FDA Approves New Immunotherapy Drug for Bladder Cancer
, by NCI Staff
UPDATE: On March 8, 2021, Roche voluntarily withdrew the indication to use atezolizumab (Tecentriq) for the treatment of people with previously treated metastatic bladder cancer in the United States. The move follows the voluntary withdrawal by AstraZeneca of the indication to use durvalumab (Imfinzi) for people with locally advanced or metastatic bladder cancer that came back or continued growing despite prior treatment.
Both drugs, which work by blocking the PD-L1 protein, had received accelerated approvals from the Food and Drug Administration (FDA) based on tumor response rates in early clinical trials. The confirmatory trials required to transition the drugs to regular FDA approval did not meet their primary endpoints of improved overall survival. Details of the atezolizumab trial that led to the accelerated approval are described in the post below.
The Food and Drug Administration (FDA) on May 18 approved atezolizumab (Tecentriq®) for the treatment of some patients with urothelial carcinoma, the most common type of bladder cancer. The drug, which strengthens the body’s immune response against cancer, is the first new treatment approved for bladder cancer in two decades.
“This is very exciting news for patients with bladder cancer,” said Piyush Agarwal, M.D., head of the Bladder Cancer Section in the NCI Center for Cancer Research's (CCR) Urologic Oncology Branch, who noted that the approval would likely open “a wave of new clinical trials” for patients with bladder cancer. The FDA approved atezolizumab for patients with locally advanced or metastatic urothelial carcinoma that has gotten worse during or after treatment with platinum chemotherapy.
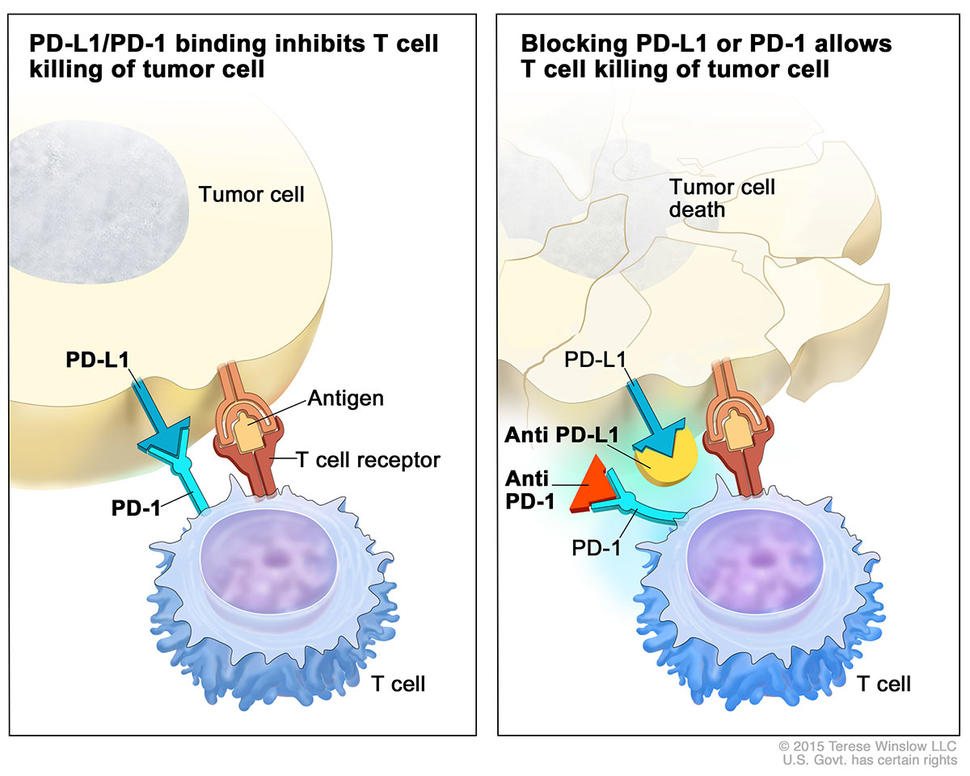
Atezolizumab belongs to a class of immunotherapy drugs known as checkpoint inhibitors. The drug prevents a protein called PD-L1 that is found on some tumor cells from binding to another protein, PD-1, on immune cells. The binding of these “checkpoint” proteins suppresses the immune response. By blocking this interaction, checkpoint inhibitors “release the brakes” on the immune system, allowing immune cells to attack tumors.
“Atezolizumab is the first PD-L1 inhibitor approved by the FDA for cancer, and it happens to be for bladder cancer,” said Andrea B. Apolo, M.D., who heads the Bladder Cancer Section in CCR's Genitourinary Malignancies Branch. Until now, she added, patients whose cancers did not respond to chemotherapy had few, if any, treatment options.
Evaluating Atezolizumab
The approval of atezolizumab was based on a study of 310 patients with locally advanced or metastatic urothelial carcinoma whose cancers had worsened during or after treatment with platinum-containing chemotherapy or within 12 months of receiving platinum-containing chemotherapy, either before or after surgery. All patients in the trial received atezolizumab.
Approximately 15% of patients had at least a partial shrinkage of their tumors, and this effect lasted from at least 2.1 months to more than 13.8 months, the study authors reported.
The most common side effects of treatment with atezolizumab were fatigue, decreased appetite, nausea, urinary tract infection, fever, and constipation. The therapy also may cause infection and serious immune system-related side effects.
In the trial, increased PD-L1 expression in patients’ tumors was associated with response to atezolizumab. Among the trial participants whose tumors were classified as “positive” for PD-L1 expression, 26% experienced a tumor response, compared with 9.5% of the participants whose tumors were “negative” for PD-L1 expression.
At the same time that it approved atezolizumab, the FDA also approved a test, called Ventana PD-L1 (SP142), to measure PD-L1 expression on patients’ tumor-infiltrating immune cells.
Dr. Apolo noted that the test is not required for patients to receive atezolizumab, and she cautioned that patients whose tumors are classified as negative for PD-L1 expression might still respond to the therapy. “I don’t think we’re ready to rule out patients as candidates for the drug based on the results of the test,” she commented.
Exploring the Immune Response
Currently, one of the main treatments for non-muscle-invasive bladder cancer is an immune-based therapy called BCG (bacillus Calmette-Guérin). Patients are given a weakened form of the bacterium Mycobacterium bovis in a solution to stimulate the immune system against cancer.
Although BCG is not new, researchers still have some unanswered questions about how the treatment works, noted Dr. Agarwal.
“It’s an exciting time,” Dr. Agarwal continued. “The most effective treatment for bladder cancer has been an immunotherapy. With the introduction of additional immunotherapies, we now have an opportunity to learn more about how BCG works.”
Based on preliminary results from clinical trials, researchers have known for several years that checkpoint inhibitors are active in some patients with bladder cancer. The field, Dr. Apolo explained, is now looking at ways to improve the response rate to immunotherapies and possibly to enhance the immune response in patients.
One strategy being explored in clinical trials is to test combinations of immunotherapies. This approach might be important for patients whose cancers do not respond to single agents or stop responding. In addition, future studies are likely to explore the use of atezolizumab at earlier stages in the disease, possibly as a first-line therapy in metastatic disease, and even in early disease states such as muscle-invasive and non-muscle-invasive disease, Dr. Apolo noted.