Does Too Much Fructose Help Colorectal Cancers Grow?
, by Sharon Reynolds
A new study suggests that excess consumption of fructose can promote both obesity and colorectal cancer.
Conducted largely in mice, the study found that large amounts of the sweetener, which is present in both table sugar and high-fructose corn syrup (HFCS), increased how long both normal and cancer cells in the intestines live.
In normal mice, this increased cell survival led to more nutrients being absorbed, leading to weight gain. And in mice prone to develop cancer, the increased cell survival led the animals to develop larger tumors and more anemia, a common tumor-related complication.
Findings from the study, which was funded in part by NCI, appeared August 18 in Nature.
The researchers, led by Marcus Goncalves, M.D., Ph.D., of Weill Cornell Medicine, are launching human studies to see if a similar phenomenon occurs in people.
“But this mouse study is really important, because it establishes a molecular mechanism for why fructose can drive tumor growth,” said Kristine Willis, Ph.D., of NCI’s Division of Cancer Biology, who was not involved with the study.
How Could Fructose Increase Tumor Growth?
In 2019, Dr. Goncalves and his colleagues published a study showing that feeding HFCS to mice prone to developing intestinal tumors could increase the size and aggressiveness of colorectal tumors. They also found that blocking the uptake of that particular sweetener by the body’s cells could prevent such growth.
But the details of the “why” behind these results remained a mystery. In the new work, the researchers thoroughly examined what was happening on the cellular level in the intestines after HFCS consumption.
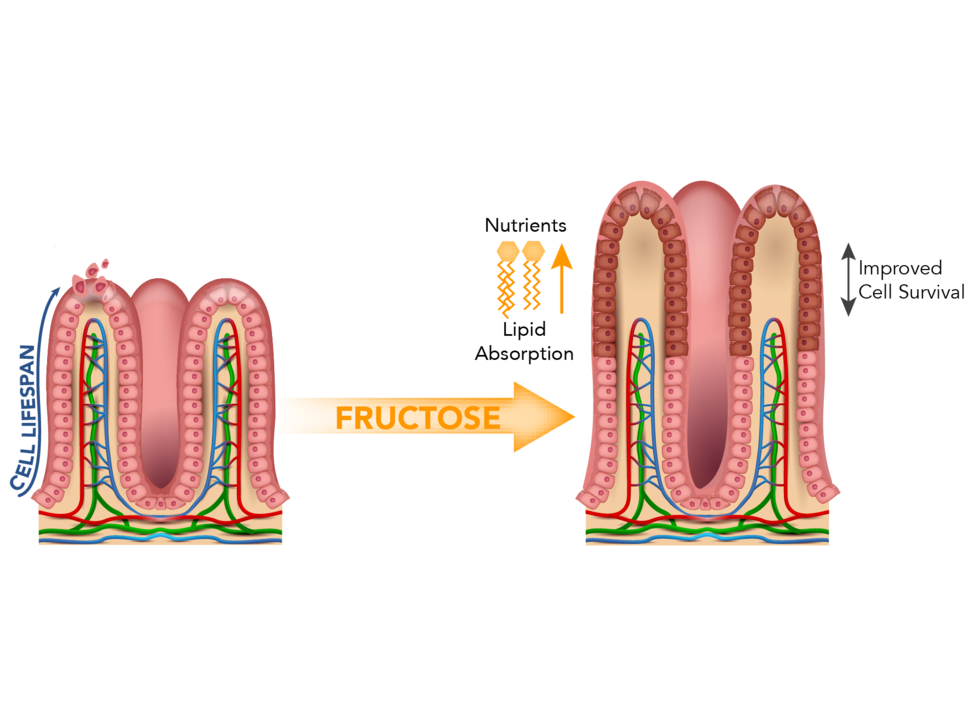
They first looked at what happened to intestinal cells in normal mice fed a diet including large amounts of HFCS. The team focused on villi: finger-like protrusions of the intestinal lining. Villi increase the surface area of the intestinal lining and maximize the amount of nutrients that can be absorbed after a meal.
The HFCS caused the villi to grow in length. Normal mice fed HFCS had villi that were around 25%–40% longer than those in mice that weren’t fed the sweetener.
When the mice were then fed a high-fat diet in addition to excess fructose, it caused them to gain far more weight than mice only fed a high-fat diet.
High-Fructose Corn Syrup Metabolism at the Cellular Level
Further experiments showed that the overabundance of HFCS kept the cells at the tips of the villi alive longer. Normally, the cells at the tip of a villus, where oxygen levels are lower (called hypoxia), die off. They’re then replaced by new cells that are produced at the base of the villus and migrate up to the tip.
But by increasing cell survival at the villi tips, and with new cells continuing to be made, the villi inevitably got longer, they found.
