December 2015 - Cancer Currents Blog
-
Approval of Osimertinib and Necitumumab Increases Lung Cancer Treatment Options
The FDA has approved two targeted therapies, osimertinib (Tagrisso™) and necitumumab (Portrazza™), for the treatment of some patients with advanced lung cancer.
-
Cancer Can Lead to Financial Hardship, Study Confirms
A substantial number of working-age cancer survivors report material or psychological financial hardship associated with their cancer.
-
What's New on NCI’s Websites?
NCI is constantly publishing new information on its websites, so periodically we provide updates on new content of interest to the cancer community.
-
Checking In on Cancer Checkpoint Inhibitors
NCI’s Dr. James Gulley discusses cancer checkpoint inhibitors, their impact on patient care, and future directions for these therapies.
-
New Drug for Pediatric Neuroblastoma Shows Promise in Preclinical Studies
An international research team has identified a possible new treatment target for pediatric neuroblastoma and is planning an early-stage clinical trial of a drug that inhibits this target.
-
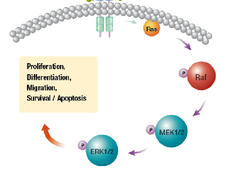
FDA Approves Cobimetinib as Part of Drug Combination for Advanced Melanoma
The FDA has approved the targeted drug cobimetinib, in combination with another targeted therapy, to treat patients with advanced melanoma whose tumors have specific genetic mutations.
-
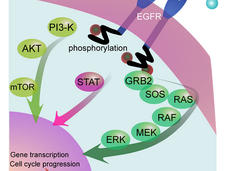
Two Small RNAs, Often Missing from Cancer Cells, May Suppress Tumors
Two small RNAs that are frequently deleted from cancer cells may help suppress tumors by interacting with RAS proteins, a new study suggests.
-
Precision Medicine Trials for Cancer: A New Era
NCI is designing smaller, quicker, and smarter clinical trials that incorporate the principal tenets of precision medicine.
-
Study Sheds Light on Role of Inherited Mutations in Childhood Cancer
In the most comprehensive study of its kind conducted to date, more than 8 percent of children with cancer were found to have inherited genetic mutations associated with a predisposition to the disease.
-
FDA Approves Trabectedin to Treat Two Types of Soft Tissue Sarcoma
The FDA has approved trabectedin for patients with advanced liposarcoma and leiomyosarcoma whose cancer has progressed after prior treatment.
-
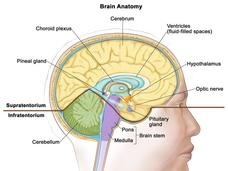
Patient with Brain Tumor Responds Dramatically to Targeted Therapy
A patient with a papillary craniopharyngioma saw his tumor shrink more than 80 percent after treatment with a targeted drug that inhibits a mutant form of the BRAF protein.