Can Immunotherapy Succeed in Glioblastoma?
, by NCI Staff
This post was updated on June 6, 2018.
Cancer treatments that work by engaging the immune system to attack tumors have proven to be effective against a growing number of cancers. One notable exception thus far, however, has been brain cancer, including glioblastoma, the most common type of brain cancer in adults.
Despite continued efforts over several decades to develop new therapies for glioblastoma, none has appreciably improved how long patients live. Most people with this type of brain cancer, in fact, survive for less than 2 years.
Researchers who study glioblastoma have been hopeful that immunotherapy might be able to succeed where other therapies have not. And in laboratory studies and human clinical trials, they are leaving no immunotherapy stone unturned.
Unfortunately, several immune-based treatments that looked highly promising in early-phase studies of patients with glioblastoma have not panned out in larger, phase 3 clinical trials. But even those failures have a silver lining, said Michael Lim, M.D., who directs the Brain Tumor Immunotherapy program at the Johns Hopkins University Sidney Kimmel Comprehensive Cancer Center.
“Just the fact that we have had some phase 3 [immunotherapy] trials in glioblastoma, where for years we had a hard time getting past phase 2 trials, is an encouraging sign,” Dr. Lim said.
There is undoubtedly optimism among researchers about the prospects for immunotherapy as an effective treatment for glioblastoma. At the same time, through their work in the lab and in the clinic, researchers are exploring a critical question: how to overcome the unique and daunting challenges presented by the brain and of glioblastoma itself.
“We’re talking about a disease that has been one of the hardest to treat in the history of oncology,” said Mark Gilbert, M.D., director of the Neuro-Oncology Branch in NCI’s Center for Cancer Research. And trying to develop and test immune-based treatments for glioblastoma, Dr. Gilbert continued, “is a whole new realm.”
From the Start, Many Challenges
When it comes to treating tumors in the brain, all cancer therapies must overcome one very serious obstacle: the blood–brain barrier.
This layer of tightly packed, specialized cells has the critical job of protecting the brain from threats, like viruses and toxins, that may be circulating in the bloodstream. But this protective blockade doesn’t distinguish between the good guys and the bad guys, so it can also prevent cancer drugs from reaching tumors.
And then there’s the matter of how the immune system operates in the brain, Dr. Lim said. “We’re understanding now that the immune environment of the brain is very different from other organs,” he explained.
For example, the brain appears to have a different mix of immune cells than other organs (there are many different types of immune cells, each with different responsibilities). For several reasons, Dr. Lim continued, the composition of immune cells in the brain doesn’t readily generate an immune response against tumors the way immune cells in other organs do.
Glioblastoma tumors also seem particularly adept at blunting an immune response. One recently published study, for instance, showed that many of the immune cells in and around glioblastoma tumors tend to be “exhausted”—that is, the molecular machinery the cells need to stimulate their activity is severely impaired.
Research has also shown that steroids, which are often used to control potentially dangerous brain swelling and manage treatment-related side effects in patients with glioblastoma, can interfere with the immune response.
As a result, said Dr. Gilbert, immunotherapy clinical trials may be “handicapped” from the start. He pointed to the first-ever phase 3 trial of an immune checkpoint inhibitor in glioblastoma, called Checkmate 143.
Results from the trial showed that the checkpoint inhibitor nivolumab (Opdivo) failed to increase how long patients with advanced glioblastoma lived compared to trial participants treated with the targeted therapy bevacizumab (Avastin). But approximately 40% of patients in the trial who received nivolumab were also taking steroids, Dr. Gilbert noted, “which means they may not have been able to mount an immune response [against their tumors].”
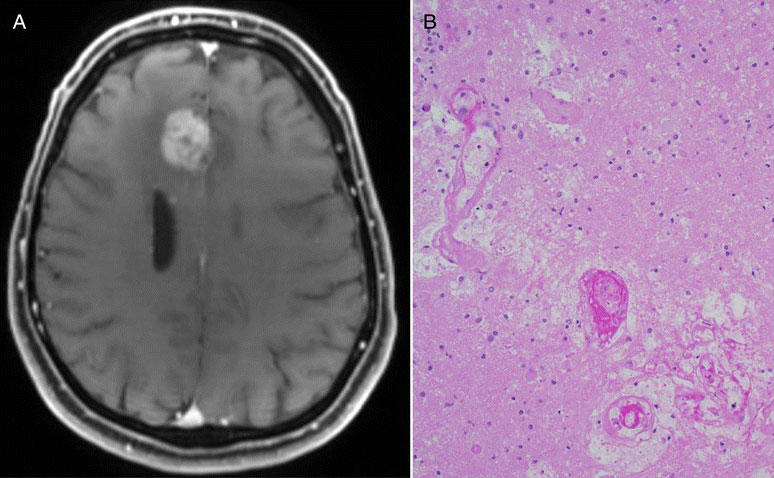
Even measuring whether an immunotherapy drug is working against glioblastoma tumors is difficult. Standard imaging techniques, such as MRI, that are used to assess whether tumors are growing or shrinking in response to treatment may not be sufficient in brain cancer, Dr. Gilbert explained.
