Multiple mRNA Vaccines Show Promise for Treating HPV-Related Cancers
, by Edward Winstead
For more than a decade, researchers have been developing vaccines that use messenger RNA (mRNA) to treat cancer. Some of these investigational vaccines have been tested in people with cancer, though none have yet been approved by the Food and Drug Administration.
As the research has progressed, various approaches for building these vaccines, such as changing the composition of the mRNA, have been tested. But few studies have directly compared mRNA cancer vaccines produced using different technologies.
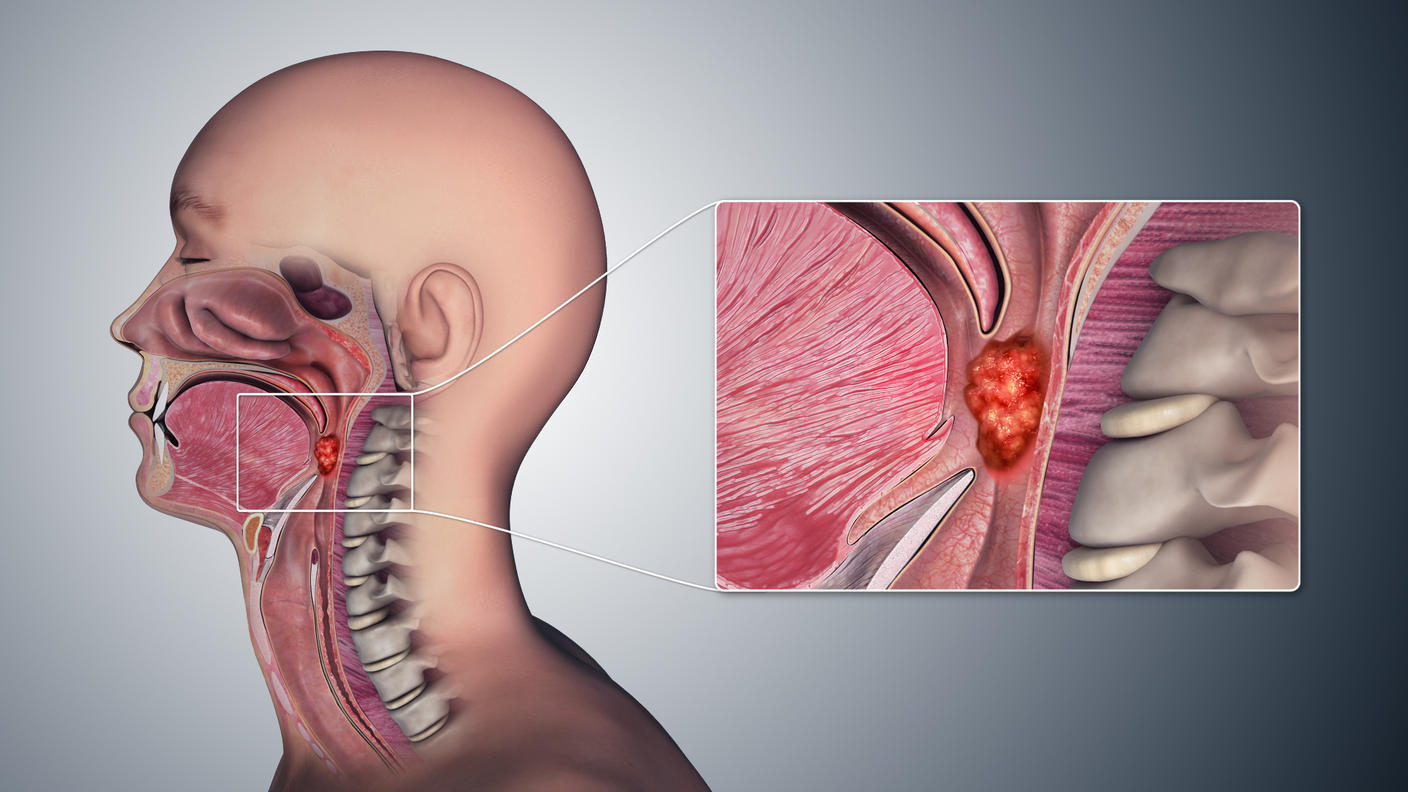
A new study in mice has done just that. Researchers developed and compared three mRNA vaccine formulations designed to treat cancers caused by human papillomavirus (HPV) infections, such as cervical and some head and neck cancers.
In the head-to-head comparison, all three experimental vaccines eradicated HPV-related cancers in the mice, the researchers found.
In fact, a single dose of any of the three vaccines made tumors in the mice go away, Jamile Ramos da Silva, Ph.D., of the University of São Paulo, and her colleagues reported in Science Translational Medicine on March 3. In most of the mice, the cancers did not come back.
Although there are vaccines to prevent HPV infections and the cancers that result, these vaccines do not treat cancers caused by existing HPV infections. And for people who develop HPV-related cancers, more treatment options are needed.
Previous studies have suggested that mRNA vaccines might be effective against HPV-related cancers, and some of these vaccines are being evaluated in people with these cancers.
Building on this work, the new study compared two vaccines with different formulations of nonreplicating mRNA—one used modified mRNA and the other unmodified mRNA. The third vaccine in the study was a self-amplifying mRNA vaccine.
