Colorectal Cancer Survival Linked to Primary Tumor Location
, by NCI Staff
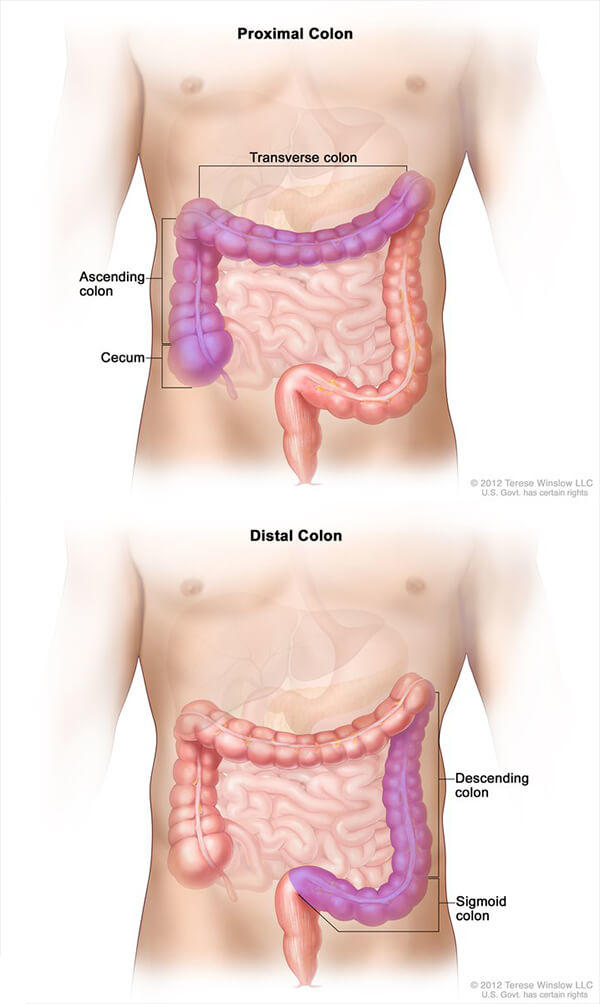
In patients with metastatic colorectal cancer, the location in the colon where the tumor originated appears to strongly influence how long patients live, according to a new study.
The study—a retrospective analysis of data from a large NCI-funded phase III clinical trial—found that patients whose cancer originated in the left side of the colon (distal colon) lived more than a year longer after initial treatment than patients whose disease originated in the right side of the colon (proximal colon).
The study also linked tumor location to the likelihood of benefit from specific targeted therapies used to treat patients with colorectal cancer. Patients with left-sided primary tumors are more likely to benefit from the targeted therapy cetuximab (Erbitux®), the study suggests, whereas patients whose tumors originated in the right colon may be better off receiving bevacizumab (Avastin®).
The findings support the idea that “colorectal cancers that originate on the right side should be treated differently than colon cancers originating on the left,” said the study’s lead investigator, Alan Venook, M.D., of the University of California, San Francisco.
Other investigators, however, caution that further confirmation is needed from prospective studies that provide more information about the underlying biology that may account for the observed survival differences.
Dr. Venook and his colleagues reported their study findings at a May 18 American Society for Clinical Oncology (ASCO) press briefing in advance of their presentation early next month at the ASCO annual meeting in Chicago.
Building on Earlier Evidence
In CALGB/SWOG 80405, the clinical trial on which this retrospective analysis was based, patients were randomly assigned to primary treatment with either cetuximab or bevacizumab in combination with one of two commonly used chemotherapy regimens (FOLFIRI or FOLFOX). Results from the trial, presented in 2014, showed no difference in overall or progression-free survival between the treatment groups.
Several previous studies had suggested, however, that tumor location in the colon may be an important clinical and biological consideration, Dr. Venook explained during the briefing. But uncertainty about its influence remained, he added, “because of the nature of the studies and [their] small numbers.”
The more than 44,000 biological samples collected in the CALGB/SWOG 80405 trial (from tumors and normal tissue), provided an ideal resource to further investigate differences between left- and right-sided colorectal cancers, Dr. Venook said.
Therapy Choice May Matter
The researchers focused their analysis on the 971 patients in the trial whose tumors had the wild-type, or normal, form of the KRAS gene. (Patients whose tumors have wild-type KRAS are more likely to respond to cetuximab than patients whose tumors have mutations in KRAS, who don’t respond at all to the therapy.)
Although patients whose tumors originated in the left colon lived substantially longer after treatment than patients whose tumors originated in the right colon, the survival improvement for patients treated with cetuximab was more pronounced. And patients with right-sided tumors had better outcomes when treated with bevacizumab. (See the table below.)
| Patients | Left-Sided Tumors | Right-Sided Tumors |
|---|---|---|
| All Patients | 33.3 months | 19.4 months |
| Patients Treated with Cetuximab | 36 months | 16.7 months |
| Patients Treated with Bevacizumab | 31.4 months | 24.2 months |
The survival difference associated with the two drugs was a “dramatic finding,” Dr. Venook said. “I think that was really surprising to most … or all of [the study investigators], given our belief beforehand that [the treatment drug] was not likely to really make a big difference.”
A ‘Surrogate Marker’
Findings from another study to be presented at the ASCO meeting also found that patients with advanced forms of colorectal cancer (stages III and IV) that originated in the left colon survived longer than patients whose tumors originated on the right side.
Although findings from both studies support the idea that primary tumor location is important, Dr. Venook said that location may well be “a surrogate marker” for underlying biological differences between tumors.
Several other studies support the notion that colorectal cancer is “not just one uniform disease,” he continued. For example, based on analyses of genomic data from numerous studies, an international consortium of researchers recently identified four molecular subtypes of the disease, each with distinct molecular features. And results from another study to be presented at the ASCO annual meeting identified molecular characteristics that are seen more frequently in right-sided colorectal tumors.
Given the size of the large bowel, “it’s not surprising that molecular differences exist across it,” said Austin Duffy, M.D., of the Thoracic and Gastrointestinal Oncology Branch in NCI’s Center for Cancer Research.
Dr. Duffy commended the researchers “for highlighting a fairly basic factor [tumor location in the colon] that is usually ignored.” He argued, however, that more data on the molecular makeup of colorectal tumors, relative to their specific location, is needed before any firm conclusions on treatment choice can be reached.
The results and conclusions seen with cetuximab treatment “are largely driven by the differences in the KRAS wild-type population,” Dr. Duffy said. The analysis was confined to the more common KRAS mutations, he continued. Information on other mutations, including less common KRAS mutations associated with lack of response to cetuximab, as well as in the NRAS and BRAF genes, would help to “complete the picture, as these would have also influenced the results,” he said.
Dr. Venook’s research team is performing molecular analyses of patient tumor samples from the CALGB/SWOG 80405 trial and expects to complete their work in the coming months, he said.