Targeted Drug Combo May Change Care for Rare Brain Tumor Craniopharyngioma
, by Sharon Reynolds
People with a rare but devastating type of brain tumor called a papillary craniopharyngioma may soon have an effective new option for treatment, according to results from a small clinical trial.
This cancer often requires surgery, radiation therapy, or both to keep the disease at bay. But the results of the NCI-funded trial suggest that, for many, the combination of the targeted therapies vemurafenib (Zelboraf) and cobimetinib (Cotellic) may substantially delay, or even eliminate, the need for added treatments.
In the early-phase trial, treatment with both drugs substantially shrank tumors in 15 of the 16 participants. After completing the combination therapy, about half of the 16 trial participants chose not to have additional surgery or radiation therapy. And the tumors of many of the participants continued to respond to the treatment for a median of 22 months, according to study results published July 13 in the New England Journal of Medicine.
Study participants all had tumors driven by a change in a gene called BRAF. The drugs used in the study specifically shut down cellular activity driven by this change, called a BRAF V600E mutation.
The drug combination hasn’t yet received Food and Drug Administration (FDA) approval for treating craniopharyngioma. But based on earlier reports from the trial, it’s already making its way into everyday patient care.
“These data support treating newly diagnosed patients with papillary craniopharyngioma with [this combination],” said Evanthia Galanis, M.D., of the Mayo Clinic, who helped lead the trial, along with Priscilla Brastianos, M.D., of Mass General Cancer Center and Harvard Medical School.
“I think the results of this trial will dramatically shift the way these patients are treated,” said Dr. Brastianos.
The trial also raises many additional questions, explained Mark Gilbert, M.D., of NCI’s Neuro-Oncology Branch, who was not involved in the study. These include how long patients should receive these targeted drugs, whether other drugs that affect the same targets may work as well—or better—and what side effects might be seen in the long term.
Nevertheless, the results to date look promising, Dr. Gilbert said.
“If we can avoid major surgery and avoid or delay radiation, then we’re potentially preventing a compromise to quality of life [for these patients],” he said.
Craniopharyngiomas: Localized but dangerous
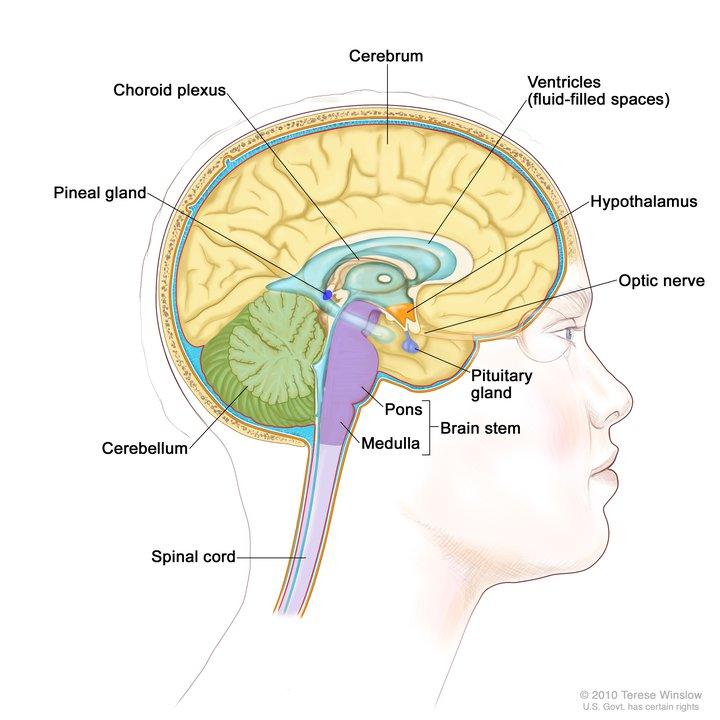
Craniopharyngiomas are brain tumors that usually form near the pituitary gland and the hypothalamus. They can occur in both adults and children. There are two subtypes, based on the brain tissue in which the tumors develop: adamantinomatous and papillary. Papillary craniopharyngiomas are more likely to occur in adults.
These tumors are very rare—only about 600 people in the entire United States will be diagnosed with craniopharyngioma in a given year.
They don’t spread (metastasize) elsewhere in the body. However, because of their aggressive local growth and location near delicate structures in the brain, craniopharyngiomas can cause devastating neurological symptoms. And their propensity to attach to vital glands, brain structures, and blood vessels makes it difficult to remove entire tumors with surgery.
That presents serious problems, because craniopharyngioma tissue left in place after surgery almost always starts to grow again. Radiation therapy is often used to kill tumor cells that can’t be removed surgically. But this treatment can also damage healthy brain tissue.
