Extra or Missing Chromosomes May Help Cancer Cells Survive Treatment
, by Nadia Jaber
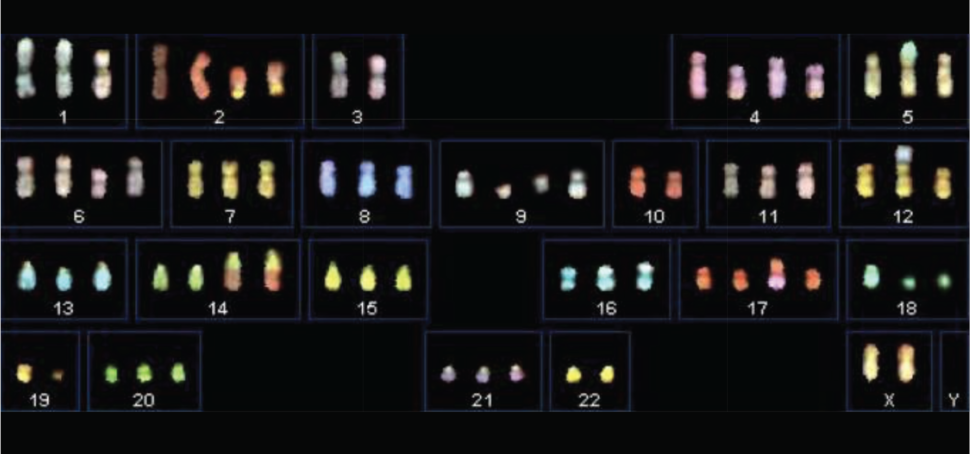
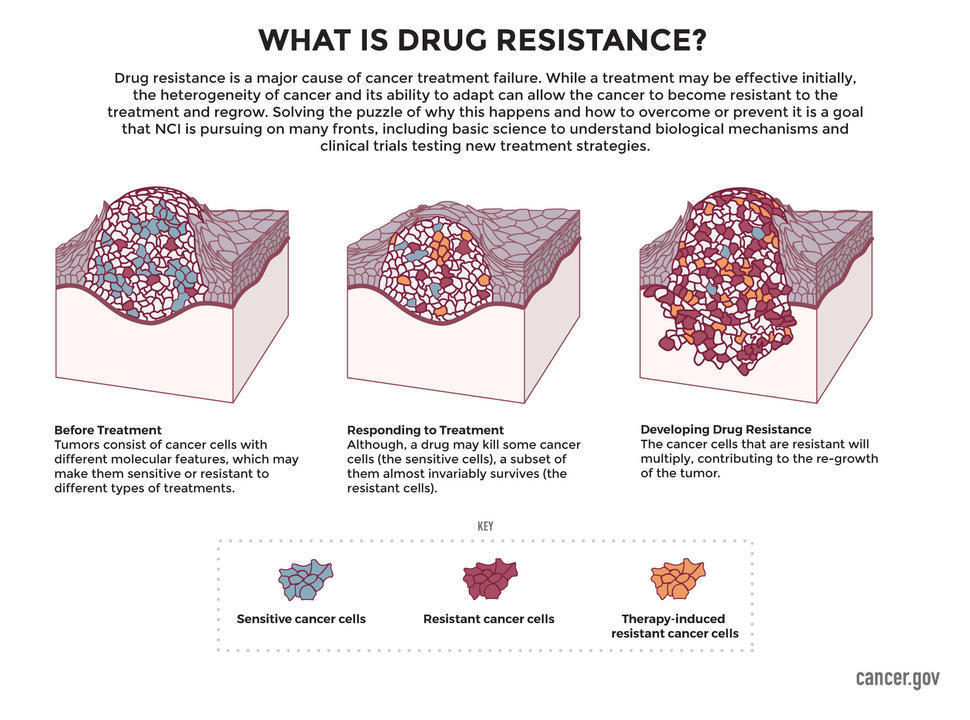
Around 90% of tumors have cancer cells with extra or missing chromosomes—a phenomenon known as aneuploidy. Despite this frequency, scientists have struggled to understand whether aneuploidy is harmful or helpful to cancer cells. But a new NCI-funded study has shown that the gain or loss of chromosomes may actually benefit cancer cells, particularly when they are exposed to cancer treatments.
“There wasn’t a good explanation as to why cancer cells would have such a high frequency of aneuploidy,” said Devon Lukow, a graduate student at Cold Spring Harbor Laboratory in New York and lead researcher of the study. There were some theories, including that aneuploidy might help tumors better adapt to stressful conditions, he added.
The new study, published August 4 in Developmental Cell, found evidence to support that idea. In lab experiments, aneuploidy helped human cancer cells survive treatment with cancer drugs like chemotherapy and targeted therapy. What’s more, the cells that thrived tended to have the same pattern of extra or missing chromosomes.
A separate research team in Europe found similar results when they examined human cancer cells with aneuploidy. Their study was also published August 4 in Developmental Cell.
“This could turn out to be a common mechanism of drug resistance and might help us identify ways to counteract that resistance,” said Keren Witkin, Ph.D., of NCI’s Division of Cancer Biology, who was not involved in the study.
