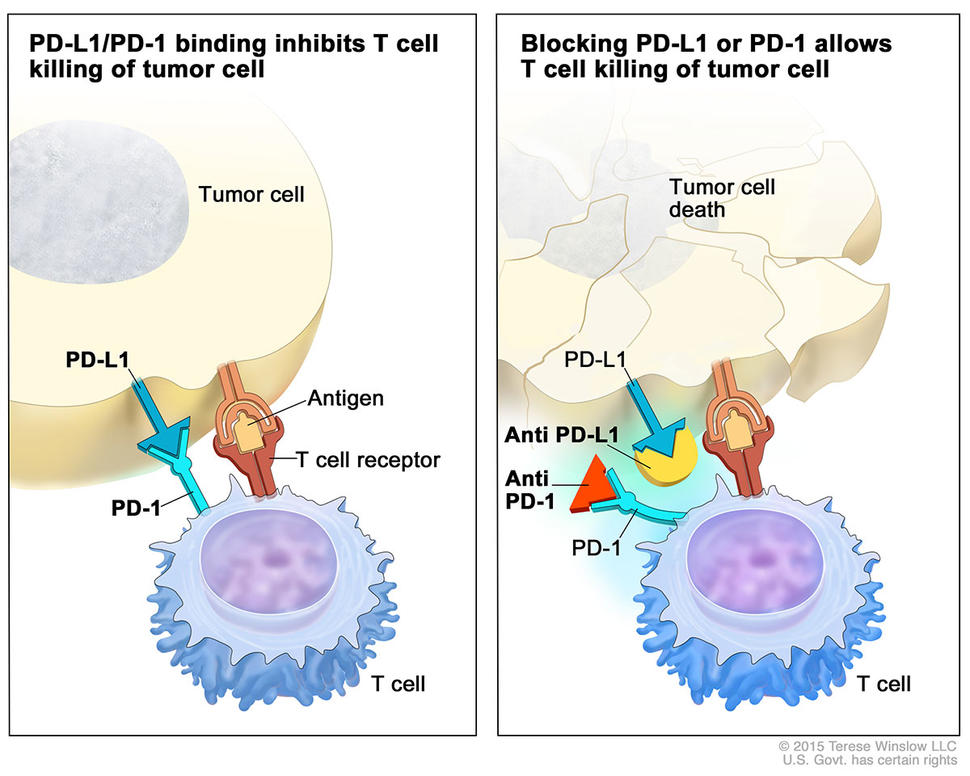
Yes, that's a great way to explain it. But it's also really important to understand that immune checkpoint inhibitors are not directed against the tumor. They have no direct antitumor activity, so if there is no underlying immune response against a tumor prior to using these drugs they won’t have any effect. They only take the brakes off of the immune response if you already have some type of immune response to begin with.
But it’s hard to tell if there is an existing immune response, and if an individual patient will respond, right?
Yes, we're still only getting responses in a minority of patients who receive these therapies. But one of the factors driving the excitement around these agents has been the deep, durable—that is, long-lasting—and rapid responses to anti-PD-1 agents like pembrolizumab and nivolumab (Opdivo®). And we’re seeing responses to these therapies across a broad range of tumor types, but, again, patients need to have an underlying immune response.
One possible way to get that underlying response is if there are many mutations in the tumor. Certain tumors—lung, melanoma, bladder—are more likely to have many mutations and so seem to be more likely to generate an immune response.
In most patients with colorectal cancer, for example, we really don’t see strong immune activity. However, we now know that if colorectal tumors have something called microsatellite instability, which produces many mutations, there is increased immune recognition of the tumor. That's where we have seen checkpoint inhibition work in patients with colorectal cancer.
These treatments are all intravenous medications. What is the administration schedule like?
Checkpoint inhibitors, all of which are monoclonal antibodies , are typically given over a period of one hour, every 2 to 3 weeks. Patients remain on the treatment as long as their disease has not progressed and as long as they are not having side effects that would require them to stop.
What are the tumor response rates to these therapies and what are the side effects like?
Tumor response rates typically range anywhere from 15 to 25 percent. You may see higher numbers quoted, but that’s in selected patient populations. In bladder cancer, for example, it can be up to 40 percent, but that’s in patients who have high PD-L1 expression within the tumor.