Both craniopharyngioma growth and treatment often cause life-altering health problems, including hormone imbalances, vision damage, cognitive difficulties, and diabetes.
With an eye to potentially reducing the side effects of treatment, researchers have been looking for specific genetic vulnerabilities in craniopharyngiomas that could be targeted with existing drugs. In 2014, a team led by Dr. Brastianos and Sandro Santagata, M.D., of Brigham and Women's Hospital, found that almost all papillary craniopharyngiomas have BRAF mutations and appear to depend on those gene mutations to grow.
This was very good news. Since 2011, both drugs that block mutant BRAF proteins and drugs that block other proteins that work in concert with BRAF have been approved for many cancer types, starting with melanoma and now including tumors as diverse as lung, colorectal, and thyroid cancers.
Running a flexible clinical trial for a very rare tumor
To test one of these drug combinations, vemurafenib and cobimetinib, in people with newly diagnosed craniopharyngioma, Dr. Brastianos, Dr. Galanis, and their colleagues joined up with the Alliance for Clinical Trials in Oncology, an NCI-sponsored clinical trials group, to search for patients with this rare tumor throughout the United States.
Between 2018 and 2020, the trial investigators recruited 16 participants from nine Alliance study sites. The original design of the trial was to give participants four 28-day cycles of treatment with the two drugs orally, then treatment with radiation or surgery as needed.
All but one of the participants had their tumor shrink. And the reduction in tumor size was substantial. The median reduction among the 15 participants who responded to treatment was more than 90%. Seven of these patients chose to have no further treatment until their tumors progressed.
Of these, six had no evidence of further tumor growth at a median of nearly 2 years. Several participants also elected to keep receiving anti-BRAF drugs after the planned four cycles.
“We started seeing these dramatic responses, and the treating physicians were coming to us and saying, ‘Is it okay if we delay surgery? Is it okay if we delay radiation?” said Dr. Galanis. “So we ended up modifying the study to allow patients to keep receiving the combination drug treatment given how impressive the response was, and how well patients were tolerating the drugs.”
The researchers estimated that, among all 16 participants, almost 60% wouldn't experience cancer progression for at least 2 years after treatment.
There was also some evidence that the combination treatment could shrink tumors enough to limit the damage from subsequent radiation therapy to areas of the brain that control critical functions, such as vision. Further analyses of these data are ongoing, explained Dr. Galanis. But if more limited radiation is needed, “that will mean less long-term side effects,” she said.
Vemurafenib and cobimetinib were not without side effects themselves. Fourteen of the 16 participants experienced a serious side effect, the most common being a severe rash. Three people had to stop treatment early because of side effects. The one participant who had no response to the drugs had stopped treatment after 8 days due to side effects.
Going forward, weighing the pros and cons of targeted therapy versus surgery or radiation, given the different characteristics of each patient’s tumor and their overall health, will be a major part of deciding what treatment to try first, Dr. Brastianos explained.
Can biopsy be avoided, too?
All patients who joined the trial had their tumor biopsied before receiving the two-drug combination, to confirm that their tumors had the required BRAF mutation.
But as part of the new study, the researchers also tested whether they could use a sophisticated blood test, called a liquid biopsy, to find BRAF V600E mutations. And in some participants, at least, this was possible.
If additional studies can improve the accuracy of these blood tests, it raises the intriguing possibility that some patients may be able to avoid a surgical biopsy, which carries its own risks, Dr. Gilbert said.
“If I had a patient with a tumor that looks very much like a craniopharyngioma [on imaging tests] and had a blood test positive for this mutation, I would [skip] a biopsy and just treat them with this [drug] combination,” he said.
As seen in this study, however, not everyone with a craniopharyngioma will have a blood test positive for BRAF, meaning that many people will likely still need their tumor biopsied for now, Dr. Gilbert cautioned.
Another unanswered question is how these drugs should be used in people who have already undergone surgery, radiation, or both for craniopharyngioma. “What do we do with patients who have received other types of treatments and are now faced with tumor recurrence?” Dr. Brastianos asked. An ongoing part of their trial, open at more than 70 hospitals across the country, is currently recruiting participants to answer that question.
And in the future, explained Dr. Galanis and Dr. Brastianos, researchers hope to develop therapies other than surgery and radiation for adamantinomatous craniopharyngiomas, which are more common in children than adults.
Like papillary tumors, almost all adamantinomatous craniopharyngiomas rely on a single gene mutation to survive and grow, in this case one called CTNNB1 (also known as beta-catenin). To date, drugs targeting this mutation have proven challenging to develop, given the potential side effects of targeting this pathway, explained Dr. Brastianos.
“So that’s a challenging target to go after, but one that we would love to conquer,” Dr. Galanis said.