November 2016 - Cancer Currents Blog
-
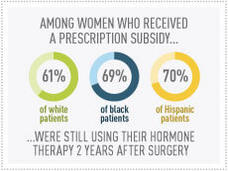
Prescription Subsidies Reduce Breast Cancer Treatment Disparities
A new study has found that subsidies for prescription drugs can improve the use of adjuvant therapy in women with early-stage breast cancer and help reduce disparities in the use of these proven treatments among black and Hispanic women.
-
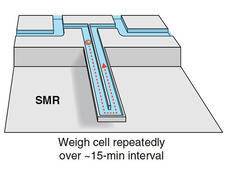
Device Uses Cancer Cells' Mass to Predict Response to Treatment
Scientists have designed a device that measures the mass of single cancer cells to predict how cells respond to drug treatment. The technology could potentially help clinicians determine whether a drug effectively kills cancer cells.
-
NCI’s Office of Cancer Survivorship: Celebrating 20 Years, Planning for Continued Progress
Dr. Julia Rowland, director of NCI’s Office of Cancer Survivorship, on the office’s 20th anniversary and efforts to ensure continued progress in survivorship research and improve survivors’ lives.
-
More Immunotherapy Options Approved for Lung Cancer
The FDA has approved atezolizumab and expanded the approval of pembrolizumab for some patients with non-small cell lung cancer.
-
Connected Health: An Important Tool for Making Progress against Cancer
A new report from the President’s Cancer Panel outlines how connective technologies can promote cancer prevention, enhance patients’ treatment experience, and accelerate progress in cancer research.
-
CDC Updates Recommendations on HPV Vaccine
Updated CDC recommendations advise those younger than age 15 need only two doses of the HPV vaccine instead of three.
-
Symposium Brings Rising Global Cancer Burden into Focus
An NCI Cancer Currents blog post about the NCI Global Cancer Research Symposium, which examined ways of increasing research collaboration, particularly in cancer prevention and screening, to reduce the cancer burden.
-
Olaratumab Approved to Treat Advanced Soft Tissue Sarcoma
The Food and Drug Administration (FDA) has granted accelerated approval to olaratumab (Lartruvo®) for the treatment of some patients with soft tissue sarcoma.