January 2016 - Cancer Currents Blog
-
Opportunities in Cancer Nanotechnology: A Conversation with NCI’s Dr. Piotr Grodzinski
The director of NCI's Office of Cancer Nanotechnology Research discusses the Cancer Nanotechnology Plan 2015, as well as new developments and clinical opportunities in the field.
-
Video Intervention Helps Prepare Patients to Participate in Cancer Clinical Trials
Patients who took part in a tailored, video-based educational program had a better understanding of and fewer concerns about participating in clinical trials than patients who received text-based educational.
-
Novel Treatment Approach Shrinks Ovarian Tumors in Mice
Researchers have developed a new approach for treating tumors that express mutant versions of the p53 protein, which are present in more than half of all cancers, including an aggressive and common subtype of ovarian cancer.
-
Supporting Vice President Biden's Cancer Initiative
During his 2016 State of the Union address to Congress, President Obama tasked Vice President Biden with leading a new initiative to accelerate cancer research. This message from NCI Acting Director Doug Lowy states that the Institute stands ready to work with partners across the cancer research community on the initiative.
-
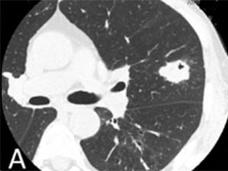
FDA Approves Alectinib for ALK-Positive Non-Small Cell Lung Cancer
The FDA has approved alectinib to treat patients with metastatic ALK-positive non-small cell lung cancer who have stopped responding to or who are unable to tolerate crizotinib.
-
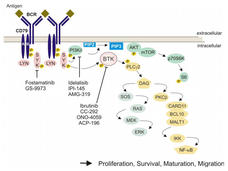
Two Drugs Show Efficacy against Common Form of Leukemia
Reports from early-stage trials of two new oral drugs provide hope for patients with high-risk chronic lymphocytic leukemia.
-
Driving New Approaches to Cancer Prevention and Early Detection
Researchers are pioneering new approaches to preventing and screening for cancer, and the management of very early-stage disease.
-
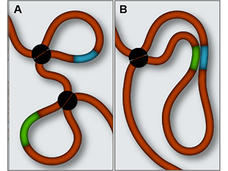
Genome Study Links Misfolded DNA to Brain Tumors
Changes in the packaging of DNA in the cell nucleus may activate growth-promoting genes that cause cancer.
-
Three New Therapies Approved for Multiple Myeloma
In November 2015, the FDA approved three new therapies for patients with multiple myeloma.