As several small studies have demonstrated, with these imaging methods, it’s very difficult to differentiate between tumors that are growing despite treatment and those encased by inflammation caused by an active immune response—a phenomenon known as pseudoprogression. In the latter case, treatment may be prematurely stopped in a patient whose tumors may actually be responding to the therapy.
According to Hideho Okada, M.D., Ph.D., director of the Brain Tumor Immunotherapy Center at the University of California, San Francisco, all of these issues highlight an important, and perhaps underappreciated, point. It’s not necessarily the case, Dr. Okada said, that “we’ll be able to apply the same approaches to brain cancer that we do for other cancers and expect them to work the same way.”
Big Effort with Immune Checkpoint Inhibitors
Perhaps not surprisingly, checkpoint inhibitors are the immunotherapy approach being tested most widely in glioblastoma. Multiple checkpoint inhibitors have been approved by the Food and Drug Administration (FDA) for a growing list of cancer types, and—despite the disappointing results from the Checkmate 143 trial—a number of these drugs are being tested in early- and late-phase clinical trials in patients with glioblastoma.
Even as the approved uses for checkpoint inhibitors in cancer continue to expand, in most cancers only a modest proportion of patients respond to these drugs. In-depth analyses from clinical trials involving checkpoint inhibitors in patients with different cancer types, however, are identifying potential biomarkers that may show which patients are most likely to respond to these agents.
The same process may eventually play out for the different immunotherapy approaches in glioblastoma, said John Sampson, M.D., Ph.D., chair of the Department of Neurosurgery at Duke University Medical Center. “Hopefully, one of the things we’ll learn from [the ongoing trials] is whether there are biomarkers that can predict success,” Dr. Sampson said.
As in other cancers, in glioblastoma, checkpoint inhibitors are being tested in combination with other therapies, including other forms of immunotherapy. And there’s some evidence from animal studies that these immunotherapy combinations may be what’s needed to make checkpoint inhibitors work in glioblastoma.
Building on this evidence, NCI’s Neuro-Oncology Branch, for example, has launched several early-stage human trials testing combinations of immunotherapies.
Patients in these trials will be monitored for treatment response using standard imaging scans, Dr. Gilbert said. In addition, further steps will be taken to decrease the chances that a tumor response isn’t mistaken for tumor progression. Namely, he explained, there will be a greater use of surgery—which carries its own risks—to more accurately assess whether what appears to be disease progression on imaging is actually inflammation resulting from an immune response against the tumor.
Vaccines and Viruses
Another form of immunotherapy being investigated as potential glioblastoma treatments are therapeutic vaccines—that is, vaccines intended to treat an existing disease.
The first therapeutic vaccine to advance to a large phase III trial in patients with glioblastoma failed, with no improvement in survival compared with the standard treatment. That vaccine, called rindopepimut, primed the immune system to attack cells that express a mutated form of the EGFR protein, called EGFRvIII, that can fuel tumor progression. Glioblastoma cells often overexpress EGFRvIII.
Another vaccine, DCVax-L, is currently being tested in a phase III trial of patients with glioblastoma whose tumors could be removed surgically. DCVax-L is a dendritic cell vaccine, meaning it uses immune cells known as dendritic cells, which are collected from patients and engineered to provoke an immune response against multiple proteins, or antigens, on tumors.
Preliminary data from the trial were recently published, but only included survival data for the entire population of patients in the trial, not by treatment group. According to a statement from Northwest Biotherapeutics, which makes the vaccine and funded the trial, another round of data collection from the trial is ongoing.
Dr. Sampson’s team at Duke, which developed rindopepimut, has been testing a dendritic cell vaccine engineered to target tumor cells that express antigens induced by infection with cytomegalovirus (CMV)—a highly common virus in people aged 40 and older. For reasons that are still unclear, glioblastoma cells, but not healthy brain cells, tend to express these CMV antigens, Dr. Sampson said.