Each vaccine delivers mRNA instructions for making an engineered protein called gDE7. Cells that take up the vaccine produce this protein, which trains the immune system to recognize and attack cells that have a protein from HPV-16 called E7. This protein interacts with other proteins in cells to drive the progression of cancer.
The gDE7 protein is a combination of E7 and a herpes simplex virus 1 protein called glycoprotein D. The herpes virus protein helps boost the immune response against E7.
Not only were all three vaccines effective, but they also outperformed two other treatments—a DNA-based gDE7 vaccine and a gDE7 protein-based vaccine—in the same mouse model, the researchers reported.
“We learned from our studies that all three mRNA platforms can potentially be used in future studies in humans,” said study co-leader Norbert Pardi, Ph.D., of the University of Pennsylvania Perelman School of Medicine, who investigates mRNA vaccines.
The benefits of comparing mRNA vaccine strategies
The effectiveness of mRNA vaccines in preventing severe COVID-19 has increased interest among researchers and companies in developing mRNA vaccines to treat different types of cancer. But research comparing the main approaches currently being used to create mRNA cancer treatment vaccines has been lacking, Dr. Pardi said.
John Schiller, Ph.D., of NCI’s Center for Cancer Research, whose research contributed to the development of preventive HPV vaccines, applauded the researchers for conducting the study.
“It’s always good to compare different strategies head to head,” said Dr. Schiller, who was not involved in the study. “Many researchers tend to focus on their own work, and they don’t want to find out that their strategy is not as good as another strategy.”
In the unmodified nonreplicating mRNA vaccine, the building blocks, or nucleosides, of mRNA have not been altered. In the other nonreplicating mRNA vaccine, the uridine component is replaced with a naturally occurring nucleoside called N1-methylpseudouridine that helps hide mRNA from the immune system.
The self-amplifying mRNA vaccine includes the instructions for making gDE7 and producing an enzyme in cells that interacts with the cell’s machinery to produce many copies of the original strand of mRNA.
“Researchers began to use self-amplifying mRNA vaccines because they realized that, at least in small animal studies, very low doses of vaccine induced very robust immune responses,” said Dr. Pardi.
New treatments needed for HPV-related cancers
Dr. Pardi partnered with Luis Carlos Ferreira, Ph.D., who directs the Institute of Biological Sciences of the University of São Paulo and has expertise in mouse models of HPV-related cancers, to develop the study.
Dr. Ramos Da Silva spent a year in Dr. Pardi’s lab to help develop and create the three mRNA vaccines.
The researchers focused on HPV-related cancers in part because current treatments for these diseases are not optimal. For example, while many cervical cancers initially respond to standard treatments, these cancers often stop responding to therapies at some point, and the disease frequently comes back. And treatments for head and neck cancers, many of which are caused by HPV, can be disfiguring and debilitating.
What’s more, many people who will be diagnosed with HPV-related cancers in the next few decades would have been past the age at which vaccination is recommended when the preventive vaccines became available or lacked access to the vaccines.
Studying how the vaccines affect the immune system
The mRNA in all three vaccines is encapsulated in lipid nanoparticles, which form a protective coating and facilitate the cellular uptake of the mRNA. Protection is needed because otherwise the extracellular RNases (RNA degrading enzymes) would destroy mRNA molecules.
Mice in the study were implanted with E7-expressing cancer cells that went on to develop tumors. The animals were then treated with one of the three mRNA vaccines. “Each one eradicated established early- and later-stage tumors in the mice,” said Dr. Pardi.
“The results from a single dose of the three new vaccines are impressive, although my optimism is tempered by the fact that other approaches that were effective in this mouse tumor model were not similarly successful in subsequent clinical trials,” said Dr. Schiller.
The researchers also analyzed how the vaccines activated the immune system. All three stimulated antigen-specific immune cells called CD8-positive T cells to attack the tumor cells. CD8-positive T cells are the primary immune cells responsible for killing infected or abnormal cells.
There were “slight differences” in the vaccines’ abilities to stimulate immune responses against established tumors, the researchers noted. But even with these differences, each vaccine worked well and protected the cancers from coming back in most of the mice.
“Looking at immune responses to mRNA vaccines did not necessarily predict how well the vaccines worked to get rid of the tumor,” Dr. Schiller cautioned. “We still don’t really understand all of the components of a complex immune response that lead to the eradication of a tumor.”
Clinical trials of mRNA vaccines: Ongoing and more to come
The new study, Dr. Schiller added, highlights the value of head-to-head comparisons of potential treatment strategies for HPV-related cancers.
Dr. Pardi agreed. When he and his colleagues began their study in 2019, it was not clear which type of mRNA treatment vaccine they should pursue. Now, they hope to test all three approaches in people, and are currently planning the clinical trials.
In the meantime, mRNA vaccines are being evaluated in people with HPV-related cancers. One trial, for example, is testing a personalized mRNA vaccine in combination with an immune checkpoint inhibitor in patients with advanced head and neck cancer. The vaccine, mRNA-4157, is manufactured by Moderna.
Another trial is evaluating a different mRNA vaccine against HPV-related head and neck cancer. This study will combine the vaccine, BNT113, with the immune checkpoint inhibitor pembrolizumab (Keytruda). BioNTech is the trial's lead sponsor.