When Cancer Resists Treatment
Most cells in our bodies have 46 chromosomes. When a cell is getting ready to divide, it makes a copy of each chromosome. The chromosomes are then paired up and evenly split between the two resulting cells.
But sometimes when cells—especially cancer cells—divide, the chromosome pairs separate unevenly, leaving one cell with more chromosomes than the other. The resulting aneuploidy generally wreaks havoc on the cells, causing widespread problems with metabolism, protein production, and growth. So why is it so common in tumors?
Lukow and his colleagues reasoned that cancer cells might use aneuploidy to circumvent treatments like chemotherapy. Although cancer treatments can be effective, most eventually stop working because cancer cells find ways to resist the effects of the treatment. Cancer cells use a variety of tactics to evade anticancer treatments, and scientists are still learning just how sneaky cancer can be in that regard.
To test their idea in the lab, the researchers exposed human cancer cells to a chemical that causes aneuploidy by disrupting chromosome separation during cell division. For comparison, the team left some cells untreated so that their chromosome number stayed the same.
In the absence of any cancer treatment, melanoma cells treated to become more aneuploid grew slower than those that were untreated. But after several days of exposure to vemurafenib (Zelboraf), a targeted therapy used to treat melanoma, the cells with more aneuploidy survived and grew faster.
Melanoma cells with aneuploidy also survived treatment with the chemotherapy drug paclitaxel. And aneuploid colorectal cancer cells were able to withstand both vemurafenib and paclitaxel treatment.
Likewise, the European research team reported that several different types of human cancer cells with aneuploidy had a survival advantage over cells without it when exposed to targeted therapies or chemotherapies.
Small DNA changes called mutations are often the culprit for drug resistance, explained Dr. Witkin. But when both research groups analyzed the DNA of different drug-resistant cancer cells, they couldn’t find any mutations that accounted for the drug resistance.
“This is a new way to think about how drug resistance occurs—not just by mutating genes, but by changing the number of copies of a gene,” Dr. Witkin said.
To see whether aneuploidy was linked with therapy resistance in people with cancer, Lukow and his colleagues turned to The Cancer Genome Atlas (TCGA), a collection of genetic and clinical data from more than 20,000 people with cancer.
In line with other study findings, the researchers noticed that people with cancer cells that had more chromosome alterations did not live as long as those whose cancer cells had fewer chromosome alterations. In addition, people with more chromosome alterations in their cancer cells were more likely to have their cancer come back after treatment.
A Pattern of Aneuploidy Emerges
Next, Lukow and his colleagues took a closer look at the pattern of extra and missing chromosomes in cancer cells that had become resistant to drug treatment.
Before treatment with vemurafenib, the aneuploid melanoma cells had a random assortment of extra or missing chromosomes. But after 3 weeks of treatment, nearly all of the cells had the same pattern of aneuploidy, namely extra copies of chromosomes 11 and 18. The researchers saw the same pattern—extra copies of chromosomes 11 and 18—each time they repeated the experiment.
Melanoma cells that developed resistance to paclitaxel also had a consistent pattern of aneuploidy, but they tended to lose chromosomes 16, 19, and 20. And colorectal cancer cells that were resistant to vemurafenib often gained an extra copy of chromosome 7.
These findings “suggest that the aneuploidies which can drive resistance are very context dependent,” meaning it depends on the type of cancer and drug treatment the cells are exposed to, Lukow said.
The European researchers also noticed consistent patterns of aneuploidy in cancer cells that had become resistant to treatment. For instance, they found that lung cancer cells that had become resistant to the chemotherapy drug topotecan tended to lose chromosomes 5 and 18 and gain an extra copy of chromosome 22.
What’s happening is a kind of survival of the fittest, Lukow explained. During drug treatment, cells with chromosome changes that confer resistance continue to survive and grow. On the other hand, cancer cells with other chromosome changes that don’t confer resistance eventually die off.
Extra Copies of Resistance Genes
The finding that certain chromosomes were consistently gained or lost suggests that “maybe there’s something on those chromosomes that’s driving resistance,” Lukow said.
Dr. Witkin agreed, saying, “it’s not all of chromosome 11, it’s probably one gene in there that’s responsible, or a combination of two [genes].” But there can be thousands of genes in a single chromosome, so it can be tough to narrow down.
The European researchers think they may have found the needle in the haystack. They painstakingly searched through extra copies of chromosome 6 for genes that might confer resistance to topotecan in lung cancer cells.
Among the many thousands of genes they reviewed, MAPK13 and MAPK14 stood out because together they produce a protein called p38. And p38 is known to raise the level of BCRP, a protein pump that expels certain toxic drugs—including topotecan—out of cells.
Indeed, the researchers saw that topotecan-resistant lung cancer cells had higher levels of BCRP than lung cancer cells that were not resistant to the drug.
“Overall, both studies have added evidence to the long-held but seldom tested theory that increased levels of aneuploidy really do give an advantage, in multiple cell lines, against multiple cancer drugs,” wrote Nadeem Shaikh and Sarah McClelland in an accompanying commentary in Developmental Cell.
The studies' findings, Drs. Shaikh and McClelland added, emphasize "the critical need to truly comprehend how [chromosome instability] functions in tumors.”