The Duke researchers have reported encouraging results from several small clinical trials testing the vaccine in patients with glioblastoma. “In two or three consecutive trials, we’ve had some patients live 2 to 5 years after treatment. That’s encouraging,” he said.
Dr. Sampson has launched a small company to move the treatment vaccine forward into larger trials.
Dr. Okada said that he is particularly excited about the prospects of another type of treatment, known as oncolytic virus therapy, which are also being tested in small clinical trials of patients with glioblastoma.
Initially, oncolytic viruses were developed as therapies that directly infect and kill cancer cells. However, researchers now believe that these viruses also work much as a vaccine does: by stimulating an immune response against tumors.
In one recent trial, several patients with advanced glioblastoma who had the oncolytic virus therapy DNX-2401 injected directly into their primary tumor had their tumors shrink by at least 95%, and 5 patients lived for at least 3 years after treatment.
At a February 2018 brain cancer immunotherapy conference Dr. Okada helped organize, several research groups reported similar results from small trials of oncolytic viruses. In these early studies, approximately 20% of patients with glioblastoma were still alive 3 years or longer after treatment.
“We’re starting to see some good long-term survival,” he said.
And other findings from these early-stage studies seem to confirm that the viruses are indeed working, in part, by stimulating an immune response against tumors. In patients who responded to the treatments, Dr. Okada said, analyses of their tumor samples showed that the tumors went from having very low levels of immune cells—known as “cold” tumors—prior to treatment to having very high levels—known as “hot” tumors—after treatment.
“Clearly, some of these [therapies] are changing the cold tumor immune environment to a hot environment,” he said.
CAR T Cells: The Search for Antigens
Like dendritic cell vaccines, another form of immunotherapy being tested against glioblastoma, chimeric antigen receptor (CAR) T-cell therapy, also involves a complicated engineering process.
With this therapy, T cells are collected from the patient and modified in the laboratory to produce a specialized receptor on their surfaces that can bind to specific antigens on the surface of tumor cells. The modified T cells are then grown in the lab and infused, typically in a one-time treatment, back into the patient.
Thus far, much of the research with CAR T-cell therapy in glioblastoma has involved CAR T cells modified to recognize EGFRvIII.
To date, clinical trials of EGFRvIII-targeted CAR T cells have not shown that the treatment can shrink glioblastoma tumors or improve how long patients live. However, said Dr. Okada, there is some evidence from these trials that the treatment can generate an immune response in the brain.
A next step, he said, will entail finding ways to make the response to the EGFRvIII CAR T cells strong enough to kill tumor cells.
CAR T cells that target different antigens are also being tested. And some research groups believe that CAR T cells that recognize only a single antigen on tumor cells may not be enough.
Investigators from the Baylor College of Medicine, for example, have developed “bi-specific” and, more recently, “tri-specific” CAR T cells—that is, CAR T cells that simultaneously target two or three antigens, respectively, on tumor cells.
One of the bi-specific CAR T cells they have developed uses T cells that naturally have a strong attraction to CMV-infected cells. And, because approximately 80% of glioblastoma cells overexpress the HER2 protein (perhaps best known for its role in breast cancer), the T cells are also engineered to express a receptor that binds to HER2. The trivalent T cells are engineered to target HER2 and two other antigens commonly found on glioblastoma cells, IL13Rα2 and EphA2.
Nabil Ahmed, M.D., who has spearheaded the development of both therapies, said his team at Baylor is moving ahead with human studies using both of these multitargeted CAR T cells.
Getting Answers from Careful Studies
Whether immunotherapy has a future in the treatment of glioblastoma, and, if so, how much of one, is still uncertain, researchers working in the field seem to agree.
Dr. Gilbert said he strongly believes that any progress is going to require very careful studies that account for the inherent challenges posed by cancers that form in the brain. Dr. Okada agreed, stressing that any progress will take time and have to build on and learn from earlier work.
He pointed, for example, to a guidelines system that he and a group of neuro-oncology researchers developed to improve the assessment of immunotherapy treatment responses in patients with brain cancer. The system, called iRANO, tries to address some of the limitations of existing methods for measuring response to immune-based treatments in patients with brain tumors.
Dr. Okada said that it’s likely the criteria will undergo changes over time as more research is conducted, including tailoring the criteria to the type of immunotherapy being used. “There aren’t going to be universal, straightforward criteria for [every immunotherapy],” he said.
It’s going to take time, effort, and patience to better understand where the field is headed, Dr. Sampson agreed.
“Is there something about glioblastoma that means [immunotherapy is] not going to work,” he said. “I don’t think so. But the only way to answer that is to keep trying to figure it out.”