When it comes to side effects, particularly with anti-PD-1 and anti-PD-L1 drugs, the side effect profile is less harsh than what’s been seen with anti-CTLA-4 checkpoint inhibitors. In general, fewer patients have severe adverse events and many patients have no side effects.
The first approved checkpoint inhibitor, ipilimumab (Yervoy®), targeted CTLA-4. But it seems like the momentum has shifted toward anti-PD-1 and anti-PD-L1 inhibitors. Why is that?
First, the side effect profile of anti-PD-1 and anti-PD-L1 drugs is much better. Patients tolerate them better, so they can remain on therapy longer.
In addition, with ipilimumab we rarely see rapid regressions. We often see progression, sometimes followed by regression. That's because the drug mostly works on T cells in the circulation, and this nonspecific activation may take time to activate enough of the antitumor T-cells to produce a response.
With the anti-PD-1 and anti-PD-L1 therapies, however, we’re seeing very rapid responses. That's because these drugs primarily act on primed T cells that are already at the tumor; their sleeves are rolled up and they're ready to go but they’re being blocked. Then we take the blinders off with these drugs and the T cells can now attack the tumor.
Those two factors are making a world of difference.
What about instances, like that of former President Carter, where patients have complete responses to treatment. In most cases, do those responses last?
The majority of responses we see with these drugs are still partial responses, at rates similar to what we see with chemotherapy and other targeted agents. But often the responses we see are very deep, 80 to 90 percent tumor shrinkage.
The more important thing—and the thing that’s blown everybody away—is how durable the responses are. Even the partial responses are very durable, compared with what we’re used to seeing with chemotherapy or targeted therapies like tyrosine kinase inhibitors.
What we are seeing in some cases are functional cures. Not in everybody, of course. But, for example, approximately 20 percent of the patients with melanoma who were first treated with ipilimumab are still alive up to 10 years after the start of treatment.
For patients who have survived for at least 3 years after starting ipilimumab, the vast majority will still be alive at 10 years. Now, melanoma patients are often younger, so there are not as many competing causes of death. But those are still intriguing numbers, and they suggest that if you get a benefit, it will often be prolonged.
What does the future hold for checkpoint inhibitors?
There need to be several areas of focus, all of which can be done in parallel.
Identifying biomarkers that predict response is one important area. For example, one thing we have found is that patients have a higher chance of responding if they have high levels of PD-L1 expression in the tumor microenvironment. That represents a "footprint" of an activated immune response. But we need to be able to look at what’s happening in patients’ tumors at the time we are going to begin therapy, not biopsy samples that might be a year old.
Biopsies are good, but they can be quite invasive and, depending on where the tumor is, may be dangerous. So we’re starting to see if we can measure PD-L1 expression on tumor cells circulating in the blood.
We’re also testing imaging modalities to identify tumors that are inflammatory [a marker of an immune response]. So if you can find a way to safely and specifically image for inflammation within tumors, that would be a big advance. If we see inflammation, then maybe we can treat with the checkpoint inhibitor. If we don’t see it, perhaps we can add a therapy that can cause an immune response at the tumor site and then follow up with a checkpoint inhibitor.
Developing combinations, including those with other checkpoint inhibitors, will also be important and we’re already starting to see those trials. I think that we’re going to find that as we interrogate the tumor microenvironment, we can start to develop more personalized approaches to treatment, where we determine that one patient requires a cocktail of therapies while another just needs a checkpoint inhibitor.
Can these drugs be used in earlier-stage disease? Are there enough mutations in tumors at that point to establish an underlying immune response?
Absolutely. The mutational burden is already high by the time you have a clinically detectable tumor, and that mutational burden helps with developing the underlying immune response, but the immune system still can’t do its job.
Because the side effect profile is so minimal compared with chemotherapy and other radiation, and because of their rapid response, these drugs should be tested in the first-line setting for many cancers and I think they will be effective.
The real advances will be building on those strong, durable responses that we’re currently seeing in 20 percent of patients and making it 40 percent, 60 percent, and so on. That’s where combinations of therapies will come in. Eventually, I think we’ll get there.