The researchers then turned to human colorectal cancer cell lines to see whether excess fructose would affect their ability to grow and survive in low-oxygen conditions. Hypoxia is a common occurrence in the middle of tumors and can limit their growth.
As with the mouse intestinal cells, the researchers found that adding fructose to colorectal cancer cells grown in a low-oxygen environment didn’t increase the rate at which they grew but helped the cells survive longer than cells to which fructose wasn’t added.
But why does fructose increase cell survival when oxygen levels are low? In further experiments in colorectal cancer cells, the researchers found that the sweetener caused a chain of cellular reactions, which resulted in lowered activity of a protein called PKM2.
PKM2 is involved in cellular metabolism. When PKM2 activity is low, as can happen under stressful conditions like hypoxia, cells adjust their metabolism so that they can stay alive.
So in order to survive, “the tumor wants to find the best way it can to inhibit PKM2,” explained Dr. Goncalves.
Several additional experiments showed that preventing fructose from inhibiting PKM2 could stop both villi and colorectal cancer cells from thriving in a low-oxygen environment. For example, when they gave HFCS-fed normal mice a drug called TEPP-46, which turns PKM2 back on, their villi didn’t elongate and they didn’t gain weight.
When the researchers then fed excess HFCS to mice predisposed to develop colorectal cancer, the mice developed bigger tumors and became more anemic—a complication associated with worse survival in both mice and people—than mice not fed the sweetener. Treating mice fed large amounts of HFCS with TEPP-46 slowed tumor growth.
The ability of a drug to activate PKM2 even in the presence of fructose, both stopping weight gain and slowing tumor growth stimulated by HFCS is exciting, said Dr. Willis.
“There’s huge potential there,” she said. “If this translates to humans, does that then represent a possible avenue of treatment for obesity as well as colorectal cancer?”
What Happens in Humans?
Dr. Goncalves and his colleagues are getting ready to launch a follow-up study in people. To date, it remains controversial whether substantial amounts of the HFCS that people consume in their diets can reach the colon, or whether most of it gets absorbed higher in the digestive tract, he explained. The mouse digestive tract is much shorter than the human version, relative to size, making it easy for HFCS to reach the colon in mice.
The team will recruit people with colon cancer scheduled to undergo surgery to have their tumors removed. Before surgery, they’ll eat a meal containing HFCS to which a heavy isotope of fructose has been attached. After the tumors are removed, this “label” will allow the researchers to measure how much of the fructose made it into the cancer cells.
This work will, the team hopes, help answer the question of whether fructose from HFCS can reach tumors in the human colon. If so, Dr. Goncalves said, “the next step is: how do we intervene?” he added. Several drugs that target PKM2 or related molecules are currently under development for other conditions.
“These could [potentially] be repurposed to reduce or shorten villi and hopefully prevent tumor growth,” he said.
Metabolism can differ vastly from one person to another, Dr. Goncalves explained. So many mechanisms—not just related to PKM2—likely drive both obesity and colorectal cancer.
“But exposure to fructose is one component of the diet that's modifiable, and we think plays a role,” he said. “There's still a lot more work to do in terms of [understanding] how big of an effect it has, and if we can use drugs to try to prevent it from happening.”
His lab also studies cachexia, a loss of body weight and muscle mass that often occurs in people with advanced cancer. People with cachexia have the reverse problem from that seen in this study: their villi shrink, Dr. Goncalves explained. That leads to the intriguing question of whether fructose could actually be a treatment for cachexia, to help people maintain weight and strength.
They’ve started studying this approach in mice. “If you give cachexic animals fructose, will they absorb [nutrients] better and ultimately maintain their body weight?” he asked.
The effects of fructose on the villi observed in the current studies were likely highly beneficial for animals in the wild and was for humans in the past, Dr. Goncalves explained. “When we didn't have agriculture and transport systems, we would get our local fruit supply only in a limited fashion, and [elongation of the villi] would help us to gain weight for the long winter coming up, when food may be harder to get,” he said.
“But now that we have continuous access to [sugar] all year long, our villi are constantly long, and we’re constantly hyper-absorbing [nutrients]. And that may underlie why [people] are gaining so much weight all the time,” he added.
Researchers are just starting to scratch the surface of how sugar metabolism affects cancer risk, growth, and treatment, Dr. Willis explained. Dietary guidelines universally recommend avoiding large amounts of added sugar in the diet, especially sugary drinks, but HFCS is almost impossible to avoid entirely in the modern world, she added.
However, a small amount is unlikely to cause long-term problems, and people shouldn’t panic, Dr. Willis explained. “If you have a slice of sandwich bread … you’re not doomed to get cancer,” she said.
And whole, unprocessed fruit—a natural source of fructose—remains a healthy part of any diet, added Dr. Willis. That’s because the amount of fructose consumed in fruit is lower than that in food containing HFCS. Fruit also contains healthy substances like fiber. “An apple is a completely different thing from a liter of soda,” she said.